Can Zika virus vaccine prevents fetal malformations?
Can Zika virus vaccine prevents fetal malformations?
Can Zika virus vaccine prevents fetal malformations? ZIKA Virus (ZIKA Virus) is the pathogen of congenital Zika syndrome. Infection with this virus during pregnancy can cause teratogenesis. Professor Andreas Suhrbier’s team is developing a series of related vaccines for target groups including pregnant women and women of childbearing age.
Professor Suhrbier’s team from the Australian Institute of Infectious Diseases developed a ZIKV vaccine BinJ/ZIKA-prME using the recently developed new insect-specific Binjari virus (BinJV) chimeric flavivirus vaccine technology, and tested this vaccine The ability to prevent fetal brain infections. They published the results of this research in the journal Vaccines.
Professor Suhrbier’s team vaccinated the female mice with the ZIKV vaccine BinJ/ZIKA-prME before mating; they were injected with Zika virus at 12.5 days of pregnancy. After testing, it was found that the mice vaccinated with the ZIKV vaccine BinJ/ZIKA-prME had no infection detected in the maternal blood, placenta or fetal head. Even using the more sensitive qRT-PCR method to detect viral RNA has the same result. More importantly, vaccination with ZIKV vaccine BinJ/ZIKA-prME will not enhance the antibody dependence of dengue infection. As a result, the ZIKV vaccine BinJ/ZIKA-prME has become a candidate vaccine to prevent congenital Zika syndrome.
Experimental process and results
Effect of Vaccination BinJ/ZIKA-prME on ZIKV Virus Content in Blood of Mice
A modified circular polymerase extension reaction was used to generate the chimeric BinJ/ZIKA-prME vaccine (Figure 1a), and the BinJ/ZIKA-prME and BinJV were proliferated in C6/36 cells, and purified using sucrose buffer and tartaric acid gradient. The researchers then vaccinated the female mice with BinJ/ZIKA-prME, BinJV and phosphate buffered saline (PBS) (control group). The ZIKV-specific antibody response in the mice was detected 5 weeks after immunization (Figure 1b). It was found that specific antibodies could be detected in mice vaccinated with BinJ/ZIKA-prME vaccine. However, no specific antibodies were detected in mice vaccinated with PBS and BinJV (Figure 1c, d). One month after the inoculation, the mice and male mice were mated, and the mice were injected with subcutaneous ZIKV every day from 12.5 days to 17.5 days after pregnancy (Figure 1b). Compared with the control group vaccinated with PBS, no ZIKV virus was detected in the blood of the mice vaccinated with BinJ/ZIKA-prME, and the virus content in the blood of the mice vaccinated with BinJV was slightly lower than that in the control group (the difference was not significant).
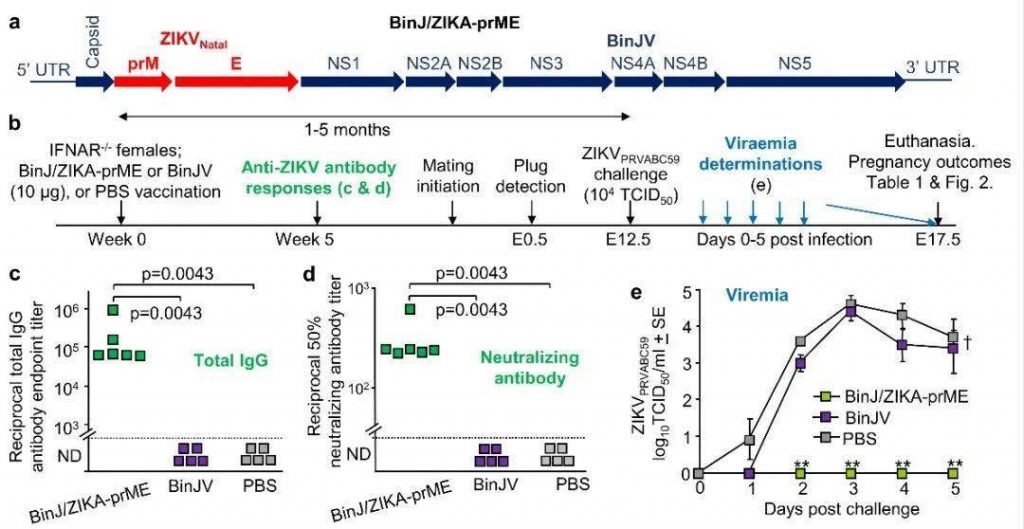
Figure 1. Experimental process and analysis of infection in pregnant mice.
Effect of vaccination BinJ/ZIKA-prME on teratogenicity of mouse fetus
Because ZIKV can cause fetal malformations. Therefore, on the 17.5th day of pregnancy, the mice were euthanized and the placenta and fetal tissues in their bodies were collected to observe and compare the abnormal conditions of the fetuses in the previous groups, as shown in Table 1. There was no significant difference in fetal abnormalities between the groups, and a small amount of placenta could not see the fetus (maybe it was absorbed).
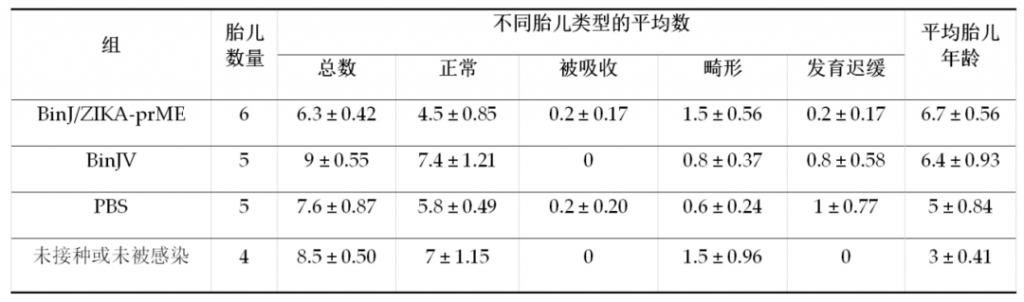
Table 1. Analysis of abnormalities in mouse fetuses.
Effect of Vaccination BinJ/ZIKA-prME on Mouse Fetal Weight
Detect and compare the collected fetal weight and ZIKV in vivo. After testing and comparison, it was found that the normal fetuses vaccinated with BinJ/ZIKA-prME weighed more than the control group and the fetuses vaccinated with BinJV (Figure 2a). Three methods were used to detect ZIKV in the fetus. High titers of ZIKV were detected in the fetuses of mice inoculated with BinJV and PBS. However, no ZIKV was detected in the fetuses of mice vaccinated with BinJ/ZIKA-prME, as shown in Figure 2b-d. Figure 2e is used to indicate that it is impossible to analyze whether the fetus contains ZIKV.
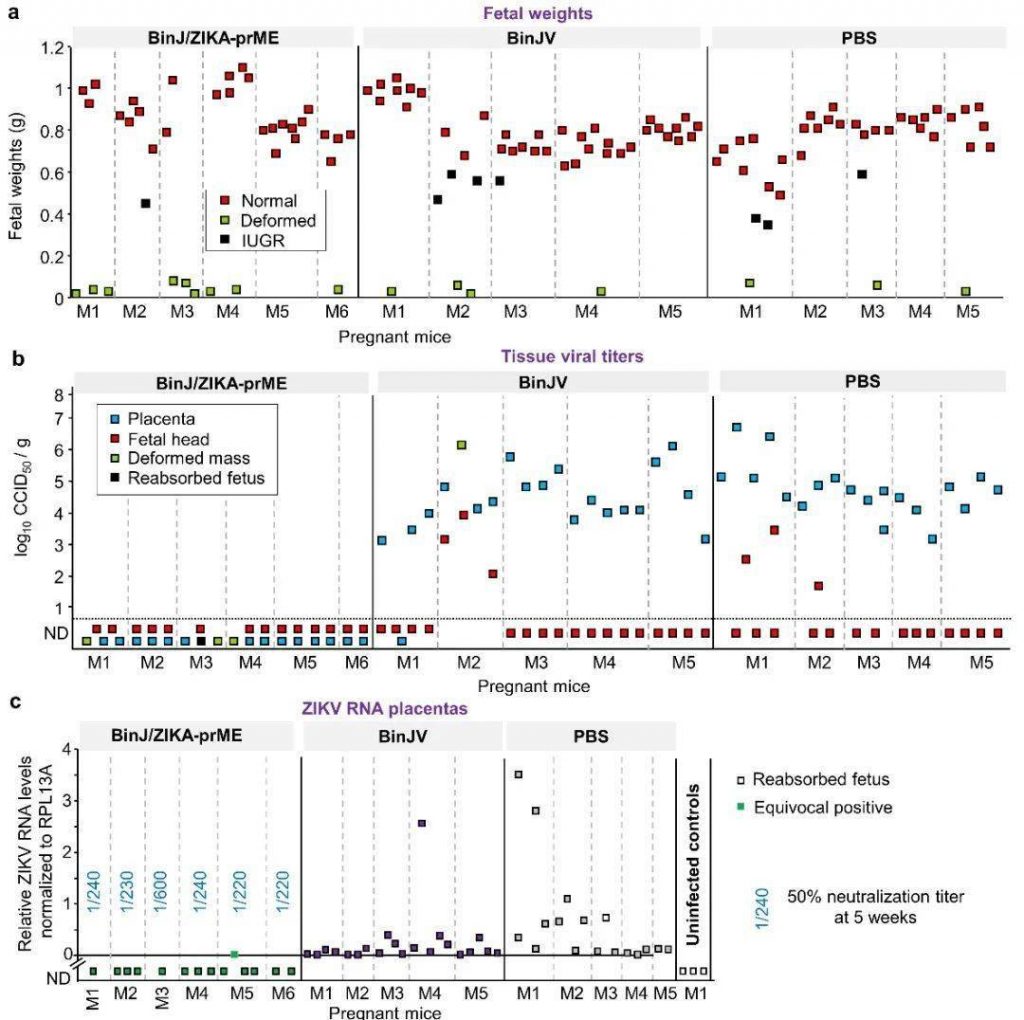
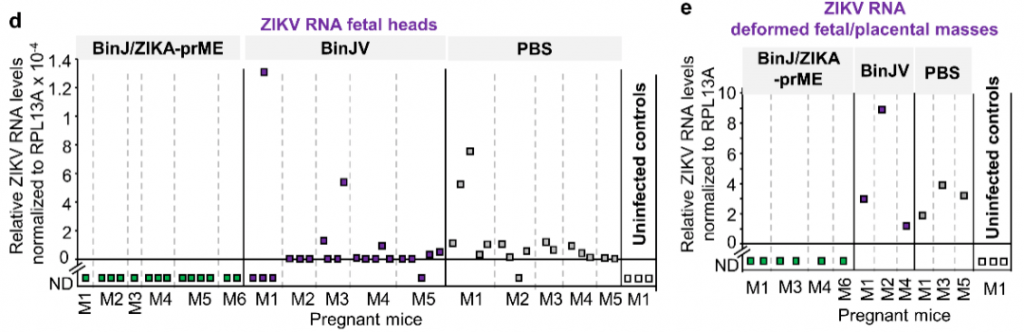
Figure 2. Analysis of fetal weight and virus status.
Antibody-dependent detection of dengue fever virus (DENV) infection
An improperly designed ZIKV vaccine can cause the antibody-dependent enhancement of Dengue Virus (DENV) infection. Using the established female mouse DENV-d220 antibody-dependent enhancement model [1], the BinJ/ZIKA-prME vaccine induces DENV. Mice vaccinated with BinJ/ZIKA-prME and PBS were injected with DENV-D220 virus, as shown in Figure 3a. It was found that mice vaccinated with BinJ/ZIKA-prME had significantly lower DENV levels than those vaccinated with PBS, as shown in Figure 3b. Antibody-dependent enhancement will obviously manifest itself in easy weightlessness. However, as shown in Figure 3c, the weight loss of the mice vaccinated with BinJ/ZIKA-prME was significantly lower than that of the control group.
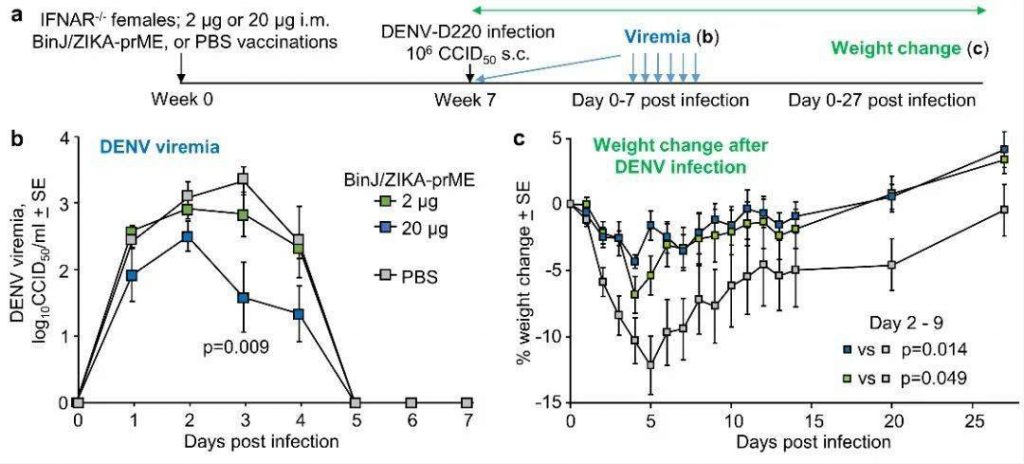
Figure 3. DENV antibody-dependent enhancement analysis.
Experimental results
Vaccination of BinJ/ZIKA-prME can not only protect pregnant mice and mouse fetuses from ZIKV infection, but also will not cause DENV antibody-dependent enhancement. Therefore, Professor Andreas Suhrbier’s team believes that the vaccine BinJ/ZIKA-prME can be used as a candidate vaccine for pregnant women with ZIKV virus.
(source:internet)
Disclaimer of medicaltrend.org



