Pfizer mRNA COVID-19 Vaccine Will Trigger Rare Severe Allergic Reaction
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Science: Pfizer mRNA COVID-19 Vaccine Will Trigger Rare Severe Allergic Reaction
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- What is the difference between Atorvastatin and Rosuvastatin?
- How long can the patient live after heart stent surgery?
Science: Pfizer mRNA COVID-19 Vaccine Will Trigger Rare Severe Allergic Reaction.
Pfizer mRNA COVID-19 Vaccine Will Trigger Rare Severe Allergic Reaction. In the past two weeks, at least 8 people have experienced severe allergic reactions after being vaccinated with Pfizer-BioNTech’s COVID-19 vaccine.
Recently, good news about coronavirus vaccines has continued. Moderna, Pfizer and BioNTech have all announced that their mRNA vaccines have achieved 95% efficacy in clinical trials involving thousands of people.
The US Food and Drug Administration (FDA) has also approved the emergency use of these two mRNA vaccines.
However, with the large-scale start of vaccination, the fastest approved vaccine in the world began to have serious side effects that could not be ignored.
In the past 2 weeks, at least eight people have experienced severe allergic reactions after receiving the mRNA COVID-19 vaccine produced by Pfizer and BioNTech.
Researchers suspect that these allergic reactions stem from the compound polyethylene glycol (PEG) contained in lipid nanoparticles (LNP) that package mRNA.
In the last month, Pfizer’s stock price has also fluctuated.
Principle of mRNA vaccine
Both of these two mRNA vaccines contain a small piece of messenger RNA (mRNA), which is used to guide the synthesis of the spike protein (S protein) of the coronavirus and is delivered through tiny liposomes composed of lipid nanoparticles (LNP).
Lipid nanoparticle (LNP) is a lipid-based delivery system.
It is composed of cationic lipids, auxiliary lipids, cholesterol and polyethylene glycol (PEG) to form stable nanoparticles. LNP is not only used to carry mRNA, but also It protects mRNA from being degraded too quickly.
At the same time, the lipophilicity of LNP can fuse the nanoparticle with the receptor cell membrane and deliver mRNA into the cell.
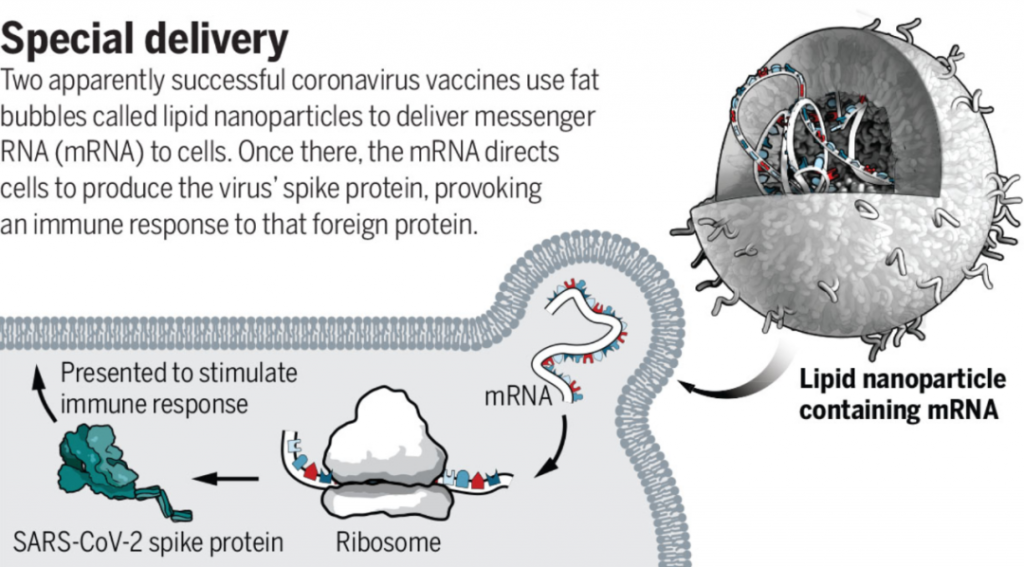 MRNA COVID-19 vaccine based on lipid nanoparticle delivery
MRNA COVID-19 vaccine based on lipid nanoparticle delivery
Is polyethylene glycol the culprit of allergies?
Prior to this, polyethylene glycol (PEG) has never been used in vaccines. Polyethylene glycol (PEG) has been found in many drugs that occasionally cause allergic reactions.
It can cause skin rashes, sudden drops in blood pressure, and shortness of breath. Symptoms such as rapid heartbeat are a potentially life-threatening reaction.
Some immunologists believe that a small number of people who have previously been exposed to polyethylene glycol (PEG) may have produced high levels of polyethylene glycol (PEG) antibodies, putting them at risk of allergic reactions to the vaccine.
According to the latest statistics from the Centers for Disease Control and Prevention (CDC) of the United States (as of December 19), there are currently 27,2001 people receiving Pfizer’s mRNA vaccine, of which 6 have severe allergic reactions.
In addition, 2 cases have occurred in the UK. In other words, in only the last two weeks, at least 8 people have developed severe allergic reactions after receiving Pfizer’s mRNA vaccine.
Any vaccine may have side effects such as allergic reactions, but it is usually extremely rare, about once per million doses.
From the current situation, the proportion of severe allergic reactions in Pfizer’s mRNA vaccine far exceeds this rate.
At present, news reports about vaccine allergic reactions have caused anxiety among the American people.
Many Americans are deeply concerned that they may not be able to receive the COVID-19 vaccine (at least these two mRNA vaccines) due to severe allergic reactions.
Due to these allergic reactions that have already occurred, the National Institute of Allergy and Infectious Diseases (NIAID) is very concerned and has held several meetings recently to discuss these allergies with representatives of Pfizer and Moderna, independent scientists and physicians, and FDA experts. reaction.
Pfizer said it is currently “actively seeking follow-up actions” and recommended that “appropriate medical treatment and supervision should be provided immediately in the event of a vaccine allergic reaction.”
Many scientists believe that polyethylene glycol (PEG) is the main culprit for allergic reactions in mRNA vaccines, but if we want to work hard to contain the current COVID-19 pandemic, we must still rely on vaccines, and humans also need to be vaccinated.
The widely used polyethylene glycol (PEG)
Polyethylene glycol (PEG) can be used as a thickener, solvent, softener, etc. in many daily products, such as toothpaste and shampoo, and has been used as a laxative for decades. More and more biopharmaceuticals also contain polyethylene glycol.
People have long believed that polyethylene glycol (PEG) is biologically inert, but more and more evidence shows that they are not.
In 2016, a study led by Samuel Lai, a pharmacological engineer at the University of North Carolina at Chapel Hill, showed that as many as 72% of people have antibodies against polyethylene glycol (PEG), which may be due to food containing polyethylene glycol ( PEG) caused by cosmetics, daily necessities or medicines. Among them, about 7% of the human body has high enough polyethylene glycol (PEG) antibody levels to easily induce allergic reactions.
However, some researchers do not believe that polyethylene glycol (PEG) is related to allergic reactions to mRNA vaccines, and believe that the risk of polyethylene glycol (PEG) is exaggerated.
But in fact, researchers from Moderna and BioNTech, which developed mRNA vaccines, have said that polyethylene glycol (PEG) has substantial shortcomings in terms of activity and safety.
Katalin Karikó, senior vice president of BioNTech, is one of the inventors of mRNA technology. She believes that due to the low dose of vaccine injection and intramuscular administration, this so-called risk is negligible.
Katalin Karikó emphasized that according to current understanding, the risk of mRNA vaccines is still very low. All vaccines have certain risks. But the benefits of vaccines far outweigh the risks.
How should we respond?
Both mRNA vaccines require two injections. Anti-polyethylene glycol (PEG) antibodies triggered by the first injection may increase the risk of allergic reactions during the second injection.
What if polyethylene glycol (PEG) is indeed the culprit?
It is obviously unrealistic to screen tens of millions of people for antibodies against polyethylene glycol (PEG) before vaccination.
The US Centers for Disease Control and Prevention (CDC) guidelines recommend that Pfizer or Moderna’s mRNA vaccine should not be administered to people who have a severe allergic reaction to any component of the vaccine.
For people who have had a severe reaction to other vaccines, the risks and benefits of vaccination should be carefully weighed.
This kind of allergic reaction happens very quickly. For those who may be at high risk of allergic reaction, they should stay at the vaccination site for 30 minutes after vaccination so that they can be treated in time when severe allergic reactions occur.
Other allergic reactions
According to statistics, after vaccinating with Pfizer and Moderna’s mRNA vaccine, 2% of vaccinators will have a severe fever of 39°C to 40°C.
Although the ratio of 2% is not high, if the two COVID-19 vaccines are approved by the regulatory authorities, their goal is to provide 35 million people worldwide with vaccination by the end of December, and 2% of them will have a severe fever, which will be 70%. Million people.
In addition to fever, other short-lived side effects may affect more people. The independent committee that conducted an interim analysis of Moderna’s large clinical trial found that serious side effects included fatigue (9.7%), muscle pain (8.9%), arthralgia (5.2%) and headache (4.5%).
In the Pfizer/BioNTech vaccine clinical trial, the proportion of side effects was even lower. Side effects included fatigue (3.8%) and headache (2%).
The proportion of these side effects is obviously much higher than that of most influenza vaccines, and it is beyond the scope of people’s habits.
Science: Pfizer mRNA COVID-19 Vaccine Will Trigger Rare Severe Allergic Reaction
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



