What antigen is for the CAR-T cell clinical trials of solid tumors?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
What antigen is for the CAR-T cell clinical trials of solid tumors?
What antigen is for the CAR-T cell clinical trials of solid tumors? CAR-T cell therapy targets a total of 20 organs for many different solid tumors (Figure 1). Especially tumors in the brain/central nervous system, liver, pancreas, and lungs are the targets of many clinical trials.
1. Target organ
At present, CAR-T cell therapy targets a total of 20 organs for many different solid tumors (Figure 1). Especially tumors in the brain/central nervous system, liver, pancreas, and lungs are the targets of many clinical trials (n=45, 43, 38, and 36, respectively). This may represent the high medical needs of tumors in these organs and/or the lack of effective alternative therapies (ie non-CAR-T therapies).
There are a total of 51 clinical trials targeting multiple organs (Figure 1), mainly because the antigens targeted by CAR-T cells are expressed on tumors in different organs (for example, epidermal growth factor receptor (EGFR), natural killer group) 2D (NKG2D)-ligand, human epidermal growth factor receptor 2 (HER2), mucin 1 (MUC1) and carcinoembryonic antigen (CEA)).
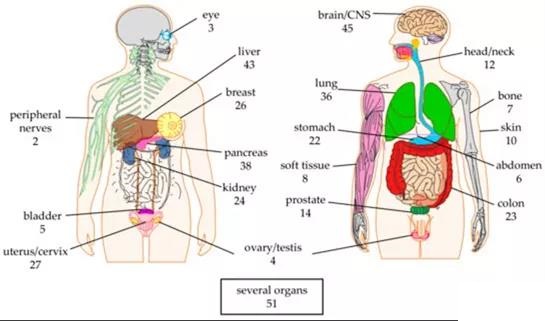
Figure 1. Organ schematic diagram of CAR-T cells targeting solid tumors. The number indicates the number of clinical trials for this organ. The data comes from clinicaltrials.gov.
2. Targeting solid tumors
As can be seen from Figure 2, CAR-T cells registered on clinicaltrials.gov targeted 57 different tumor entities. Nine different tumor entities are described in the brain, six in the liver, and five in the lung (Figure 2). Unfortunately, many registered clinical trials do not accurately describe the tumor entities targeted. These files only indicate organs (for example, “brain”; which type of tumor is not specified) (Figure 2). In addition, 34 registered trials only indicated “solid tumors” (Figure 2).
The four most targeted tumor entities are pancreatic cancer (n=34), gastric cancer (n=22), ovarian cancer (n=21), and colorectal cancer (n=20) (Figure 2). This does not reflect the incidence of cancer worldwide. In 2018, the top 3 newly diagnosed cancer types for men and women were: (1) lung cancer (12.3%), (2) breast cancer (12.3%), and (3) colorectal cancer (10.6%) [31,32,33 ]. This may be caused by local differences in the incidence of cancer (for example, in China, gastric cancer is the third most commonly diagnosed cancer after lung cancer and colorectal cancer, and even the second most common cause of cancer-related deaths. There is a higher interest in clinical trials for this cancer entity).
In fact, of the 22 clinical trials for gastric cancer, 15 were conducted in China.
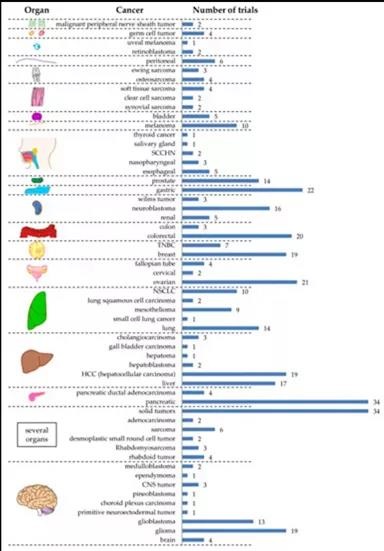
Figure 2. Schematic overview of tumor entities targeted by CAR-T cells against solid tumors grouped by organ.
3. Target antigen
The ideal target antigen on solid tumors unifies three basic properties: (i) evenly exists on the surface of malignant cells, reducing the risk of antigen-negative escape mutation; (ii) there is no expression on non-malignant host cells, which excludes targeting/ Non-tumor activity, which may bring serious and potentially fatal side effects; (iii) As a key role as a carcinogenic driver in cancer cells, due to the selective survival advantage conferred on malignant cells, this may compound antigen closure.
Co-expression on bystander cells that maintain the tumor microenvironment, such as tumor-associated vasculature, fibroblasts, and macrophages, represents another valuable feature.
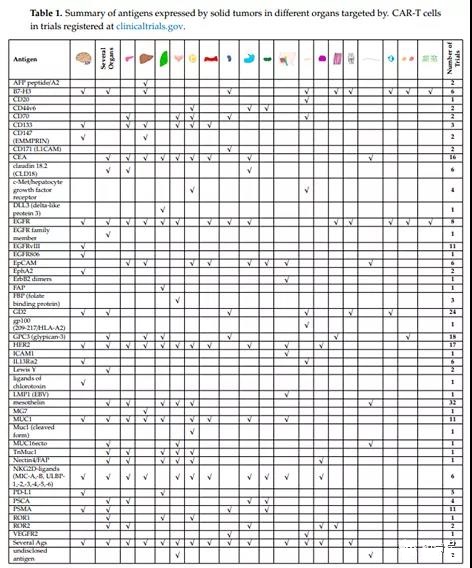
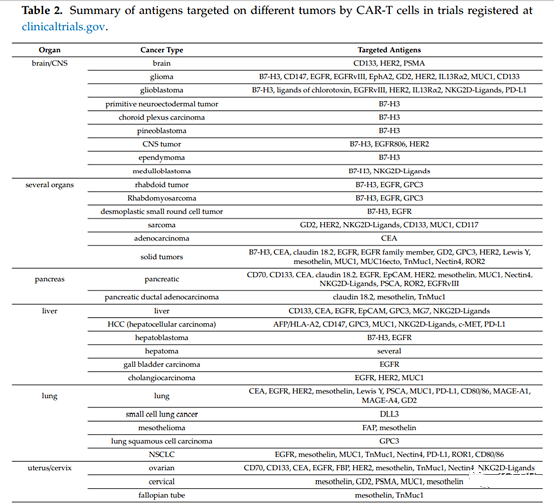
EGFR and HER2 are members of the ErbB family of receptor tyrosine kinases (ie EGFR (ErbB-1), HER2/(neu) (ErbB-2), Her 3 (ErbB-3) and Her 4 (ErbB-4). Mutations in EGFR lead to its overexpression, leading to its continuous activation and uncontrolled cell division in many different cancers (for example, non-small cell lung cancer (NSCLC), colorectal cancer, pancreatic cancer, etc.). The functions of HER2 are similar, mainly in It is overexpressed in breast cancer, but it is also overexpressed in other cancer types, such as ovarian cancer, glioma, etc. [62-64].
NKG2D ligands MIC-A, MIC-B and ULBPs 1, 2, 3, 4, 5, 6 are self-inducing proteins, and they are upregulated in stressed, infected and transformed cells. These ligands can be recognized by NKG2D receptors expressed on NK cells, NKT cells, γ/δ T cells, and activated CD8+ αβ T cells [65,66]. Colorectal cancer, ovarian cancer, and other cancers express higher levels of NKG2D ligand and can be targeted by CAR-T cells that bind NKG2D in a chimeric receptor.
B7-H3 (ie CD276 or B7 homolog 3) is a costimulatory molecule for T cells, such as expressed on activated dendritic cells and monocytes. When TCR signal is combined with B7-H3 costimulation, T cells stimulated by B7-H3 proliferate and differentiate into cytotoxic T cells and selectively secrete IFNγ. It has only limited expression in healthy tissues. However, B7-H3 is overexpressed on neuroblastoma, which inhibits NK cells from recognizing and killing tumor cells.
In addition, CD276 is overexpressed in several other tumors, such as pancreatic ductal adenocarcinoma (PDAC), prostate cancer, ovarian cancer, lung cancer, and renal clear cell carcinoma, as well as tumor-related vasculature and stromal fibroblasts. Mucin 1 (MUC1) is a highly glycosylated membrane protein that is expressed on the surface of epithelial cells in the intestine, stomach, lung, eye and other organs. It binds pathogens with oligosaccharides to inhibit pathogens from reaching the cell membrane. MUC1 in the junction It is overexpressed in rectal cancer, breast cancer, ovarian cancer, lung cancer and pancreatic cancer.
Carcinoembryonic antigen (CEA, also known as CEACAM5) is a glycoprotein that is widely expressed during fetal development and on some adult tissues (such as the epithelium of the colon, stomach, and esophagus). In normal epithelial cells of the lung and gastrointestinal tract, CEA has apical polarity, faces the lumen, and cannot be recognized by CAR-T cells. Its function and signal transduction in normal tissues are still not fully understood. CEA is overexpressed in colorectal cancer, pancreatic cancer, gastric cancer, lung cancer and breast cancer, and plays a role in tumor metastasis. In cancer, CEA has lost its apical polarity and even partly falls off, leading to increased serum levels. At this stage, CEA expressed on tumor cells can be targeted by CAR-T cells.
Sum up:
This summary review of all clinical trials that use CAR-T cells to treat solid tumors registered with clinicaltrials.gov shows that the use of many different CAR structures, application pathways, and the introduction of additional functions of T cells follow many strategies.
This may indicate that an ideal strategy for the treatment of solid tumors with CAR-T cells has not yet been found. This can also be seen in the clinical results of the trial that reported this; only 52 of the 375 patients responded.
Nevertheless, the use of CAR-T cells to treat solid tumors contains huge opportunities, and further development and clinical testing are needed to meet the high medical needs for the treatment of such cancers.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



