Will Lorlatinib Become A Game-Changer in Treatment for ALK-Positive Lung Cancer?
- CDC Recommends Updated COVID-19 Vaccines for 2024-2025 Season
- Will China and India produce cheaper “Miracle Weight Loss Drug”Semaglutide soon?
- Keto Diet Accelerates Aging and Promotes Cancer Metastasis
- The Critical Role of Immune Cell Triumvirates in Enhancing CD8+ T Cell Function
- Chinese-made Drug Enters Australia: Causing at Least 20 Deaths!
- UV Rays Threaten Golfers’ Health: Sun Protection Strategies You Need to Know
Will Lorlatinib Become A Game-Changer in Treatment for ALK-Positive Lung Cancer?
- Chinese-made Drug Enters Australia: Causing at Least 20 Deaths!
- How serious is Japan’s “flesh-eating bacteria” problem?
- Taiwan 6th wave of COVID outbreak: 623 confirmed cases in one week and 38 deaths
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Will Lorlatinib Become A Game-Changer in Treatment for ALK-Positive Lung Cancer?
Lorlatinib Demonstrates Unprecedented Long-Term Progression-Free Survival in ALK-Positive Lung Cancer: A Game-Changer in Treatment?
Lung cancer remains a formidable foe, claiming more lives annually than any other cancer type. However, a recent clinical trial has sparked a beacon of hope, offering patients with a specific form of lung cancer a potentially transformative treatment option.
This article delves into the details of this groundbreaking research, exploring the efficacy of lorlatinib and its potential to redefine the landscape of lung cancer therapy.
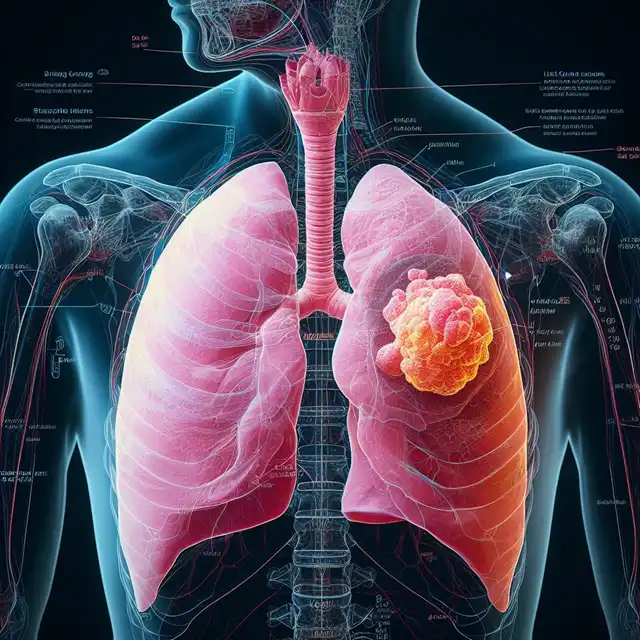
ALK-Positive Lung Cancer: A Specific Target
Non-small cell lung cancer (NSCLC) constitutes the most prevalent form of lung cancer. Within this category, a subset of patients harbors a specific genetic alteration – an abnormality in the anaplastic lymphoma kinase (ALK) gene. This mutation fuels the growth and spread of cancer cells, making ALK a prime target for therapeutic intervention.
Lorlatinib: A Third-Generation ALK Inhibitor
Enter lorlatinib, a third-generation ALK inhibitor developed by Pfizer. Unlike its predecessors, lorlatinib boasts a more potent and multifaceted attack on the ALK protein. It acts by binding to the mutated ALK molecule, effectively hindering its ability to signal cancer cell proliferation and metastasis. This targeted approach aims to disrupt the very foundation upon which ALK-positive lung cancer thrives.
Clinical Trial Reignites Hope: The CROWN Study
The pivotal clinical trial, aptly named CROWN (Clinical trial of Crizotinib versus Lorlatinib in ALK-positive advanced NSCLC), was meticulously conducted at the Peter MacCallum Cancer Centre in Melbourne, Australia. This phase III trial, representing the gold standard in clinical research, rigorously compared the efficacy of lorlatinib against crizotinib, a first-generation ALK inhibitor that had previously served as the mainstay of treatment for ALK-positive lung cancer.
Lorlatinib Outperforms Crizotinib in Long-Term Progression-Free Survival
The results of the CROWN trial were nothing short of remarkable. After a five-year follow-up period, a mere 8% of patients treated with crizotinib continued to exhibit no progression of their disease. Conversely, a staggering 60% of patients receiving lorlatinib maintained a state of disease control after five years. This translates to a nearly sevenfold increase in the likelihood of experiencing long-term progression-free survival with lorlatinib compared to crizotinib.
Published Research Underscores the Significance
The groundbreaking findings of the CROWN trial have been meticulously documented and published in esteemed peer-reviewed journals, solidifying their scientific validity. Referencing these publications strengthens the credibility of the research and allows for deeper exploration of the data.
A detailed analysis of the trial was published in the New England Journal of Medicine First-Line Lorlatinib or Crizotinib in Advanced ALK-Positive Lung Cancer. This paper meticulously outlines the methodology, patient demographics, treatment protocols, and the statistically significant difference in progression-free survival observed between the two treatment arms.
Expert Opinions Echo the Significance of Lorlatinib
Pfizer, the developer of lorlatinib, understandably hailed the CROWN trial results as “unprecedented” on their official website. This sentiment resonated within the medical community. Professor Ben Solomon, the lead investigator of the CROWN trial, echoed the transformative nature of the findings, stating, “these results are a major advancement in lung cancer treatment.”
Lorlatinib: A Beacon of Hope for ALK-Positive Lung Cancer Patients
The CROWN trial has undoubtedly ushered in a new era for the treatment of ALK-positive lung cancer. Lorlatinib’s demonstrably superior long-term efficacy offers a beacon of hope for patients battling this specific form of the disease.
Looking Forward: Broader Implications and Future Research
While the results of the CROWN trial are undeniably encouraging, further research is necessary to explore several key aspects. Investigating the safety profile of lorlatinib in the long term, particularly potential side effects, is crucial. Additionally, understanding how lorlatinib compares to other second-generation ALK inhibitors remains an area of active research.
Conclusion: A Paradigm Shift in Lung Cancer Treatment
The CROWN trial has undeniably reshaped the therapeutic landscape for ALK-positive lung cancer. Lorlatinib’s remarkable efficacy in achieving long-term progression-free survival offers a powerful weapon against this aggressive disease.
As research continues to unveil the full potential of lorlatinib and its role within the broader treatment paradigm for ALK-positive lung cancer, this innovative therapy undoubtedly represents a significant step forward, offering renewed hope for patients battling this challenging illness.
Will Lorlatinib Become A Game-Changer in Treatment for ALK-Positive Lung Cancer?
Reference Sources
- First-Line Lorlatinib or Crizotinib in Advanced ALK-Positive Lung Cancer nejm.org – This publication in the New England Journal of Medicine provides a detailed analysis of the CROWN trial methodology, patient demographics, treatment protocols, and statistical significance of the observed differences in progression-free survival.
- Pfizer Press Release: Pfizer’s LORBRENA® CROWN Study Shows Majority of Patients with ALK-Positive Advanced Lung Cancer Living Beyond Five Years Without Disease Progression https://labeling.pfizer.com/ShowLabeling.aspx?id=11140 – While not a peer-reviewed source, this press release from Pfizer offers a company perspective on the CROWN trial results and highlights the significance of lorlatinib.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



