Science: How does vitamin A treat digestive system diseases?
- Did Cloud Seeding Unleash a Deluge in Dubai?
- Scientists Identify Gut Bacteria and Metabolites that Lower Diabetes Risk
- OpenAI’s Model Matches Doctors in Assessing Eye Conditions
- UK: A Smoke-Free Generation by Banning Sales to Those Born After 2009
- Deadly Mutation: A New Monkeypox Variant Emerges in the DRC
- EPA Announces First-Ever Regulation for “Forever Chemicals” in Drinking Water
Science: How does vitamin A enter the intestinal immune cells and treat digestive system diseases
- Red Yeast Rice Scare Grips Japan: Over 114 Hospitalized and 5 Deaths
- Long COVID Brain Fog: Blood-Brain Barrier Damage and Persistent Inflammation
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Science: How does vitamin A treat digestive system diseases?
Vitamin A can be used to treat digestive system diseases, did you know? How does vitamin A enter the intestinal immune cells?
Introduction: Vitamin A is a fat-soluble nutrient that can be absorbed by intestinal epithelial cells and converted into retinol. Vitamin A metabolites are essential for intestinal adaptive immunity because it directs the development of B cells and T cells and promotes their entry into the intestine. Therefore, lack of vitamin A will increase the susceptibility of intestinal infections.
Immunologists and geneticists at the University of Texas Southwestern Medical Center have discovered how vitamin A enters the immune cells of the intestine. These findings can provide insights for the treatment of digestive diseases and may also help improve the effectiveness of certain vaccines. Curative effect.
The research paper was published today in “Science”, and the paper is titled “Serum amyloid A delivers retinol to intestinal myeloid cells to promote adaptive immunity”.
“Now that we have a better understanding of this important aspect of immune function, we may eventually be able to manipulate how vitamin A is delivered to the immune system for disease treatment or prevention,” the Howard Hughes Medical Institute (Howard Hughes Medical Said Lora Hooper, a researcher at the University of Texas Southwestern Medical Center and Professor of Immunology.
Dr. Hooper, a professor at the Center for Host Defense Genetics at the University of Texas Southwestern Medical Center, said that vitamin A is a fat-soluble nutrient that the human body converts into a molecule called retinol before using it, and then converts it into a molecule called retinol.
As retinoic acid, vitamin A is very important to every tissue of the human body. It is especially important for the adaptive immune system. The adaptive immune system is part of the broader immune system. It responds to specific pathogens based on immune memory, which is formed through exposure to diseases or vaccines.
Dr. Hooper said that although researchers know that some intestinal immune cells called bone marrow cells can convert retinol into retinoic acid, how they can obtain retinol to accomplish this task is still a mystery. Her laboratory studies how gut bacteria affect the physiological habits of humans and other mammalian hosts.

The lead author, Dr. Ye-Ji Bang, is a postdoctoral researcher in Hooper’s laboratory. He and his colleagues focused on serum amyloid A, a retinol-binding protein that is produced by some organs during infection. They used biochemical techniques to determine the cell surface proteins to which they were attached and identified low-density lipoprotein (LDL) receptor-related protein 1 (LRP1).
LRP1 was discovered more than 30 years ago by Joachim Herz, MD, a researcher at the University of Texas Southwestern Medical Center. He is the director of the Neurodegenerative Transformation Research Center and a professor of molecular genetics, neurology and neuroscience.
Herz laboratory mainly studies the molecular mechanisms by which members of the LDL receptor gene family act as signals and endocytosis receptors in the brain and blood vessel walls. The discovery of LDL receptors at the University of Texas Southwestern Medical Center in the United States earned Michael Brown and Joseph Goldstein, MD, the Nobel Prize in Physiology or Medicine in 1985.
Dr. Bang, Dr. Hooper, Dr. Herz, and his colleagues found that LRP1 is present on the intestinal myeloid cells and seems to be able to transport retinol to and from. Dr. Hooper said that when researchers used genetic technology to delete this receptor gene in mice to prevent their bone marrow cells from taking vitamin A derivatives, the adaptive immune system in their intestines almost disappeared, T cells and B cells.
Cellular and the key component of adaptive immunity-molecular immunoglobulin A is significantly reduced. The researchers then compared the response of mice with LRP1 and mice without LRP1 to Salmonella infection. Those mice without LRP1 were quickly overcome by infection.
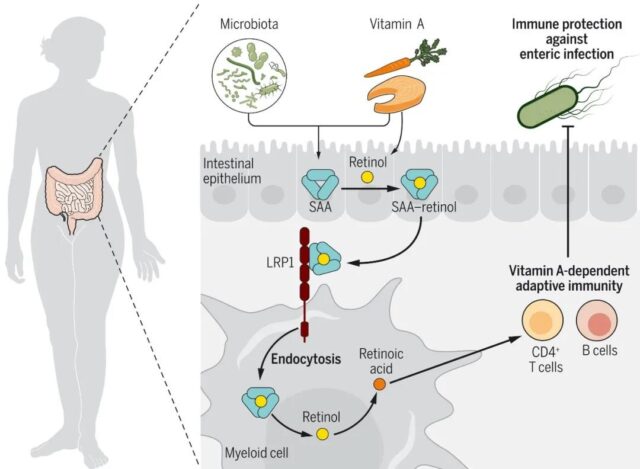
The results of the study published in “Science” show that LRP1 is the carrier of retinol into bone marrow cells. Dr. Hooper said that if researchers can develop a way to inhibit this process, they can inhibit the immune response to inflammatory diseases that affect the intestines, such as inflammatory bowel disease and Crohn’s disease.
Alternatively, finding a way to enhance the activity of LRP1 can enhance immune activity and make oral vaccines more effective.
Reference:
https://www.science.org/doi/10.1126/science.abf9232
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



