Cancer cell: Research and treatment progress of metastatic breast cancer
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Cancer cell: Research and treatment progress of metastatic breast cancer
Cancer cell: Research and treatment progress of metastatic breast cancer. The concept of precision oncology is based on the assumption that understanding the genomic basis of a patient’s cancer will guide the choice of potentially effective targeted therapies.
In recent years, as the pace of society and the pressure of life have steadily increased, breast cancer, as a major member of cancer, has become the world’s most common cancer.
In 2021, A large-scale study by the top international oncology journal “JAMA Oncology” showed that the mortality rate of men with breast cancer is higher, 19% higher than that of women.
Breast cancer has invisibly become one of the killers of contemporary youth and middle-aged and elderly people.
Recently, “Cancer cell”, a subsidiary of the internationally renowned magazine “CELL”, selected 8 latest advances in cancer research in the latest issue of the magazine on July 12, including an article on breast cancer. research paper.
In this article, researchers have obtained genome-wide sequencing data from 170 breast cancer patients. A series of studies have shown that the “cancer seeds” that cause cancer metastasis or recurrence have a similar mutation process to the original cancer cells. However, cancer cells that metastasize far away have mutations not seen in primary cancer cells, and these mutated cancer cells can continue to drive the development of cancer.
Metastatic breast cancer has almost left the medical community at a loss in the past 5-10 years. This frustrating phenomenon has not undergone much research changes and progress in the past 20-30 years. The research results of this article are for metastatic Breast cancer has brought some new treatment ideas.
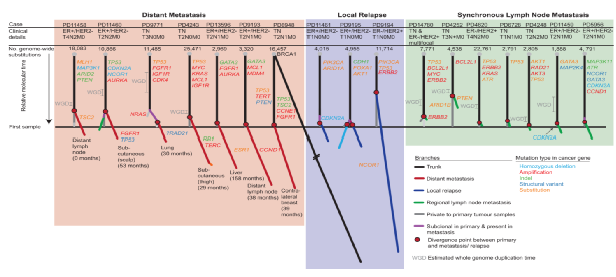
Figure 1. Evolutionary tree diagram of primary breast cancer metastasis or local recurrence
This article discusses the evolution of 17 cases of primary breast cancer metastasis or local recurrence, and each dendrogram region represents a matching normal sample of a patient and 2-4 tumor samples per case (a total of 40 tumor samples) , According to the results to group the dendrogram: remote metastasis group (red area), local recurrence group (blue area) or synchronized axillary lymph node metastasis group (green area).
Among them, the branches of metastasis or recurrence follow the same color theme, while the branches that represent specific clones of the primary tumor are grayed out. The black lines represent clonal virus mutations that are present in 100% of the cells in each sample. The purple branches represent subcloned metastases or mutations within the primary tumor. The length of the branches reflects the proportion of cluster somatic mutations caused by the subcloning. The red circle identifies the point of divergence between metastatic/recurring seeds.
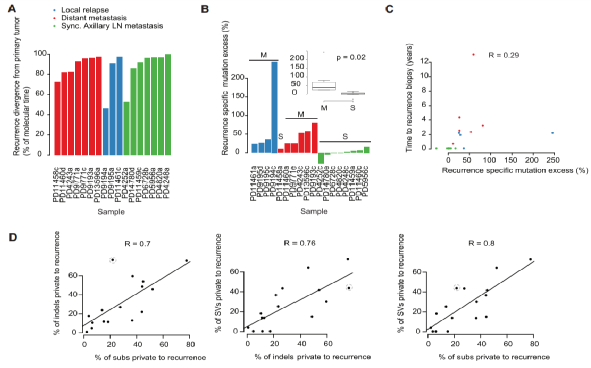
Figure 2. Genome-wide somatic mutation time of the 16 cases of breast cancer experimental group
On this basis, the researchers carried out further studies on 16 breast cancer cases, using bar graph 2 (A) to show the different molecular time points between recurrent seeding clones and primary tumors (the molecular time is replaced by bases). Determined by the number of breast cancers), and using range differences and scatter plots in Figures B, C, and D to show the recurrence specificity and radiation mutation characteristics in 16 samples, and draw 10 breast cancer samples with local recurrence or distant metastasis Each phylogenetic tree branch of the whole genome case illustrates the characteristic composition of the studied mutation (HRD, homologous recombination defect; MMR, mismatch repair defect).
Breast cancer can acquire many different mutation processes and a wide range of cancer genes during its development. The result is often quite large differences in genomic characteristics, even more pronounced than the already daunting levels in primary breast cancer. In order to verify the primary and recurrent mutation processes of breast cancer and the possible impact of radiotherapy on the genome, the team conducted a study on the structural mutation driving factor mutations in three cases of breast cancer recurrence PD9193 and found that the structural mutation factors were driven The CCND1 re-amplification, the loss of TP53 copies are shown in Figure 3 (A) and FGFR1 amplification (B). Interestingly, these data show that the complex catastrophic events in the development of metastasis are consistent with the results of single-cell sequencing studies tested by the team that the discontinuous copy number evolution of primary breast cancer lesions is actually consistent.
This means that even if the primary tumor is treated optimally, about 10% of patients will still be at risk of local recurrence, and the remaining 30% and 60% of cases may also be in the future Accompanied by varying degrees of distant metastatic disease.
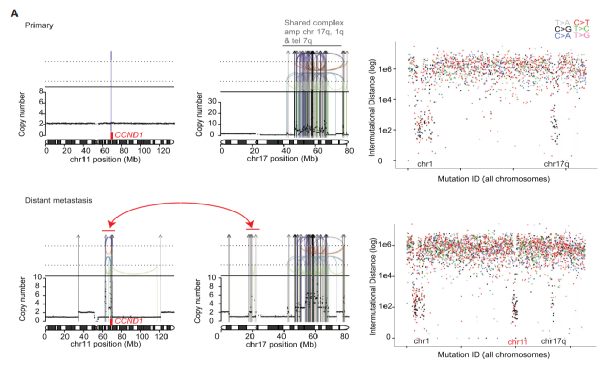
Figure 3. Three cases of breast cancer recurrence (A) cases of structural variant driver mutation results in PD9193
The article mentioned at the end that the concept of precision oncology is based on the assumption that understanding the genomic basis of the patient’s cancer will guide the choice of possible effective targeted therapies.
Based on this hypothesis, this means that samples of tumor cells that represent our treatment targets can be obtained. At present, the world has good detection and characterization results for the genome of primary breast cancer, but the analysis data for recurrent or metastatic breast cancer is much less. Due to the small sample size, it is difficult to extract the general evolutionary pattern between primary and recurrence in contemporary medical applications. These unresolved problems have important biological and clinical significance for human treatment of breast cancer.
The author of the article also stated that the research in this article is mainly to solve some of the existing problems mentioned above, including metastasis and its primary lesions closely related to breast cancer; recurrence in the same area and caused by lymphatic transmission Whether there is a difference in the evolution of axillary metastasis and distant metastasis caused by blood cell dissemination; whether the driving landscape of metastasis is different from that of the primary cancer; and whether there are cancer genes specific to metastasis. Because the survival rate of patients with metastatic breast cancer is very low, it is particularly important to determine whether newly emerging metastatic driver mutations may provide opportunities for personalized treatment.
When looking for a genome that is very suitable for autonomous living in remote areas of the body, breast cancer can enter many different mutation processes and an extensive cancer gene pool. There are often considerable differences between patients in the genome profiles, even more obvious than the differences in the genome profiles of primary breast cancer.
To count and confirm this extremely complex genome profile usually requires recruiting a large number of patients with metastatic disease, and combining analysis with transcription, epigenome and clinical data. The research in this article shows that this research method has potential clinical impact, and can provide the clonal evolutionary model of breast cancer mutation process, the mechanism of treatment failure and the direction of new treatment targets.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



