First time: CAR-T cell therapy successfully treats lupus erythematosus
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
First time: CAR-T cell therapy successfully treats lupus erythematosus
First time: CAR-T cell therapy successfully treats lupus erythematosus. NEJM: CAR-T cell therapy successfully treats lupus erythematosus for the first time.
CAR-T cell therapy, the full name is Chimeric Antigen Receptor T-Cell Immunotherapy, refers to chimeric antigen receptor T-cell immunotherapy. To put it simply, the patient’s T cells are genetically engineered in vitro to make them recognize tumor cell surface antigens, and then these modified T cells are infused back into the patient to achieve the therapeutic effect of identifying and killing cancer cells.
Since the FDA approved Novartis’s CAR-T therapy Kymriah (CTL-019) on August 30, 2017, the world has approved 5 CAR-T therapies for marketing, of which 4 target CD19 and 1 target BCMA. It is worth mentioning that on June 22, 2021, the National Food and Drug Administration (NMPA) of China approved Fosun Kate’s CAR-T cell therapy product Yiqi Lirensai injection (also known as Akirensai, code name: FKC876), marking China’s first CAR-T cell therapy product approved for marketing.
On August 5, 2021, researchers from the University of Erlangen-Nuremberg in Germany published an article titled CD19-Targeted CAR T Cells in Refractory Systemic Lupus Erythematosus in the New England Journal of Medicine (NEJM).
The research team performed CAR-T cell therapy on a 20-year-old female patient with severe systemic lupus erythematosus. The therapy quickly relieved his condition without obvious side effects. This patient also became the first lupus patient in the world to receive CAR-T cell therapy.
The research team said that although it was only one patient, the test results showed that the CAR-T therapy responded quickly and the patient’s symptoms subsided quickly. The patient can be said to have “completely recovered” and has not needed treatment for more than four months. It is reported that the research team is now using CAR-T cells to treat two other patients with severe lupus erythematosus.
The research team published the results of this treatment in the New England Journal of Medicine (NEJM),

For lupus patients, their enemy is not the tumor, but their own immune system. Their own immune system mistakenly produces antibodies and attacks their own body tissues. The most common form of lupus is systemic lupus erythematosus, which can severely damage a series of organs.
An untreated 20-year-old female patient suffers from severe systemic lupus erythematosus. Attacks from the autoimmune system have caused her to suffer from arthritis, kidney damage, and inflammation of the lungs and heart. The conventional treatment drugs are ineffective.
In desperation, the treatment team decided to try CAR-T cell therapy. Like the previous CAR-T cell therapy used for cancer treatment, the doctor collected the patient’s T cell samples and then genetically modified these T cells in the laboratory. , Allowing patients’ T cells to recognize the CD19 protein on B cells. B cells normally produce antibodies to help the body fight pathogen infections, but in lupus erythematosus, B cells produce antibodies that attack themselves.
The transformed T cells were infused back into the patient. After 44 days, the antibodies that attacked her body disappeared, her condition was significantly alleviated, and there were no obvious side effects.
Using the current existing treatment methods, most systemic lupus erythematosus is difficult to maintain remission, let alone cure, but this study shows that CAR-T therapy is expected to completely cure lupus erythematosus, the “undead cancer.”
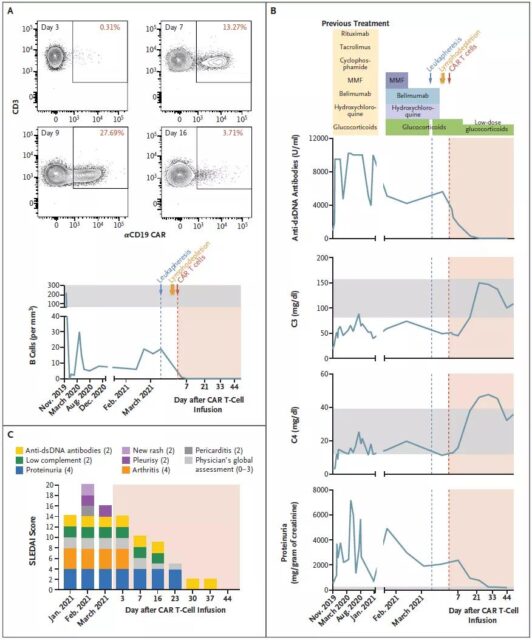
The research team also pointed out that the infusion cost of CAR-T cell therapy exceeds $200,000, which does not include hospitalization costs. Moreover, lupus erythematosus is a highly heterogeneous disease, which means that patients often need targeted treatment, which further increases the cost of treatment. Therefore, if you want to provide patients with lupus erythematosus with a safer, effective, and low-cost CAR -T therapy also needs to find the immune abnormalities that are common in patients with lupus erythematosus.
This study shows that CAR-T cell therapy targeting CD19 can induce rapid remission of refractory systemic lupus erythematosus. In view of the role of B cells in a variety of severe autoimmune diseases, this also suggests to us that CAR-T cell therapy targeting B cell antigens may have wider applications.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



