Monoclonal antibodies may end the raging COVID-19 variant strains
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Monoclonal antibodies may end the raging COVID-19 variant strains
Monoclonal antibodies may end the raging COVID-19 variant strains. Since it was first discovered in India in October 2020, the COVID-19 pneumonia delta (Delta) mutant strain (B.1.617.2) has quickly replaced other variants and has become the main epidemic strain in most countries and regions around the world. In May of this year, Delta “lived up to expectations” and was on the World Health Organization (WHO) “Variant of Concern” (Variant of Concern, VOC) list.
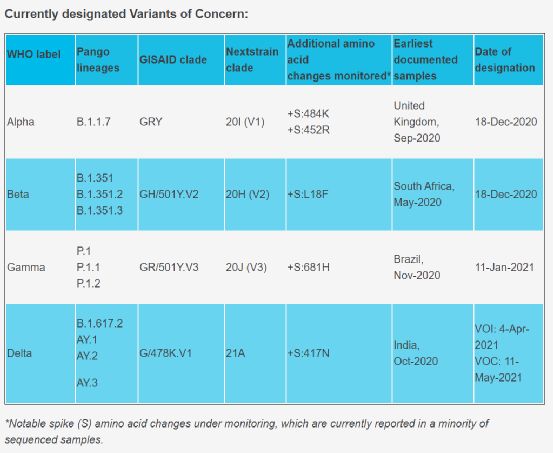 Figure 1. WHO’s method of labeling new coronavirus variants. Image source: WHO
Figure 1. WHO’s method of labeling new coronavirus variants. Image source: WHO
What is it that makes Delta variant so rampant?
Studies have shown that Delta has three mutations on the Spike protein (S protein) that play a key role in its propagation characteristics, namely L452R, E484Q and P681R.
Among them, the mutations of L452R and E484Q occurred in the receptor-binding domain (RBD) of the spike protein and angiotensin-converting enzyme 2 (ACE-2) of human cells. L452R can improve the ability of viruses to invade cells, and E484Q helps to enhance the immune escape of viruses. In addition, P681R has the function of enabling viruses to enter cells more effectively.
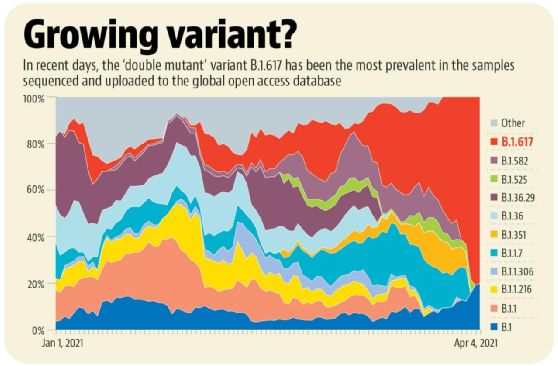 Figure 2. The growth trend of different new coronavirus variants. Image source: Hindustan Times
Figure 2. The growth trend of different new coronavirus variants. Image source: Hindustan Times
On July 23, Lu Jing and others from the Guangdong Provincial Center for Disease Control and Prevention published a paper titled “Viral infection and Transmission in a large well-traced outbreak caused by the Delta SARS-CoV-2 variant” on the preprint website MedRxiv. .
The team found that it took an average of 4 days from the first exposure of the Delta mutant strain to a positive PCR test, compared with 6 days for the original strain in 2020. In addition, people infected with the Delta mutant strain produced more viruses, and the viral load was more than 1,000 times that of the original strain.
The above data shows that the combination of higher viral load and shorter incubation period will cause Delta virus to become more easily spread. It is reported that the increasing number of patients has also increased the possibility of “breakthrough infections” in the vaccinated population. These are the important reasons why the Delta mutant strain was able to spread so “successfully” quickly.
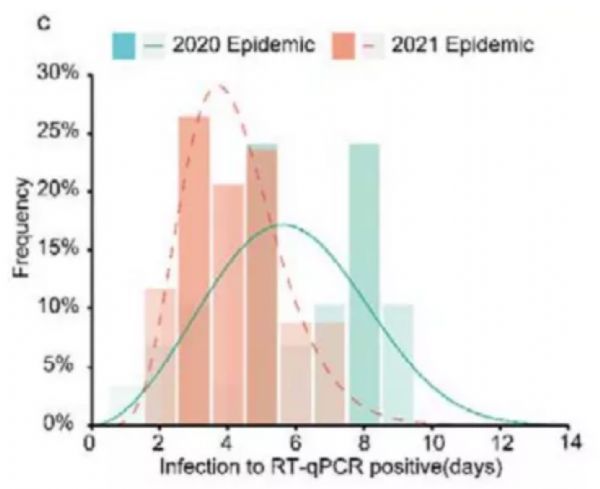
Figure 3. Comparison of the incubation period between the Delta mutant strain and the original strain. Image source: Medrxiv
We wait to die?
Of course the answer is no!
On August 4, the official Weibo of “China BioCNBG” released an exciting news: The research team of Sinopharm China Biotech Yang Xiaoming has newly discovered the effective monoclonal antibody “2B11” against the mutant strain of Delta, and its neutralizing activity can be as high as IC50. 5ng/ml.
Relevant research results were published online on July 27 in the sub-Journal of Nature, Cell Discovery. The title of the research paper is “Screening of potent neutralizing antibodies against SARS-CoV-2 using convalescent patients-derived” phage-display libraries”. The partners of this project are Shanghai Advanced Institute of Chinese Academy of Sciences and Wuhan Institute of Virology, Chinese Academy of Sciences.

Figure 4. “Screening of potent neutralizing antibodies against SARS-CoV-2 using convalescent patients-derived phage-display libraries” Image source: Cell Discovery
The above results mean that monoclonal antibodies are effective against mutated strains of new coronavirus such as Delta, and the treatment of such mutated strains is expected to usher in “specific drugs.”
How effective is “special medicine”?
Monoclonal antibody (mAb) is a type of antibody produced by only one type of immune cell. It is produced by the fusion of immune cells that can produce this antibody with cancer cells.
As a targeted therapy drug, monoclonal antibody has the characteristics of strong specificity, significant curative effect and low toxicity. It can effectively prevent the virus from entering the cell to proliferate. It can be used as a short-term prevention for high-risk populations and can also be used for diseases after virus infection. Therefore, it is also a hot spot in global research on the prevention and control of the COVID-19 epidemic.
In this study, the experimental team used phage display technology to construct two high-capacity human phage libraries using Peripheral Blood Mononuclear Cell (PBMC) from 8 patients who recovered from the new coronavirus infection. Enzyme-linked immunosorbent assay (ELISA) was used to determine the RBD-specific antibody titers in each serum sample. In the end, 10 monoclonal antibodies with high neutralizing activity against the new coronavirus were successfully screened.
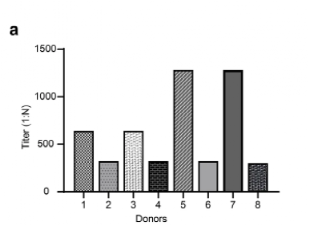
Figure 5. Plasma RBD-specific antibody titers in patients with different recovery periods. Image source: Cell Discovery
The crystal structure analysis data shows that the neutralizing antibody “2B11” has the strongest neutralizing activity against the original wild-type (WT) new coronavirus and its variant B.1.1.7, and the recognized RBD epitope binds to ACE-2. The high overlap of the dots can effectively block the binding of the new coronavirus to ACE-2 on the cell surface, thereby preventing it from infecting cells.
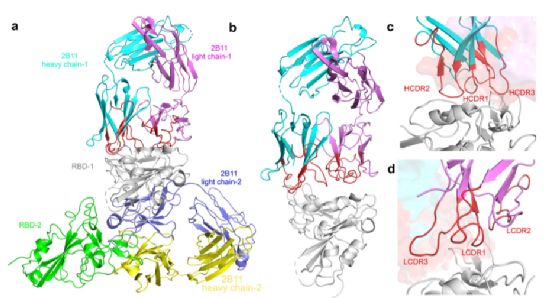
Figure 6. The overall structure of 2B11 and the new coronavirus RBD complex. Image source: Cell Discovery
At the same time, the research team also used a mouse model transduced with hACE2-adenovirus (hACE2-ADV) to evaluate the preventive and therapeutic effects of 2B11 on new coronavirus infections. The results showed that compared with the control group, the administration of 2B11 before or after the challenge could significantly reduce the weight loss and lung viral load caused by viral infection; further analysis of lung pathological tissue sections showed that the administration of 2B11 could significantly reduce Inflammation of the lungs caused by a viral infection.
For the Delta mutant strain, which has become the main variant of the global COVID-19 pneumonia transmission, the recent supplementary research results of this study show that 2B11 has a highly consistent neutralizing activity with the wild strain, indicating that 2B11 is mutated by Delta. The short-term prevention and early treatment of COVID-19 pneumonia caused by the virus strain has great application value.
In addition, another neutralizing antibody “1E10” against a different epitope on RBD showed complete inhibition of the new coronavirus mutants Alpha, Beta and Gamma in vitro in this experiment. The researchers said that while further using the neutralizing antibody 2B11, alone or in combination with other different epitope-targeting antibodies (such as 1E10), it may help to better short-term prevention and early treatment of new coronavirus infections.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



