In vivo gene editing therapy by virus-like mRNA technology
- Engineered Soybeans with Pig Protein: A Promising Alternative or Pandora’s Dish?
- Severe Fever with Thrombocytopenia Syndrome (SFTS): A Tick-Borne Threat with High Mortality
- Why Isolating Bananas Extends Their Shelf Life?
- This common vitamin benefits the brain and prevents cognitive decline
- New report reveals Nestlé adding sugar to infant formula sold in poor countries
- Did Cloud Seeding Unleash a Deluge in Dubai?
In vivo gene editing therapy by virus-like mRNA technology
In vivo gene editing therapy by virus-like mRNA technology. NBT : the first preclinical study of in vivo gene editing therapy based on viroid-mRNA technology.
Herpes simplex virus (HSV) is a typical representative of herpes virus, named after vesicular dermatitis that occurs in the acute phase of infection. According to the difference in antigenic characteristics, HSV is divided into two serotypes, HSV-1 and HSV-2. Humans are the natural host of herpes simplex virus (HSV). HSV infection is extremely common in the population, among which herpes simplex virus type I (HSV-1) is one of the most common human viruses in the world, and 50-90% of the world’s population has been infected with the virus. HSV virus infection is still a medical problem that has not been overcome. So far, neither vaccine is available nor medicine can cure it.
HSV-1 infection can cause a variety of diseases, including herpes simplex encephalitis. If not treated in time, the mortality rate is high. In recent years, HSV-1 infection has also been considered an important factor leading to Parkinson’s disease and Alzheimer’s disease. HSV-1 infection of the cornea can cause herpetic stromal keratitis (HSK), which is the leading cause of infectious blindness. After HSV-1 infects the cornea, it can hide in the trigeminal ganglion and form a virus reservoir. Globally, it is estimated that 1.5 million cases of corneal HSV recurrence each year, causing at least 40,000 blindness. In clinical practice, there are many cases of viral keratitis patients receiving corneal transplantation, and soon after they become transparent, HSV virus particles are released from the ganglion again, causing corneal transplantation failure.
At present, the first-line treatment for viral keratitis caused by HSV-1 infection is acyclovir (ACV). However, in the half century since the advent of acyclovir, there is still no one that can clear the cornea. And the medical technology of the virus in the trigeminal ganglion. Therefore, viral keratitis is still an incurable ophthalmic disease. Therefore, how to directly degrade the virus genome on the basis of ensuring safety, and even effectively remove the latent HSV from the root trigeminal ganglion, has become a new research direction.
At a Harvard-Boston academic event, Professor Yujia Cai, who has a background in gene editing and virology, met with Hong Jiaxu, a clinician who studies corneal diseases. After the exchanges between the two parties, they found that they were both exploring the gene editing treatment of viral keratitis. At that time, Cai Yujia was worried about clinical samples, and was not sure whether the technology could be applied to the clinic in the future, and there was no answer to how to administer the drug in the clinic; while Hong Jiaxu was worried that there was no suitable gene editing delivery system, and he was using The AAV did not achieve therapeutic effects on animals. The two parties were a little worried about competition at first, but after each went back and thought, they soon decided to join forces to overcome the medical problem of viral keratitis.
On January 11, 2021, Professor Cai Yujia from the Institute of Systematic Biomedicine of Shanghai Jiaotong University and Director Hong Jiaxu of the Eye, Ear, Nose and Throat Hospital of Fudan University jointly published an article titled: Targeting herpes simplex virus with CRISPR in Nature Biotechnology. –Cas9 cures herpetic stromal keratitis in mice research paper, reported the joint team using a new delivery technology to use CRISPR–Cas9 to target herpes simplex virus to cure herpetic stromal keratitis in mice.

The research is based on some countries’s original gene therapy delivery technology developed by Cai Yujia’s team not long ago-a virion-mRNA delivery platform (Cai Yujia’s team invented a viroid delivery between viral and non-viral vectors Technology) (VLP-mRNA). The VLP-mRNA technology has recently obtained a national patent authorization and has successfully achieved the transformation of results. Using this delivery platform, the research team conducted a preclinical study of CRISPR in-vivo gene editing to treat viral keratitis, which effectively inhibited HSV-1 virus replication, successfully cured herpes keratitis model mice, and prevented virus recurrence. The research team named the technology HELP (HSV-1-Erasing Lentiviral Particles). The HELP technology breaks through the limitations of the existing traditional small molecule drugs such as acyclovir and can directly remove the latent virus in the body, which is expected to bring a fundamental cure to patients with viral keratitis.
The research team first designed two guide RNAs (gRNAs) that target key genes in the HSV-1 life cycle, so that Cas9 can be directly cut on the HSV-1 genome, causing its degradation. In order to make gene editing safer, the researchers used a new gene editing delivery technology, namely VLP-mRNA, to deliver Cas9 mRNA. VLP-mRNA can deliver Cas9 and gRNA at the same time, which overcomes the long-term problem of low AAV delivery capacity and the safety risks caused by long-term expression of Cas9. This technology can minimize the off-target risk of CRISPR gene editing. It is currently a more advanced in vivo gene editing delivery technology and is expected to lead the clinical application of gene editing.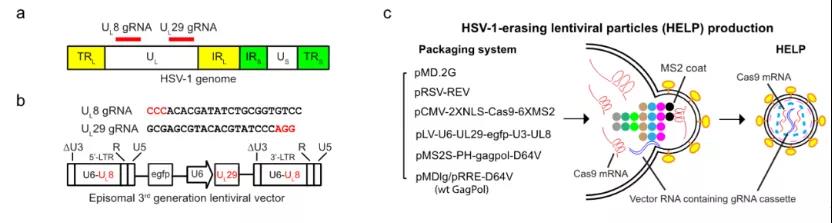
Figure 1: The working principle of HELP and its production process
In order to prove the antiviral effect of HELP, the research team of Cai Yujia and Hong Jiaxu used three different mouse infection models: preventive, therapeutic and relapsed. The researchers found that HELP effectively prevented the replication of HSV-1 and prevented the occurrence of herpetic stromal keratitis in these three different HSV infection models (Figure 2).
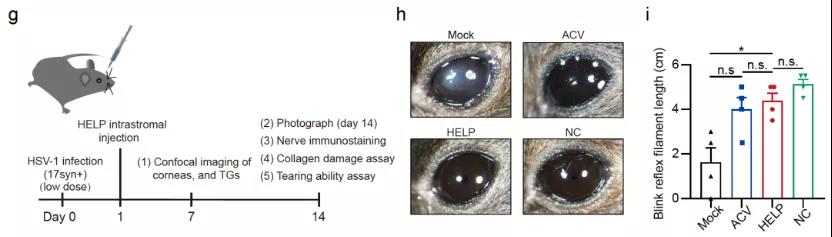
Figure 2: The therapeutic effect of HELP on the viral infection treatment model
Excitingly, the researchers found evidence that HELP can be transported retrogradely from the cornea to the trigeminal ganglion, clearing the HSV-1 virus reservoir (Figure 3). As a control, although the traditional drug acyclovir (ACV) can also inhibit virus replication in the cornea, it cannot limit the virus in the trigeminal ganglion (Figure 3). In addition, researchers have also observed that HELP can effectively eliminate the HSV-1 virus in the human cornea by using the donor cornea.
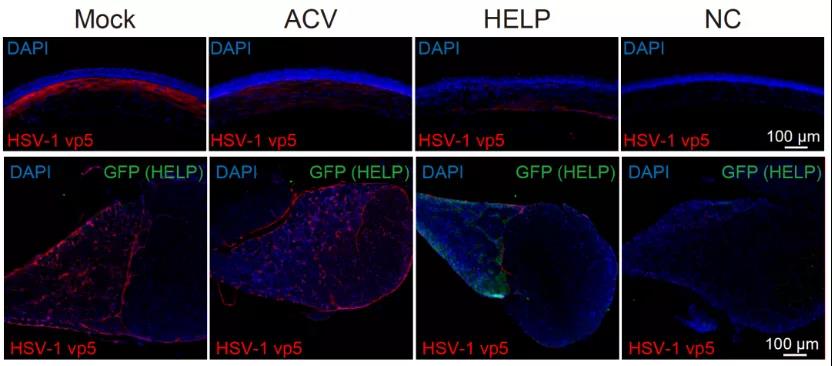
Figure 3: HELP clears the herpes virus in the cornea and trigeminal ganglia
The birth of CRISPR gene editing technology has brought new insights into the treatment of many diseases, but its potential off-target has largely limited the progress of clinical applications. Taking into account the safety issues that off-target may bring, the research team conducted a comprehensive review. Genome sequencing did not find that HELP caused off-target phenomena. This may be related to the delivery of Cas9 in the form of mRNA by HELP technology. Based on CRISPR delivered by mRNA, the gene editing enzyme Cas9 stays in the body for a short time. Therefore, gene editing is instantaneous, which can minimize the risk of off-target and reduce immune response.
It is reported that the review experts believe that the results of the work are mutually confirmed by a variety of technical means, and they rarely express high appreciation: the quality of the data is near exceptional; make an excellent contribution to the field.
Director Hong Jiaxu of the Eye, Ear, Nose and Throat Hospital of Fudan University is currently presiding over the clinical research on the application of HELP technology to refractory viral keratitis with the approval of the ethics committee.
In general, this study strongly supports the clinical potential of HELP as a new antiviral therapy and brings bright hope to patients with refractory viral keratitis.
(sourceinternet, reference only)
Disclaimer of medicaltrend.org



