AAA GENE therapy treats rare genetic diseases (Only 135 found in the world)
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
AAA GENE therapy treats rare genetic diseases (Only 135 found in the world)
AAA GENE therapy treats rare genetic diseases (Only 135 found in the world). Only 135 patients with rare genetic diseases were found in the world, which was overcome by AAV gene therapy.
Aromatic L-amino acid decarboxylase deficiency, abbreviated as AADC deficiency, is due to a deletion mutation in the ADDC gene, leading to a lack of AADC enzyme responsible for the metabolism of levodopa and serotonin, resulting in a lack of dopamine and serotonin in the body. Symptoms begin to appear in the next 6 months, leading to severe developmental delays. They cannot control their muscles, usually cannot speak, eat, or even lift their heads. They often suffer from an eye movement crisis that lasts for several hours. Seizures are often accompanied by sleep disturbances and emotional out-of-control.
AADC deficiency is an extremely rare autosomal recessive genetic disease, and only 135 cases have been found worldwide. For this extremely rare genetic disease, there is still a lack of effective treatment methods.
With the gradual maturity and application of gene therapy, new hope has been brought to these patients with AADC deficiency who are suffering from great pain.
On July 12, 2021, researchers from the University of California, San Francisco and Ohio State University published the title: Gene therapy for aromatic L-amino acid decarboxylase deficiency by MR-guided direct delivery of AAV2- in the journal Nature Communications. AADC to midbrain dopaminergic neurons gene therapy clinical trial paper.
The research team first loaded the human AADC gene into an adeno-associated virus (AAV) vector, and then, with the help of MRI technology, accurately injected the AAV virus into the substantia nigra compact area (SNc) and ventral area of the midbrain of the patient. Covered area (VTA).
A total of 7 children (4-9 years old, 4 women and 3 men) received this gene therapy this time. A follow-up 2 years after the treatment found that the symptoms, motor function and quality of life of these patients have been significantly improved. This therapy is expected to change the treatment of AADC deficiency and other similar brain diseases in the future.

The previous AAV gene therapy for more than 20 children with AADC deficiency in Japan and Taiwan, China was to inject the AAV virus into the putamen area of the patient’s brain, but the results of the treatment were not satisfactory.
In this clinical trial, the research team decided to inject the AAV virus vector into the substantia nigra compact area (SNc) and the ventral tegmental area (VTA) of the midbrain to increase the AADC enzyme activity in the midbrain dopaminergic neurons, thereby saving Dopamine biosynthesis and dopaminergic neurotransmission in the substantia nigra striatum, midbrain limbus, and midcortical pathways.
In order to maximize the safety and targeting accuracy of injections, the research team developed and used an MRI-guided gene delivery platform to accurately deliver the AAV virus to a specific area and observe the delivery effect in real time.
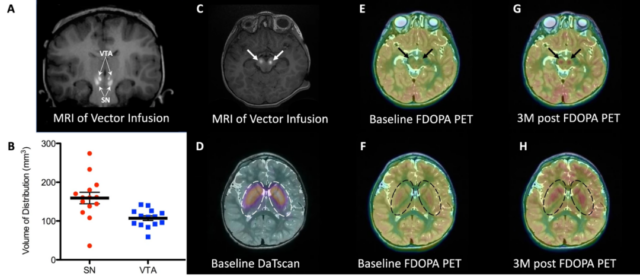
In gene therapy delivery, all 7 subjects showed measurable clinical improvement in symptoms and motor function.
In the third month after treatment, 6 out of 7 subjects had the eye movement crisis completely disappeared, and in the 12th month after treatment, 6 out of 7 subjects were able to control their head normally, and 4 out of 7 subjects were able to control their heads normally. sit down. At the 18th month after treatment, 2 people can walk with the support of both hands. Their sleep disorders and emotional out-of-control have also been significantly improved.
This therapy is expected to change the treatment of AADC deficiency and other similar brain diseases in the future. The research team believes that the gene therapy method can also be used to treat other genetic diseases and common neurodegenerative diseases, such as Parkinson’s disease and Alzheimer’s disease.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



