30 Questions about COVID-19 Vaccination
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
30 Questions about COVID-19 Vaccination
30 Questions about COVID-19 Vaccination. Is it necessary to vaccinate every year? Is it necessary to inoculate the new coronavirus vaccine? How to get the new coronavirus vaccine and where to get it?
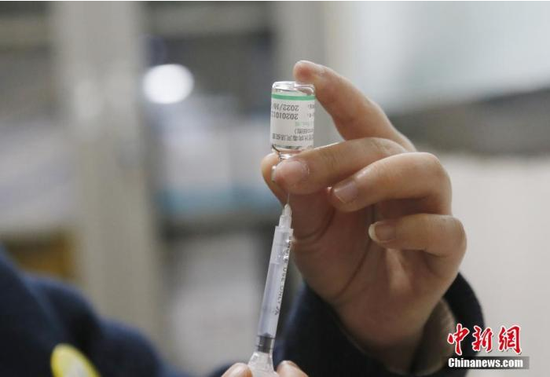
Chinese Center for Disease Control and Prevention released the “COVID-19 Vaccination Questions and Answers” on the 7th. It is mentioned in the question and answer that you should continue to wear a mask after vaccination; it is recommended to use the same type of vaccine from the same manufacturer to complete the vaccination at this stage; about two weeks after the second dose of inactivated vaccine can produce a better immune effect; for new coronavirus pneumonia For confirmed cases and asymptomatic infections, it is currently not recommended to receive the new coronavirus vaccine; those who are vaccinated do not need to be tested for the presence of antibodies before vaccination.
On January 7, at the First People’s Hospital of Xining City, Qinghai Province, a nurse is preparing to inject new coronavirus vaccines for vaccinators. China News Agency reporter Ma Mingyan photo On January 7, at the First People’s Hospital of Xining City, Qinghai Province, a nurse is preparing to inject COVID-19 virus vaccines for vaccinators. Photo by China News Agency reporter Ma Mingyan
The specific questions and answers are as follows:
1. At this stage, who are the key populations for new coronavirus vaccination?
The situation of the COVID-19 epidemic in some countries is different from that of foreign countries, and the vaccine use strategy is also different. The main strategy of new coronavirus vaccination needs to be considered in conjunction with the domestic epidemic situation and the goals of prevention and control. At present, the significance of vaccinating key populations is to protect this part of the population on the one hand, and on the other hand, it is conducive to “external import and internal prevention of rebound” and is conducive to the overall epidemic prevention and control in some countries.
At this stage, the key populations for COVID-19 virus vaccination mainly include those who are engaged in import cold chain, port quarantine, ship piloting, aviation, fresh food market, public transportation, medical disease control and other industries with relatively high risk of infection; going to medium-to-high-risk countries or Regional work, study and other personnel.
2. Is it necessary to inoculate the new coronavirus vaccine?
necessary. On the one hand, almost everyone in our country has no immunity against the new coronavirus and is susceptible to the new coronavirus. After the infection becomes onset, some people will develop critical illness and even cause death. After vaccination, on the one hand, the vast majority of people can gain immunity; on the other hand, through orderly inoculation of the new coronavirus vaccine, an immune barrier can be gradually established among the population to block the epidemic of new coronavirus pneumonia.
3. How to get the new coronavirus vaccine and where to get it?
The vaccinations of the new coronavirus vaccine are all carried out in the vaccination unit approved by the local health administrative department. Normally, the vaccination unit is located in the health service center, township health center or general hospital in the jurisdiction. If the vaccination involves some departments or enterprises where the key objects are relatively concentrated, some temporary vaccination units will also be set up locally according to the situation.
The administrative department of health or disease prevention and control of the jurisdiction will also announce the vaccination units that can be vaccinated against the new coronavirus as required, including the location and service hours. Please pay attention to the relevant information release platform.
For most key populations, the unit where the key populations are located will organize surveys, make appointments, and assist in carrying out the vaccination work. For individuals who go to work or study in high-risk countries or regions, they can pay attention to the relevant service information of local COVID-19 virus vaccination.
4. What are the contraindications for vaccination?
The contraindications of vaccination refer to situations where vaccination should not be taken. Because most contraindications are temporary, the vaccine can be vaccinated at a later time when the condition that caused the contraindication no longer exists.
Before the new coronavirus vaccination plan and vaccination guide are not specifically stipulated, the contraindications of the new coronavirus vaccination shall be implemented in accordance with the vaccine instructions. The usual contraindications for vaccination include: 1. Those who are allergic to vaccines or vaccine components; 2. Suffering from acute illness; 3. Those who are in the acute stage of chronic disease; 4. Those who are feverish; 5. Women during pregnancy.
5. How to discover and grasp the contraindications of vaccination?
During the implementation of the operation, if a severe allergic reaction occurs during the first dose of the vaccine, and it cannot be ruled out that it is caused by the vaccine, the second dose is not recommended. To understand the ingredients of the vaccine, people who have a previous allergy to the ingredients of the vaccine cannot be vaccinated.
During vaccination, the vaccination doctor should carefully ask the recipient about the health status and past allergies. Recipients should truthfully report their health status, disease history, and history of allergies to the vaccination doctor. Vaccine contraindications should be included in the informed consent form.
6. Do I no longer need to wear a mask after being vaccinated with the new coronavirus vaccine?
Before the population’s immune barrier is established, even if some people are vaccinated, everyone’s awareness of prevention and control and prevention and control measures cannot be relaxed. On the one hand, the vaccine immunization success rate is not 100%, and a small number of people who have been vaccinated may become ill during the epidemic. On the other hand, in the absence of an immune barrier, the new coronavirus is still easy to spread. Therefore, you should continue to wear masks after vaccination, especially in public places and crowded places; other protective measures such as hand hygiene, ventilation, and social distancing also need to be maintained.
7. How to form herd immunity in the crowd through vaccination?
Different infectious diseases have different infectivity, and people who block the epidemic of infectious diseases have different levels of immunity. Generally speaking, the stronger the infectious power of an infectious disease, the higher the immunity of the population is required. For example, measles and pertussis are highly contagious. If they are to be blocked, the population’s immunity must reach 90%-95%. To eliminate smallpox and poliomyelitis, the population’s immunity must reach more than 80%. When the population’s immunity reaches the above threshold, an immune barrier is established to block the spread of measles, whooping cough, smallpox and polio.
Population immunity is directly proportional to the protective efficacy of the vaccine and the vaccination rate. Therefore, to achieve sufficient population immunity, it is necessary to have a sufficiently high vaccination rate, which means that most people are vaccinated. Conversely, if there are more people who do not vaccinate or most people are unwilling to vaccinate, a strong immune barrier will not be formed, and the spread of diseases will easily occur when there is a source of infection.
8. Does the new coronavirus vaccine require a cold chain? How to ensure that the new coronavirus vaccine is safe and effective during transportation and storage?
Vaccine is a biological product. To ensure the quality of biological products, it must be stored and transported under the specified cold chain. The new coronavirus vaccine should do the same. The “Vaccine Management Law”, “Vaccine Storage and Transportation Management Regulations” and “Vaccination Work Regulations” all have specific regulations on the cold chain requirements for vaccine storage and transportation.
During vaccine transportation, vaccine transportation companies shall monitor and record the temperature regularly during the transportation process to ensure that the vaccine is in the specified temperature environment. When the vaccine is received, the receiving unit shall request and check the temperature monitoring record during the transportation.
During the vaccine storage process, the disease control agency and vaccination unit use thermometers or automatic temperature recorders to monitor the temperature of the refrigerator storing the vaccine, and measure the temperature once a day in the morning and afternoon (at an interval of not less than 6 hours), and fill in the cold chain equipment Temperature record table.
During the use of vaccines, the vaccination unit uses refrigerators and refrigerators (bags) to store vaccines. When storing and taking vaccines, they should close the refrigerators and refrigerators (bags) doors/covers in time, and minimize the number of times of opening the refrigeration equipment.
All relevant units must strictly abide by the above-mentioned specifications and requirements in each link in order to keep the vaccine in the full cold chain state and the quality of the vaccine can be guaranteed.
9. What is the suspected abnormal response to vaccination? What situations are included?
Suspected Vaccination Abnormal Reaction (AEFI) refers to a reaction or event suspected to be related to vaccination after vaccination, also known as a suspected adverse vaccine reaction. Including the following situations: adverse reactions of vaccines, reactions related to vaccine quality problems, reactions related to vaccination errors, psychogenic reactions, and coupling disorders (coupling reactions).
10. What is an adverse vaccine reaction?
Vaccine adverse reactions refer to reactions that are not related to the purpose of vaccination or accidents caused by the characteristics of the vaccine itself, and are related to individual differences in the recipient. Vaccine adverse reactions include general reactions and abnormal reactions. General reactions mainly refer to transient and mild body reactions in the recipient, such as local reactions such as redness, swelling, induration, and pain at the inoculation site, and systemic reactions such as fever, fatigue, and headache. Abnormal reactions mainly refer to related reactions that cause damage to the recipient’s organs or functions, which occur rarely, such as acute severe allergic reactions.
11. What is psychogenic response?
Psychogenic reaction refers to the reaction that occurs due to the psychological factors of the recipient after vaccination, which is mainly caused by psychological stress and anxiety during vaccination, without organic damage, and has nothing to do with the vaccine. Some are “fainting”-like manifestations, and some are “hysteria”-like manifestations. Group psychogenic reactions can occur during group vaccination activities.
12. What is the accidental reaction?
Coupling disease (coupling reaction) means that during the vaccination process, the recipient happens to be in the incubation period of a disease or at the early stage of the onset of the disease, and it happens by chance after vaccination. Therefore, the coupling disease (coupling reaction) is not caused by vaccination, has nothing to do with the vaccine, nor is it an adverse reaction after vaccination. Coincident diseases after vaccination sometimes cannot be judged immediately and need to be reported in a timely manner. It also requires investigation by disease control and other institutions and a diagnosis by an investigation and diagnosis expert group.
13. What circumstances are not an abnormal response to vaccination?
An abnormal response to preventive vaccination refers to an adverse drug reaction in which a qualified vaccine causes damage to the tissues, organs, and functions of the recipient during or after the implementation of the standardized vaccination, and the relevant parties are not at fault. The following situations do not belong to abnormal vaccination reactions: general reactions, vaccine quality accidents, vaccination accidents, coupling disorders (coupling reactions), and psychogenic reactions.
14. How is the surveillance of suspected abnormal response to vaccination carried out in China?
The “Vaccine Management Law”, “National Suspected Vaccination Abnormal Reaction Monitoring Program”, “Vaccination Abnormal Reaction Identification Method” and other laws and regulations have clear regulations on the monitoring and reporting of suspected vaccination abnormal reactions (suspected vaccine adverse reactions).
The specific measures include clarifying the responsible reporting unit, responsible reporter, the content of the report, and the time limit of the report, stipulating the suspected vaccine adverse reaction standards that need to be investigated, and the diagnosis of abnormal reactions needs to be completed by the investigation and diagnosis expert team, and the identification needs to be done by the province or city. Level medical society to complete. The monitoring of suspected vaccine adverse reactions is achieved through the monitoring information system established by the China Centers for Disease Control and Prevention, and the disease control institutions and the adverse drug reaction monitoring institutions realize information sharing. Disease control agencies and adverse drug reaction monitoring agencies at all levels will also regularly analyze and evaluate the monitored information. If a major event is encountered, timely analysis and evaluation will be conducted.
The systematic and standardized monitoring of adverse vaccine reactions in some countries started in 2005. With the development and deepening of work, the level of vaccine adverse reaction monitoring has been greatly improved. In 2011 and 2014, respectively, through the World Health Organization (WHO) assessment of the capacity of the adverse reaction monitoring part of some countries’s national vaccine regulatory system, the monitoring indicators for vaccine adverse reactions met or exceeded WHO evaluation standards.
15. How long is the protection of the new coronavirus vaccine currently in use in some countries?
The new coronavirus vaccine is a newly developed vaccine that has been put into use. It requires continuous monitoring and related research after mass vaccination to accumulate more scientific evidence and evaluate the protection durability of the new coronavirus vaccine.
16. Can new coronavirus vaccines from different manufacturers be used as an alternative to vaccination?
There is currently no evidence that different manufacturers or different types of new coronavirus vaccines can replace vaccination. It is recommended to use the same type of vaccine from the same manufacturer to complete vaccination.
17. What should I pay attention to during the new coronavirus vaccination process?
During the vaccination process, the recipient should pay attention to and cooperate with the following matters:
Before vaccination, you should understand the COVID-19 disease, COVID-19 virus vaccine related knowledge and vaccination process in advance.
When vaccinating, you need to bring relevant documents (ID card, passport, etc.), and in accordance with local prevention and control requirements, take personal protection, cooperate with on-site vaccination staff to inquire, and truthfully provide information such as your health status and vaccination contraindications.
After vaccination, stay for 30 minutes; keep the skin of the vaccination area clean and avoid scratching the vaccination site with your hands; if there is a suspected adverse reaction, report to the vaccination unit and seek medical treatment in time.
18. Why should I stay for half an hour after vaccination?
After vaccination, a very small number of people may experience acute allergic reactions and syncope. Severe life-threatening acute allergic reactions usually occur within 30 minutes after vaccination. If an acute allergic reaction occurs, timely treatment measures can be taken at the scene. Syncope also mostly occurs within half an hour after vaccination. If you leave the observation site immediately after vaccination, it may cause accidental harm to the recipient due to syncope. Therefore, the recipient needs to stay in the designated area of the vaccination unit for half an hour after vaccination.
19. How does the new coronavirus vaccine work in the human body?
After being vaccinated, the human body will produce protective antibodies, and some vaccines will also allow the body to produce cellular immunity and form a corresponding immune memory. In this way, the human body has immunity against diseases. Once the new coronavirus invades the human body, the antibodies produced by the vaccine and the cytokines released by cellular immunity can recognize, neutralize or kill the virus, and immune memory also quickly mobilizes the immune system to function, preventing the virus from continuing to multiply in the body, thereby To achieve the purpose of disease prevention.
20. After vaccination, how long does it take to produce antibodies against the new coronavirus?
According to the previous clinical trial study of the inactivated new coronavirus vaccine, about two weeks after the second dose of the inactivated vaccine, the vaccinated population can have a better immune effect.
21. After the new coronavirus has mutated, does vaccination against the new coronavirus still work?
Virus is one of the simplest organisms, and its proliferation depends on living cells. In the process of multiplication, the virus will mutate. Judging from the global monitoring of the mutation of the new coronavirus, there is currently no evidence that the virus mutation will invalidate the existing new coronavirus vaccine. However, the World Health Organization, national research institutions, vaccine manufacturers, etc. are closely monitoring the mutation of the new coronavirus and are also carrying out related research, which will provide early warning and scientific analysis basis for subsequent vaccine development and application.
22. If ordinary people are willing to be vaccinated against the new coronavirus, can they sign up for vaccination?
some countries’s current vaccination strategy is to follow the “two-step” plan, and the first step is to vaccinate key populations. In the second step, as vaccines are approved for marketing and the production of vaccines gradually increases, more vaccines will be put into use. By carrying out vaccination in an orderly manner, eligible members of the public will be able to “do everything they need”, gradually build an immune barrier among the population, and control the spread of new coronavirus pneumonia in the country.
23. What kind of protective measures are needed for people who are not included in the key population and who have not been vaccinated against the new coronavirus?
some countries’s current vaccination strategy is to follow the “two-step” plan. The first step is the vaccination of key groups, and the second step is the vaccination of other groups. In the process of prevention and control of the COVID-19 epidemic, the non-vaccine prevention and control measures that some countries has implemented are very effective. For the public, although there may be no vaccinations for the time being, there are still many effective prevention and control measures, such as wearing masks, maintaining social distancing, washing hands frequently, and ventilating.
24. What are the common adverse reactions of new coronavirus vaccination?
From the results of the preliminary clinical trials of the new coronavirus vaccine and the information collected during emergency use, the occurrence of common adverse reactions of the new coronavirus vaccine is basically similar to that of other vaccines that have been widely used. Common adverse reactions mainly include redness, swelling, induration, and pain at the site of inoculation, as well as clinical manifestations such as fever, fatigue, nausea, headache, and muscle aches.
25. What factors may affect the effectiveness of the new coronavirus vaccine?
Under normal circumstances, factors such as pathogens, vaccine characteristics, and recipient status affect the effectiveness of vaccine vaccination. In the process of vaccine development and use, in order to ensure the effect of vaccination, the above-mentioned influencing factors are all within the scope of consideration. The new coronavirus vaccine is a brand new vaccine, and the influence of related factors on its effect needs further observation and research.
26. Do I still need to be vaccinated against the COVID-19 virus if I have been infected with the COVID-19 virus?
For most infectious diseases, the human body will develop a certain degree of immunity after being infected with pathogens. This part of the population is usually not the target of vaccination, such as smallpox, measles, rubella, chickenpox and other diseases are no longer the target of vaccination. At present, although there have been reports of secondary infections in people who have been infected with the new coronavirus, the problem is still a case and has not generally appeared, and more follow-up studies are still needed to reach a conclusion. For confirmed cases of new coronavirus pneumonia and asymptomatic infections known before vaccination, it is not recommended to receive the new coronavirus vaccine; for those who have not been clearly infected with the COVID-19 virus or have suffered from the COVID-19 pneumonia, those who meet the vaccination requirements can be vaccinated.
27. Will our prevention and control measures be adjusted after the vaccination starts?
For individuals, the protective effect of vaccination is not 100%, and it takes a certain amount of time to produce protective antibodies; for groups, the new coronavirus is still easy to spread without the formation of an immune barrier. Therefore, to prevent the COVID-19 pneumonia epidemic from rebounding, other prevention and control measures must be adhered to at this stage, including protective measures such as wearing masks, maintaining social distancing, washing hands frequently, and ventilation.
28. How does the new coronavirus vaccine trace the entire process?
The Vaccine Management Law requires the country to implement an electronic traceability system for the entire vaccine process. After the vaccine is on the market, there should be accurate and standardized records in all aspects of production, transportation, storage and use, etc. The information recorded throughout the traceability includes vaccine varieties, vaccine manufacturers, dosage forms, specifications, batch numbers, expiration dates, and vaccination case information Etc., areas that realize informatization management will be entered into the electronic information system in a timely manner, and the above information will be traced back to the entire process of vaccine circulation and use through electronic information systems and other methods.
29. Do I need to test for antibodies before deciding whether to vaccinate against the new coronavirus?
The production of specific antibodies in the human body is generally obtained through natural infection or vaccination. At present, it is not completely clear what level of antibody needs to be able to prevent new coronavirus pneumonia. It is recommended that anyone who meets the vaccination requirements can be vaccinated as long as they have not been clearly infected with the COVID-19 virus or have suffered from the COVID-19 pneumonia. There is no need to test for the presence of antibodies before vaccination.
30. Will the new coronavirus vaccine be vaccinated every year like the flu vaccine?
Under normal circumstances, factors such as pathogens, vaccine characteristics, and recipient status affect the effectiveness of vaccine vaccination. Influenza viruses mutate faster and the protection period of influenza vaccines is short, so it needs to be vaccinated every year. Although the new coronavirus has also undergone a certain degree of mutation, according to the information currently released on the World Health Organization website, it is shown that there is no evidence that the existing new coronavirus vaccine has failed in response to the mutation of the new coronavirus that has emerged in countries such as the United Kingdom and South Africa. . Whether the new coronavirus vaccine will be vaccinated every year like the flu vaccine, it is necessary to continue to conduct research on the impact of virus mutation on the vaccination effect and the durability of vaccine protection.
(source:internet, reference only)
Disclaimer of medicaltrend.org



