How does the immune system receive “Training”?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
How does the immune system receive “Training”?
How does the immune system receive “Training”? Nature reveals the “behind-the-scenes work” of gut bacteria.
We and our gut microbiome (flora) have jointly evolved a mutually beneficial relationship, which is a very complex relationship.
Although the human host provides a friendly environment for bacteria, and the bacteria provide the host with many beneficial functions including nutrient supply and prevention of pathogen infection, however, maintaining this relationship requires a careful immune balance: it is necessary to control the symbiotic microorganisms locally, but also At the same time limit the inflammatory response.
Because once certain bacteria enter the tissues or organs where they shouldn’t appear, even good bacteria can have bad effects.
In a new study published in the top international academic journal “Nature” on May 13, a research team led by the Memorial Sloan Kettering Cancer Center in New York, USA revealed how the body maintains this balanced relationship.
The study shows that early life is a critical period when the immune system learns to recognize intestinal bacteria and establish a monitoring mechanism.
When these mechanisms are defective, the immune system will attack beneficial bacteria in the wrong place, causing chronic inflammation, which is also the cause of inflammatory bowel disease.

Previously, there have been reports on the antigen-specific recognition of intestinal bacteria by immune T cells. Although the local environment determines the differentiation of effector cells, it is not clear how the microbiota-specific T cells in the thymus are cultured.
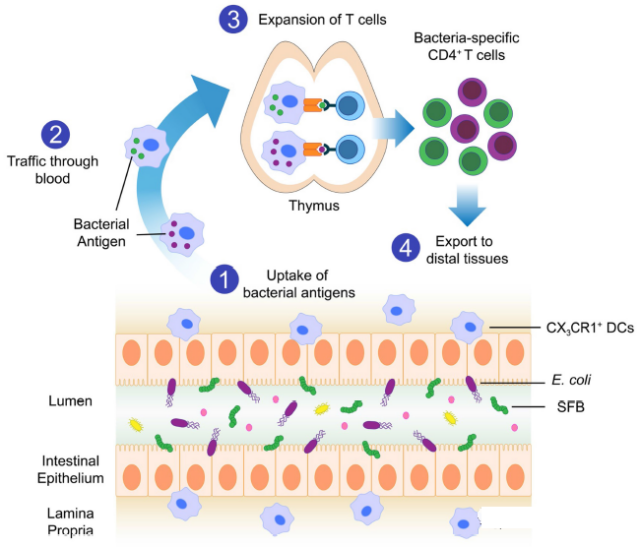
In this study, researchers tried to understand how the body maintains a healthy relationship with bacteria and discovered how the intestinal flora shapes the developing immune system. The dendritic cells that colonize the intestine early in life travel long distances to transport the captured microbial antigens from the intestine to the thymus.
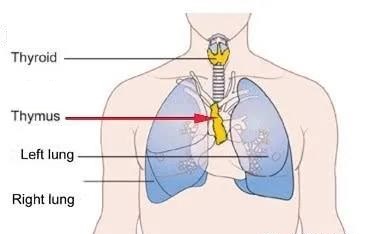
The thymus is located in the upper part of the anterior mediastinum behind the sternal stem. It is a gland responsible for “education” of immune T cells. The delivered microbial antigen induces the thymus to produce T cells targeting the microbiome.
Then, the T cells leave the thymus and enter the lymph nodes, intestines and other parts to maintain control of the bacteria. In this way, the developing microbiome forms and expands the thymus and peripheral T cell pools, thereby enhancing the recognition of intestinal microbes and pathogens.
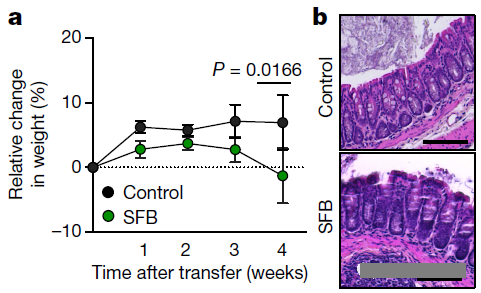
In the first three years of human life, the composition of the intestinal flora gradually stabilized, almost similar to that of adults. This is equivalent to an expansion phase of the T cell bank.
The researchers determined this process by implanting certain bacteria in the intestines of mice. They found that the thymus produced T cells that specifically recognize these bacteria. However, how this happened is still unknown.
Finding DNA from bacteria in the thymus and lymph nodes is the first clue to the migration of bacteria to these sites. To track their trajectory, the researchers used specially designed mice whose cells emit red fluorescence when exposed to laser light. In the two days after light activation, the red intestinal cells finally reach the thymus, lymph nodes and spleen. And the researchers found that these processes are strong in the first few weeks after the mouse is born, but significantly weakened after it reaches adulthood.
Matthew Bettini, co-corresponding author of the study and associate professor in the Department of Pathology at the University of Utah School of Medicine, said: “Our research challenges the previous assumption that potential pathogens have no effect on the immune cells that develop in the thymus. Instead, we see the Learning in bacteria. Although these events that determine T cell types occur early in life, they have a greater impact later in life.”
This view is obvious because researchers have discovered that T cells that target beneficial bacteria can also defend against harmful bacteria that are closely related to it. Mice that carried E. coli at a young age were more than six times more likely to survive a lethal dose of Salmonella later in life than other mice. Research results show that building immunity to the microbiome can also enhance the body’s protection against harmful bacteria that have not yet been encountered.
From the moment we are born, our immune system has been established. In this process, it will learn as much as possible to identify beneficial and harmful bacteria. This study shows that the intestinal flora has a window period to enter the immune “education” process. This opens up the possibility of designing treatments that can affect the trajectory of the immune system at an early stage.
Researchers say that by studying these early communications between the body and the microbiome, you will discover how important it is to activate the immune system from the beginning. This early education of immune T cells is absolutely necessary for the rapid cultivation of an extensive immune cell pool to protect the body.
Bettini said: “Our research results may be extended to certain areas, in these areas, certain bacteria are found to be protective or pathogenic to other diseases, such as type 1 and type 2 diabetes. Next, we will have to understand Is the window of bacterial exposure and T cell development also important in triggering these diseases?”
(source:internet, reference only)
Disclaimer of medicaltrend.org



