Addressing the failure of CAR-T and CAR-NK cell therapy in solid tumors
- Aspirin: Study Finds Greater Benefits for These Colorectal Cancer Patients
- Cancer Can Occur Without Genetic Mutations?
- Statins Lower Blood Lipids: How Long is a Course?
- Warning: Smartwatch Blood Sugar Measurement Deemed Dangerous
- Mifepristone: A Safe and Effective Abortion Option Amidst Controversy
- Asbestos Detected in Buildings Damaged in Ukraine: Analyzed by Japanese Company
Addressing the failure of CAR-T and CAR-NK cell therapy in solid tumors
- Red Yeast Rice Scare Grips Japan: Over 114 Hospitalized and 5 Deaths
- Long COVID Brain Fog: Blood-Brain Barrier Damage and Persistent Inflammation
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Addressing the failure of CAR-T and CAR-NK cell therapy in solid tumors.
The advent of engineered adoptive cell therapy (ACT) has changed the treatment landscape for hematological tumors and has brought hope for the treatment of solid tumors.
However, the application of cell therapy in solid tumors is hampered by poor tumor migration, penetration of physical barriers, and active tumor suppression.
Therefore, precise directing of endogenous or adoptively transferred lymphocytes into solid tumor masses is essential for optimal antitumor efficacy, while also improving patient safety.
Traditional therapies, including chemotherapy and radiation, have been shown to stimulate T-cell migration into tumors to enhance treatment outcomes, while radiation can have distant effects on distant metastases.
Similarly, intratumoral injection of pathogen-associated molecular patterns or oncolytic viruses directly induces inflammatory events within the tumor, stimulating systemic and local events at the tumor site to encourage lymphocyte migration and antigen-specific antitumor responses.
Furthermore, antigen recognition itself is a major stimulator of migration.
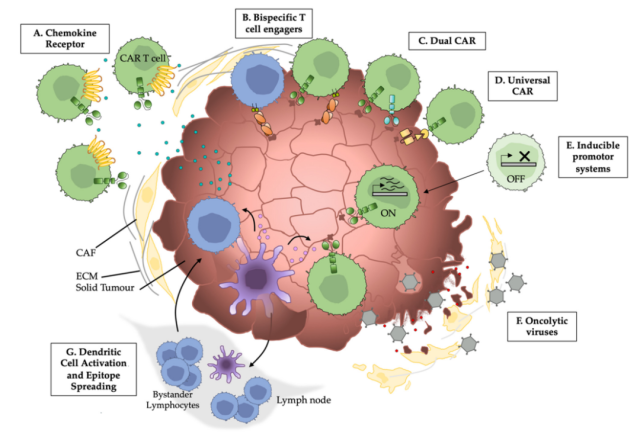
To date, many explorations and attempts have been made, and many strategies have emerged that can be used to enhance the migration and infiltration of adoptively transferred cells, including CAR-NK and CAR-T cells, in immunotherapy of solid tumors.
Chemokines and their receptors
Chemokines are cytokines that regulate the migration and trafficking of immune cells. Chemotactic gradients are critical for the recruitment of effector cells to sites of inflammation, including the tumor microenvironment (TME) .
Chemokine expression in solid tumors is secreted by stromal cells, tumor cells, and tumor-associated immune cells to determine which immune cells are recruited to the TME to help or hinder tumor growth.
Multiple chemokine/chemokine receptor strategies have been used in immunotherapeutic T-cell preclinical studies to facilitate tumor targeting by CAR-T cells, including leveraging the CXCR3, CXCR2, CCR5, CCR2, and CCR3 axes. There is currently no approved cancer chemokine immunotherapy strategy, but multiple studies have shown good clinical potential.
CXCR3
The CXCR3 axis is a key pathway for the recruitment of immune cells in solid tumors, and CXCR3 ligands include CXCL9, CXCL10, and CXCL11. After activation, CXCR3 was induced and highly expressed on effector TH1-polarized CD4+ T cells, CD8+ T cells, NK cells, and NKT cells, CXCR3 ligand expression in tumors, elevated serum CXCR3 ligand levels, and T CXCR3 on cells enhances T cell recruitment with positive preclinical results in a range of cancers.
Tumor-local expression of CXCL11 by oncolytic vaccinia virus in a mouse model of mesothelioma successfully increases the transport of endogenous cytotoxic T lymphocytes to the tumor and induces systemic antitumor immunity, highlighting the role of the CXCR3 axis on lymphocytes The importance of cell migration.
Furthermore, the use of epigenetic modulators (DNA methylation and H3K27 trimethylation inhibitors) restored tumor expression of CXCL9 and CXCL10, increased CAR-T cell efficiency in mouse models of ovarian cancer, and improved T Cellular infiltration and tumor growth control.
In a mouse model of myeloma, co-injection of two oncolytic adenoviruses encoding CXCL10 and IL-18 into established tumors resulted in smaller tumor growth and complete tumor regression in 80% of mice.
CXCR2
Regulation of CXCR2 expression on T cells has been shown to improve therapeutic efficacy in a range of tumor models. CXCR2 is specific for ligands overexpressed on a variety of tumors, including CXCL1, CXCL2, CXCL5, and CXCL8. These ligands are also expressed by various infiltrating immune cells and are generally considered to be tumor-promoting chemokines.
For example, CXCL8 can directly promote invasion and metastasis, and can promote a pro-metastatic niche by recruiting granulocyte-derived suppressor cells and neutrophils.
One strategy to exploit the expression of tumor-promoting cytokines is to ectopically express their receptors, such as CXCR2, on desirable effector cells, thereby targeting them to tumors.
Expression of CXCR2 in primary T cells enhanced T cell accumulation in tumors in a mouse model of melanoma and improved tumor regression and survival.
An ongoing phase I/II trial aimed at treating patients with metastatic melanoma with CXCR2-transduced tumor-infiltrating lymphocytes followed by high-dose IL-2 has not yet reported clinical results (NCT01740557) . CXCR2 expression also shows promise in CAR-T cell therapy.
In a hepatocellular carcinoma tumor model, enforced expression of CXCR2 on CAR-T cells increased intratumoral migration and accumulation, improving therapeutic outcomes. Similarly, ectopic expression of CXCR1 or CXCR2 enhanced metastasis to tumors and induced tumor regression and improved survival in glioblastoma, pancreatic, and ovarian cancer models.
CCR4
The receptor CCR4 is low expressed on CD8+ T cells, and its ligands are CCL22 and CCL17. In a mouse model of Hodgkin lymphoma, retroviral transduction of anti-CD30 CAR-T cells enhanced CCR4 expression, enhanced in vitro migration to lymphoma cells, and enhanced the efficacy of CAR-T cell therapy.
Although this study used a blood cancer model, this approach can also be applied to solid tumors, as upregulation of CCL22 and/or CCL17 expression was found in gastrointestinal, ovarian, and pancreatic cancers.
In a mouse model of pancreatic cancer, retroviral transduction of antigen-specific cytotoxic T cells with CCR4 enhanced migration to the tumor site and eliminated tumors in 40% of mice.
Interestingly, studies have shown that CCR4 modification enhances the interaction with DCs and enhances the binding of T-cell LFA-1 to DC ICAM-1. Therefore, this strategy has the potential not only to enhance tumor infiltration, but also to enhance the activation and support of immune cells within the tumor.
CCL2
CCL2 is secreted by a range of tumors and tumor-supporting immune cells, and induces the migration of tumor-promoting immune cells such as macrophages, TH2, and regulatory T cells. However, its cognate receptor CCR2b is only weakly expressed on activated T cells. Translocation to CCL2-secreting neuroblastoma was enhanced 10-fold on CCR2b-expressing GDH-CAR-T cells compared with CAR-T cells not expressing CCR2b, and increased relative antitumor activity. However, targeting the CCL2 pathway may have some unintended toxic consequences, as CCL2 is expressed in a range of non-tumor cell types, including endothelial cells, smooth muscle, and fibroblasts, and has been implicated in a variety of diseases, such as rheumatoid arthritis, Asthma, inflammatory bowel disease and SARS-CoV-2 infection. Considering that the level of CCL2 produced by tumors is higher than that of normal tissues, CCR2b-transduced CAR-T cells still need to be evaluated experimentally.
CXCR4
The chemokine receptor CXCR4 is widely expressed in a variety of hematological and solid tumors. Its binding to the corresponding ligand CXCL12 (SDF1) promotes tumor cell proliferation, survival and metastasis. Inhibition of CXCR4 resulted in enhanced T cell and NK cell infiltration, along with reduced recruitment of cancer-associated fibroblasts (CAFs) . Therefore, inhibition of CXCR4 is a potential strategy to enhance the infiltration of CAR-T and CAR-NK cells to tumor sites.
Recent studies have shown that the combined use of anti-EGFRvIII CAR-T cells and the PARP inhibitor olaparib improves efficacy. Olaparib reduces CAF secretion of CXCL12, thereby reducing the migration and infiltration of MDSCs, and improving the efficacy of CAR-T cell therapy in a mouse model of breast cancer.
CCL5
CCL5 is recognized by a series of chemokine receptors that are expressed on activated T cells, such as CCR1, CCR3, and CCR5, making CCL5 a good candidate for tumor-specific expression to promote adoptive-transferred lymphocyte migration. CCL5-armed oncolytic virus and CAR-T cell therapy have been successfully used in neuroblastoma mouse models.
Harnessing the CCL5 axis has also been shown to improve efficacy in a preclinical mouse model of NK cell therapy. Injected NK cells were engineered to overexpress CCR5, and intratumoral injection of CCL5-carrying vaccinia virus resulted in increased NK cell infiltration and tumor regression.
Although modulation of chemokines has shown promise in immunotherapy models, serious application problems remain that may limit their efficacy.
An important consideration is that there may be nonspecific recruitment of most chemokines.
For example, although CCL5 can enhance antitumor immune responses by recruiting CCR5+ cells such as T, NK, and DC cells, it can also recruit monocytes, macrophages, and regulatory T cells, and promote cancer invasion.
In conclusion, the role of selected chemokines needs to be clarified to prevent accidental recruitment of suppressor cells to tumors.
Oncolytic virus
Oncolytic viruses (OVs) have important immunomodulatory effects and have the potential to be used in combination strategies with adoptive cell therapy. The ability of OVs to modulate the TME and influence the host antitumor immune response provides a strong rationale for using OVs to target CAR-T cells for tumor therapy.
Mechanistically, OVs induce immunogenic cell death of tumor cells, release damage- and pathogen-associated molecular patterns (DAMPs and PAMPs) , tumor-associated antigens or neoantigens, and induce cytokine and chemokine expression at tumor sites.
OVs can induce the expression of the major histocompatibility complex (MHC) on tumor cells , thereby increasing antigen presentation.
Furthermore, OV-mediated TME remodeling affects innate and adaptive immune cell function, enhances dendritic cell activation, lymphocyte infiltration and activation, and stimulates epitope spreading.
In addition to the natural immune regulation of OVs, their genetic modification allows tumors to locally express a broad range of polypeptides or noncoding RNAs to further enhance the tumor’s inflammatory profile, supporting immune cell migration and activation.
In a series of preclinical studies, OVs carrying an array of cytokines, chemokines, and bispecific T-cell engagers have been used to remodel the TME and enhance the efficacy of CAR-T cell therapy.
TILT-123, an oncolytic adenovirus carrying TNF-α and IL-2, has the potential to be a combination strategy with CAR-T cells.
Showing compelling synergy with PD-L1 inhibition in a range of preclinical models, TILT-123 is currently enrolling in two clinical trials for clinical investigation as a single agent or in combination with tumor-infiltrating lymphocytes in metastatic melanoma (NCT04695327, NCT04217473) .
Another use of OV and CAR-T cell therapy is the use of transgenic OVs to induce target antigen expression in tumors.
This principle has been demonstrated in preclinical studies, where CD19 antigen is displayed on the surface of solid tumor cells through OV vectors, enabling effective targeting by anti-CD19 CAR-T cell therapy.
There are currently two Phase I clinical trials (NCT01953900, NCT03740256) that are testing the synergistic effect of oncolytic virus and CAR-T cells in the treatment of solid tumors.
Tumor-inducing promoter
In addition to constitutive expression of genes that have been used to enhance CAR-T cell therapy, tumor-targeted genes can also be specifically expressed through the use of inducible promoter systems.
Triggering the site-specific release of inflammatory mediators , including chemokines, by tumor antigens is a useful way to encourage the migration of other CAR-T cells and bystander cells to the tumor.
Inducible promoters activated by recognizing tumor antigens, either through CARs or TCRs in T cells or TILs, have been used to overexpress a variety of genes, especially antitumor cytokines, and have been shown to improve safety At the same time, the anti-tumor effect was enhanced.
IL-12 has been shown to enhance CAR-T cell activity in vivo, but the toxicity of IL-12 is also evident. Preclinical testing of targeted expression mainly involves the IL-2-based NFAT promoter to target IL-12 expression to tumor sites.
Using these antigen-triggered promoters to fix IL-12 expression at the tumor site improves tumor killing by CAR-T cells in vivo and reduces the number of CAR-T cells required for effective tumor killing.
This strategy minimizes the systemic toxicity observed with IL-12 expression. Local expression of IL-12 by CAR-T cells has also been shown to activate innate immune cells at the tumor site, such as macrophages, to help eliminate antigen-negative tumor cells.
NFAT-inducible promoters also showed advantages in non-CAR-ACTs, such as TCR and TIL-ACT. TILs selectively expressing IL-12 at the tumor site produced enhanced antitumor immune responses compared to non-engineered TILs and required 10- to 100-fold lower doses in a clinical trial in metastatic melanoma (NCT01236573) .
Although some toxicity has been observed, the inducible system it uses shows potential for application in human therapy.
The development of promoters that respond to signals other than TAAs is another growing area of research that can exploit alterations in tumor metabolite profiles.
A study has developed a hypoxic tumor microenvironment sensor using the hypoxia-inducible element within the CMV promoter.
The “HiTA system” restricts CAR-T cell gene expression to a hypoxic environment while ignoring normoxic tissues that express TAA. Hypoxia-inducible expression systems can also be used to express other transgenes or the CAR itself to focus and enhance immune responses.
Modification of CAR Structure
The selection and design of CAR structures are key considerations for optimizing migration and antitumor activity. Systems such as Dual CARs and Universal CARs (UniCAR) are effective approaches to improve the tumor targeting and safety of CAR-T cells.
Dual CAR-T cells express CARs targeting different antigens, and can express more than one CAR on a single T cell or T cells transduced with two groups of different CAR structures to target multiple antigens. These approaches have been successfully applied in the clinical treatment of a range of in vivo solid tumors with promising results in clinical trials.
For example, by targeting CD38 and BCMA in vivo, the combination of two independent CAR-T cells has successfully treated multiple myeloid cancer in clinical, and the objective response rate reached 87.5%.
After nearly 1 year of follow-up, 76.9% of patients did not experience recurrence transfer.
In addition, a dual-target CAR-T for CD19/CD22 has also entered clinical trials (NCT03233854) .
Early results showed that 88% of patients had a successful response, and all had complete remission; of the 21 LBCL patients, 62% had a response and 29% had a complete remission.
UniCAR creates a safety “switch” for clinical control of immune responses by introducing a second component, called a targeting module (TM) .
TMs are fusion protein molecules of UniCAR-T cell target antigen and TAA binding domain (such as single-chain antibody) . TMs link CAR to TAA. Due to the short half-life of TM (15–45 minutes) , removing TM can quickly eliminate UniCAR-T cell activity.
Currently, several UniCAR systems have entered clinical trials, almost all of which focus on hematological malignancies targeting a range of targets, including CD19 (NCT02808442 and NCT02746952) , CD22 (NCT04150497), and CD123 (NCT03190278) .
While the CD19 trial was largely successful, with 67% of patients achieving complete remission, other trials, such as the CD123 UniCAR trial (NCT04106076) , have encountered safety concerns. For solid tumors, only a few UniCAR clinical trials are registered, such as one for PMSA in prostate cancer (NCT04633148) .
Targeting the tumor microenvironment
Cancer-associated fibroblasts are a phenotypically heterogeneous cell population that builds and remodels the TME extracellular matrix (ECM) .
Depleting or altering the function of CAFs is currently a well-studied area and has the potential to be an attractive option for augmentation of ACT and immunotherapy.
Since CAFs contribute to the ECM/cell barrier, reducing their number and activity has the potential to synergize with CAR-T/NK cell therapy to increase lymphocyte migration to tumors.
Several CARs have been developed that exert antitumor effects by targeting fibroblast activation protein alpha (FAP-α) to directly deplete CAFs.
Anti-FAP-α CAR-T cell therapy has shown convincing efficacy in preclinical models of various solid tumors.
There are two clinical trials utilizing anti-FAP-α CAR-T cells, a completed Phase 1 clinical trial in pleural mesothelioma (NCT01722149) and a Phase 1 clinical trial utilizing a fourth-generation CAR (NCT03932565) .
A different strategy is to remove immunosuppressive cells such as MDSCs and TAMs. In a mouse model of neuroblastoma, removal of tumor-localized MDSCs enhanced CAR-T cell efficacy and tumor infiltration.
Increased infiltration of endogenous tumor-specific CD8+ T cells was found by depletion of TAMs using antifolate receptor β-CAR-T cells.
TAM-targeted CAR-T cell pretreatment enhanced the efficacy of tumor-specific anti-mesothelin CAR-T cells, resulting in tumor regression and prolonged survival.
In addition, solid tumors often exhibit metabolic disturbances that lead to the release of immunosuppressive metabolites, including lactate, adenosine, and reactive oxygen species (ROS) , which can effectively inhibit lymphocyte migration.
The combination of CAR-T/NK cell therapy with tumor metabolism inhibitors may enhance their migration to tumors and their killing effects on tumor cells.
Enhance DC cell activity
Dendritic cells (DCs) contribute to the homeostasis, activation and migratory potential of the T cell compartment.
Concurrent with adoptive T cell transfer, the use of strategies to stimulate and enhance endogenous DC activity will maximize T cell engagement and activation, encourage epitope spreading, and alter the TME in a way that is more conducive to local and systemic immune activity.
Flt3L is a key growth factor for mobilizing and expanding DC cells, and the secretion of Flt3L by engineered CAR-T cells increased DC precursor cells in bone marrow and tumors, increased intratumoral DC secretion of IL-12 and TNF, and suppressed tumors grow.
Crucially, CAR-T cells secreted Flt3L while promoting tumor infiltration of endogenous CD8+ T cells and leading to epitope spreading, indicating that the use of Flt3L activates host DC cells and enhances lymphocyte migration into solid tumors.
Summary
CAR-T and CAR-NK cell therapy is a very promising approach to improve the prognosis of cancer patients. Although they are effective against hematological malignancies, overcoming solid tumors remains difficult.
More recently, drugs that stimulate migration, improve the tumor microenvironment, and remove tumor barriers have been combined with CAR-T/NK-based therapy to improve the disappointing performance of CAR-T and NK cells in solid tumors.
These strategies will provide critical avenues for advancing engineered T cell and NK cell therapy against solid tumors.
Reference :
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8870056/
Addressing the failure of CAR-T and CAR-NK cell therapy in solid tumors
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



