High-precision measurement on COVID-19 nucleic acid by Digital PCR method
- Statins Lower Blood Lipids: How Long is a Course?
- Warning: Smartwatch Blood Sugar Measurement Deemed Dangerous
- Mifepristone: A Safe and Effective Abortion Option Amidst Controversy
- Asbestos Detected in Buildings Damaged in Ukraine: Analyzed by Japanese Company
- New Ocrevus Subcutaneous Injection Therapy Shows Promising Results in Multiple Sclerosis Treatmen
- Dutch Man Infected with COVID-19 for 613 Days Dies: Accumulating Over 50 Virus Mutations
High-precision measurement on COVID-19 nucleic acid by Digital PCR method
High-precision measurement on COVID-19 nucleic acid by Digital PCR method. China Guangming Daily reported on January 1st, Beijing. The reporter learned from the Chinese Academy of Metrology that the International Bureau of Weights and Measures recently announced the results of the international comparison of the new coronavirus nucleic acid measurement on its website. The comparison results show that the digital PCR method can achieve high-precision measurement of the nucleic acid content of the new coronavirus worldwide.
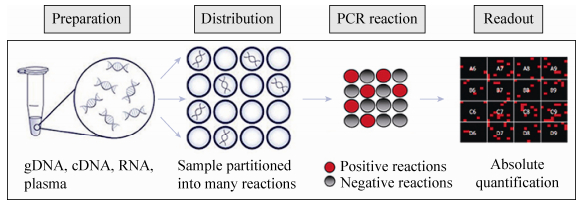
The comparison was proposed and jointly led by the Chinese Institute of Metrology, with the participation of measurement institutions in many countries around the world, which is conducive to improving the consistency and accuracy of the new coronavirus nucleic acid measurement results in countries around the world.
PCR is short for polymerase chain reaction, which is an enzymatic chemical reaction that can be used for genetic diagnosis. At present, the detection of new coronavirus nucleic acid mainly uses the fluorescent quantitative PCR method. Compared with the conventional fluorescent quantitative PCR method, digital PCR adopts single-molecule detection technology with higher sensitivity. It is an absolute quantitative technology that can directly measure the number of nucleic acid molecules. It is especially suitable for mild and asymptomatic diseases with low virus content. Quick and accurate investigation of the person.
In order to verify the measurement accuracy of the digital PCR method and the global consistency of the measurement results, the Chinese Institute of Metrology has taken the initiative to carry out an international comparison of the nucleic acid measurement of the new coronavirus, which has been highly recognized by the International Bureau of Metrology and measurement institutions in many countries, and has won the comparison. Right joint leadership.
A total of 21 internationally renowned laboratories from 16 countries participated in this international comparison. Among them, the British Government Chemist Laboratory, the Chinese Metrology Institute, the British Institute of Biological Products and the American Institute of Standards and Technology jointly act as the leading laboratories and lead Comparison work. As the joint leading laboratory, the China Metrology Institute promptly sent comparison samples to another 20 reference laboratories for free.
In this comparison, the China Metrology Institute used the self-developed digital PCR method to measure four new coronavirus RNA comparison samples, and the self-developed isotope dilution mass spectrometry method was used to measure one of the comparison samples. 4 The measurement results of this comparison sample are excellent, and the results of digital PCR method and isotope dilution mass spectrometry are in good agreement.
Moreover, the measurement results of the above-mentioned 21 reference laboratories in the world, including the China Metrology Institute, also have good consistency, achieving global equivalent. This means that the digital PCR method can achieve highly accurate measurement and international mutual recognition of the nucleic acid content of the new coronavirus worldwide.
Experts from the Chinese Institute of Metrology said that this international measurement comparison has greatly improved the consistency and comparability of the nucleic acid measurement results of the new coronavirus in countries around the world, and provided a measurement basis for jointly building a human health community.
At present, the digital PCR method and comparison results have been used by many national metrology agencies to develop new coronavirus nucleic acid standard materials, which can be used for the quality control, evaluation and traceability of nucleic acid in vitro diagnostic reagents, which can improve the accuracy of new coronavirus nucleic acid measurement It is of great significance for countries to effectively respond to the COVID-19 pneumonia epidemic.
(source:chinanet, reference only)
Disclaimer of medicaltrend.org



