HER2 Exon 20 YVMA Insertion Mutant Organoid Model
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
HER2 Exon 20 YVMA Insertion Mutant Organoid Model
HER2 Exon 20 YVMA Insertion Mutant Organoid Model. The research of HER2 targeted drugs is full of opportunities and challenges, and the further development of new therapeutic drugs is imminent.
Human epidermal growth factor receptor 2 (Her-2) belongs to the ERBB receptor tyrosine kinase family. The family includes four members ERBB1, ERBB2, ERBB3 and ERBB4. Among them, ERBB2 is also called HER2. When it is combined with other ERBB family members, it forms a heterodimer. After the ligand binds to HER2, HER2’s own tyrosine kinase is activated, which in turn phosphorylates its substrate and activates downstream signal path.
Introduction to HER2 gene mutation and Current status of targeted drug therapy
HER2 gene mutation is a carcinogenic driver of lung cancer, which is mainly seen in women, non-smokers and patients with lung adenocarcinoma. It can also be found in other tumors, including breast cancer, ovarian cancer, bladder cancer, salivary gland tumors, endometrial cancer, pancreatic cancer, etc. Among them, the insertion mutation in exon 20 of the HER2 gene is the most common mutation type in HER2 mutant non-small cell lung cancer. It is more common in p.A775_G776insYVMA, and also includes p.G776>VC, p.P780_Y781insGSP, p.V777_G778insCG, p.M774delinsWLV , P.G776>LC and point mutations p.L755S, p.G776C and p.V777L.
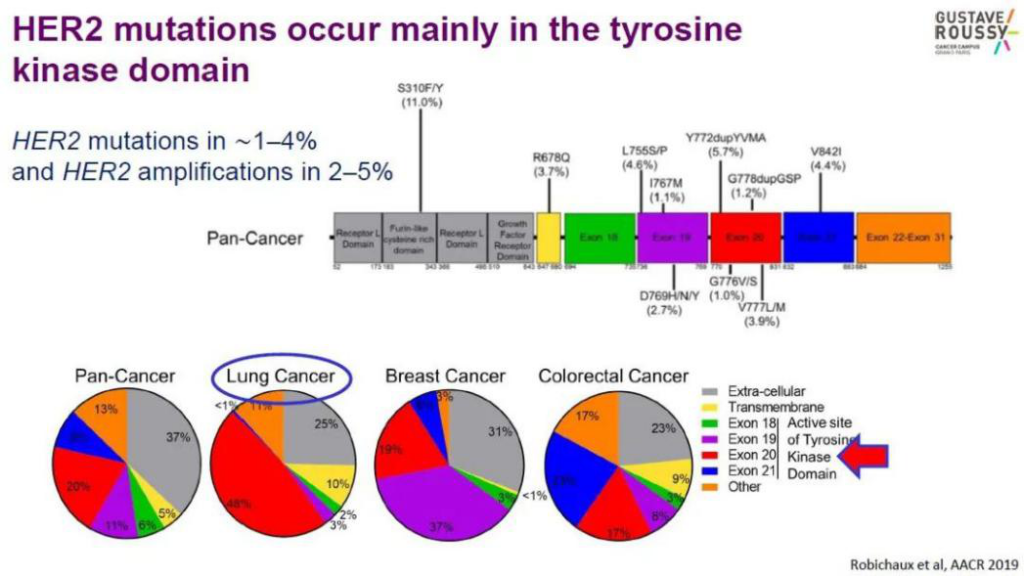
At present, there is no standard treatment plan for HER2 gene mutant non-small cell carcinoma. According to reports, the drug treatments targeting HER2 mutations are shown in Table 1:
Table 1 Therapeutic effects of drugs targeting HER2 mutations
|
name |
Test target |
ORR |
Median progression-free survival |
|
TDM-1 |
HER2 mutant NSCLC |
44% |
5 months |
|
Pozitinib |
HER2 20 exon insertion mutation NSCLC |
50% |
5.1 months |
|
Afatinib |
G778_P780dup and G776delinsVC mutant subtypes |
40% |
7.6 months |
|
Dacomitinib |
HER2 exon 20 mutation |
12% |
no |
|
TAK-788 |
EGFR exon 20 insertion mutation |
43% |
7.3 months |
|
DS-8201 |
HER2 mutant breast and stomach cancer |
61.9% |
14 months |
Previous studies have found that the median overall survival of patients with HER2 mutant advanced NSCLC is less than 2 years. Since the existing targeted drugs are not developed for mutant HER2, the clinical results of the treatment effect for a wide range of HER2 mutant lung cancers are quite different. . Therefore, in non-small cell lung cancer, the research of HER2 targeted drugs is full of opportunities and challenges, and the further development of new therapeutic drugs is imminent.
HER2 Exon 20 YVMA , Insertion mutant organoid model
On December 31, 2018, Professor Zhou Caicun and Ren Shengxiang from Shanghai Pulmonary Hospital Affiliated to Tongji University and Professor Ji Hongbin from Shanghai Institute of Biochemical Cell Research, Chinese Academy of Sciences collaborated in an article published online on “AnnalsofOncology”. The researchers successfully used the established The HER2-20 exon A775_G776YVMA insertion mutant lung adenocarcinoma organoids (organoids) model and human tumor xenograft (PDX) models have verified the anti-tumor activity of pyrrotinib, and are also in follow-up clinical trials (showing that pyrrotinib’s ORR 53.3%, median PFS was 6.4 months, median DOR was 7.2 months) further proved the correctness of the test, indicating that tumor organoids in vitro drug sensitivity test has a strong correlation with the results of clinical trials.
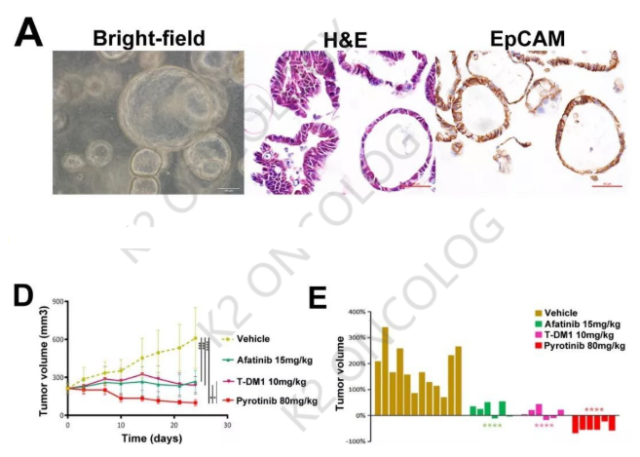
Recently, Ketu Medical established a HER220 exon YVMA insert organoid model using samples of malignant pleural and ascites fluid. The mutation abundance reached 45.05%. It successfully maintained the gene mutation characteristics in the original tumor cells, which can be used for the majority of pharmaceutical companies and scientific research. The institute provides integrated CRO services such as HER2 targeted drug screening, new drug development and targeted therapy. Part of its product information is as follows:
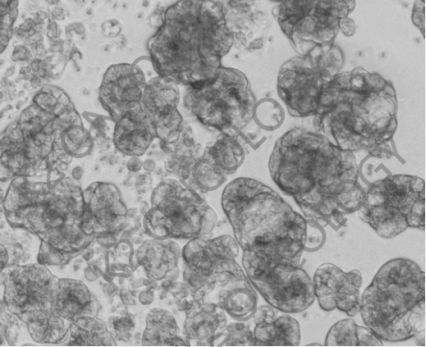
HER220 Exon YVMA Brightfield photos of introgression organoids:
HE staining results:
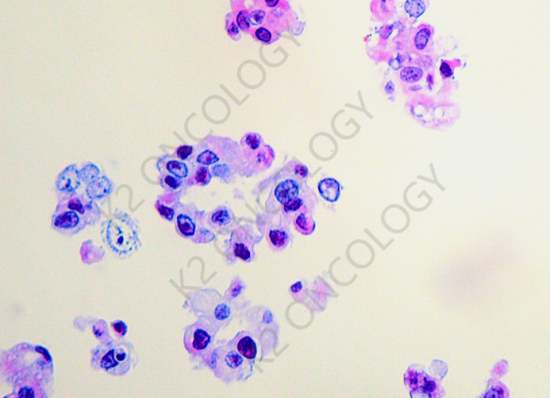
Tumor cells isolated from malignant pleural effusion
Organoids (P3 generation)
Sanger sequencing:
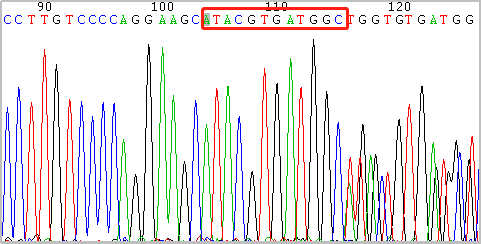
Drug testing experiment:
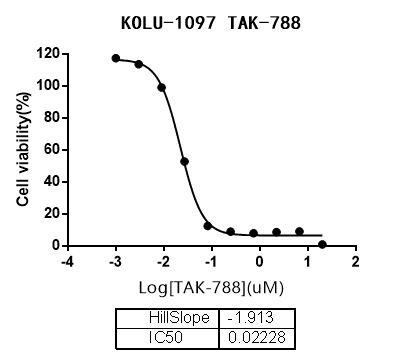
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



