Why Molnupiravir brings new hope for development of COVID-19 drugs?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Why Molnupiravir brings new hope for development of COVID-19 drugs?
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Why Molnupiravir brings new hope for development of COVID-19 drugs?
Since the outbreak of COVID-19 pneumonia in early 2020, scientists have been looking for effective prevention and treatment drugs.
Have you ever remembered that Remdesivir has been regarded as “the hope of the people” for a long time.
Fapilavir tablets were urgently produced, and chloroquine phosphate was unanimously recommended by experts to be included in the COVID-19 pneumonia diagnosis and treatment plan,
with an mRNA vaccine Let Moderna’s market value quickly jump above GSK…
Countries around the world have been looking for new hopes for the prevention and treatment of COVID-19 pneumonia.
Now, new hope is directed to Merck’s oral antiviral drug Molnupiravir. The origin of Molnupiravir, its characteristics, its clinical trials, its similar products, its imitation, etc., today we will analyze them separately.

Why Molnupiravir brings new hope for development of COVID-19 drugs?
Can Merck’s Molnupiravir defeat COVID-19?—No, but it will reduce hospitalization and mortality
On October 1, 2021, Merck announced that their oral antiviral drug Molnupiravir has reduced the hospitalization rate or death risk of patients with mild to moderate COVID by approximately 50%.
Merck plans to seek emergency use authorization in the United States and submit marketing applications to other drug regulatory agencies around the world.
This is the most appropriate time. Nearly 5 million people worldwide have died from COVID infection. In the United States alone, the death toll of COVID-infected people exceeds 700,000, more than the death toll caused by the infamous Spanish flu in 1918, which killed 675,000 Americans.
Considering that the United States has the world’s best medical resources, including well-trained doctors and the world’s most advanced medical equipment, this is indeed sad and shocking.
Why?
The key factor is selfishness disguised as a love of freedom. Those who refuse to receive safe and effective vaccines are too individualistic and pay too little attention to the community and humanity as a whole.
During the American Revolutionary War, Patrick Henry’s famous battle cry was “Either give me freedom or give me death!” Today, I suspect that this battle cry may be “Give me freedom, give me death!”, because super 99 % Of COVID-related deaths have not been vaccinated.
But still want to put aside the political field and return to the scientific level for professional analysis.
1. Discovered drugs for flu treatment at Emory University
Emory University in Atlanta, Georgia is a small private school. But in the past few decades, it has become famous for several antiviral drugs. Drugs earned the university a lot of royalties.
In the 1990s, inspired by a drug called lamivudine (Epivir), chemistry professor Dennis Liotta and virologist Raymond Schinazi discovered emtricitabine (Emit riva), an HIV nucleoside reverse transcriptase inhibitor Agent. In the end, it was “carried forward” in the hands of Gilead Sciences, and it was approved by the FDA in 2003.
It is worth noting that more than 90% of HIV-infected people receiving treatment in the United States take drugs containing lamivudine or emtricitabine. Emory University received more than 500 million U.S. dollars in royalties, and three inventors received 40% of royalties.
Professors Liotta and Schinazi once again used Pharmasett’s sofosbuvir for gold panning, which is an inhibitor of anti-hepatitis C virus NS5B polymerase. In 2011, even though only Sofosbuvir was a real asset, Pharmasett’s market value still reached 7 billion US dollars, which was on par with Vertex.
As one of the founders, Professor Schinazi holds 4.4% of the company’s shares and received US$440 million from the transaction. At the end of 2013, sofosbuvir was approved by the FDA to treat hepatitis C in combination with ribavirin and sold at a price of $1,000 per tablet.
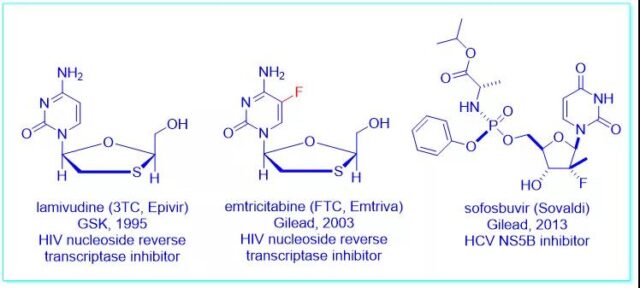
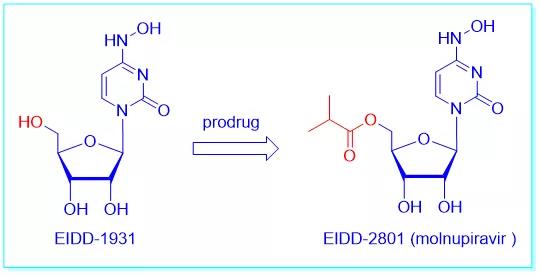
In 2013, Emory University established the Emory Institute for Drug Discovery (EIDD), and George Painter, a veteran of GlaxoSmithKline, served as the CEO. In 2014, they discovered through screening that N-hydroxycytidine (NHC, EIDD-1931) is an antiviral drug.
But EIDD-1931 is too polar to penetrate the cell membrane. In order to improve bioavailability, they used an old trick in drug design: creating a prodrug. The hydroxyl is converted to the corresponding isobutyrate to produce EIDD-2801, which will be converted to Molnupiravir when appropriate.
When Painter and his collaborators started testing EIDD-2801, they were surprised to find that the drug showed broad-spectrum activity against many RNA viruses, including influenza, hepatitis C, Ebola virus, chikungunya virus, and equine brain. Inflammation and coronaviruses, such as SARS-CoV-1 and MERS.
For example, EIDD-2801 shows a significant therapeutic effect on ferrets infected with influenza virus (an animal commonly used to study human respiratory viruses).
In 2015, the Emory team contacted researchers at Vanderbilt University to verify whether EIDD-2801 could fight the coronavirus through research, and he did it.
COVID-19 pneumonia came out in early 2020, and the focus of the medical and scientific communities quickly shifted to the evaluation of EIDD-2801 as a treatment method for large-scale drug reuse on a global scale. In March 2020, Emory authorized EIDD-2801 to Ridgeback, a small biotherapy technology company in Miami, for human research.
Ironically, many people think that Molnupiravir will be too toxic to be used as a drug because of its mutagenic properties. In April 2020, Rick Bright, the director of the U.S. Biomedical Advanced Research and Development Agency (BARDA) who was fired by Trump, complained that Ridgeback Biotherapeutics had pressured him to provide funds to manufacture EIDD-2801, despite Rick Bright’s complaints. Its safety expressed concern.
In May 2020, Ridgeback Biotherapeutics began a clinical trial of EIDD-2801 in cooperation with Merck. Merck gave it a code name: MK-4482, which later became Molnupiravir, and began phase II/III clinical trials in October 2020.
Currently, Merck is forced to terminate the clinical trial of Molnupiravir. Although the drug is safe and effective for the treatment of patients with mild to moderate COVID, it reduces the hospitalization rate and mortality rate. However, it is unethical to continue to give placebos to patients enrolled in the trial. So Merck now administers Molnupiravir to the subjects.
Merck will now issue an emergency use application to the FDA. Hope the approval is coming soon!
2. The mechanism of action of Molnupiravir
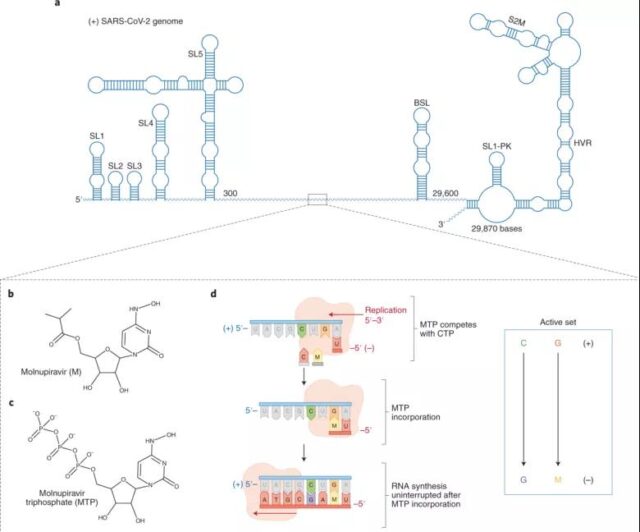
The mechanism of action of Molnupiravir is well known: Just like Gilead’s Remdesivir, Molnupiravir kills the coronavirus by blocking the synthesis of RNA polymerase (RdRp).
Molnupiravir is easily metabolized by plasma esterase into the active drug EIDD-1931 after being taken orally. It can exist in two forms: one form mimics cytidine and forms a single bond between carbon and N-OH group; the other form mimics uridine and forms between carbon and N-OH group Double bond.
These two forms are called tautomers, and the conversion between them can cause mismatches in the transcription process. Therefore, when the virus grows in the presence of EIDD-1931, its RNA-dependent RNA polymerase reads the compound in the form of uridine instead of cytidine, thereby replacing uridine with cytidine.
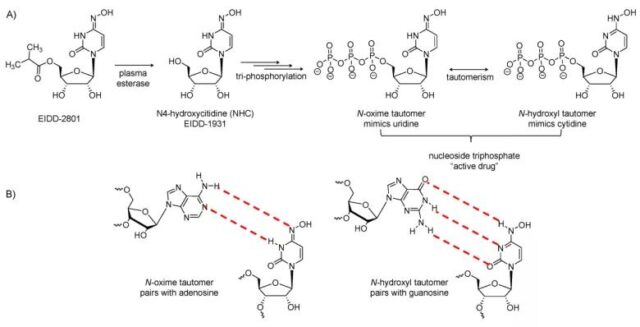
In short, EIDD-1931 works by interfering with the synthesis of RNA. In the process of viral RNA synthesis, the incorporation of this molecule will lead to subsequent misreading of base pairs, resulting in a high mutation rate, and ultimately the death of the genome.
Because the drug forces the coronavirus to make a lot of mistakes when it replicates, the virus cannot be synthesized. Therefore, some people are worried that it is mutagenic and has potential risks to humans. However, Merck’s comprehensive tests have shown that high-dose drugs will not have mutagenic effects on animals, and the Phase 3 clinical trials have not found that they have mutagenic effects on humans.
Now, Merck has proven that Molnupiravir is safe and effective, and it has changed the way we fight the coronavirus.
First of all, it is an oral medicine, which is much more convenient than remdesivir and neutralizing antibodies. Second, Molnupiravir can fight a variety of viruses, which means that it can also be used to treat other viral infections. The third and most intriguing feature is that Molnupiravir seems to have a high barrier to resistance.
Usually a drug will force the virus to rapidly develop a mutant that is not affected by the drug, and then make the drug useless. In laboratory tests, although “expected” to produce mutants, it was not found that Molnupiravir promotes resistance in animals, which means that Molnupiravir is effective against all variants, including the Delta mutant strain.
3. In the future, there will be antiviral drugs from Pfizer and Roche
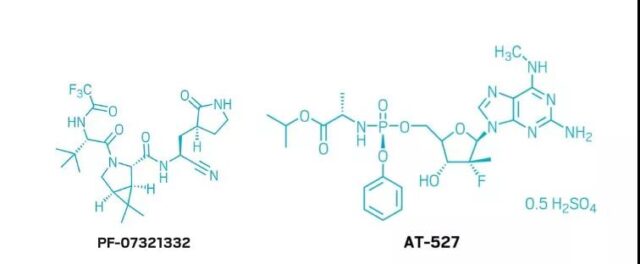
Pfizer’s PF-07321332 followed closely behind, and Phase II/III clinical trials began in early September 2021. Unlike Molnupiravir, PF-07321332 inhibits the 3CL protease of coronavirus. It is speculated that PF-07321332 is also a CYP3A4 substrate, which is why it can be co-administered with AbbVie’s ritonavir to increase its oral availability.
At the same time, Atea Pharmaceuticals cooperated with Roche to develop AT-27. AT-27, like Remdesivir, can inhibit the production of RNA-dependent RNA polymerase. But it also has certain activity on viral non-structural protein-12 (NSP 12) polymerase. AT-27 is farther in the R&D pipeline.
In any case, the success of Molnupiravir is great news that the world desperately needs. Through the combination of vaccines and oral antiviral drugs, I hope to defeat the invisible enemy of coronavirus in the near future.
Science will win!
Slow resistance and low production cost
—The success of Malnupravir is another victory for nucleoside antiviral drugs
Malnupravir is expected to become the first oral anti-coronavirus drug approved for marketing, which is another verification and victory of nucleoside antiviral drugs. In the treatment of many major viruses such as HIV, HBV, HCV and other diseases, nucleoside antiviral drugs are undoubtedly the most important class of antiviral drugs.
Generally speaking, virus polymerase is less prone to mutation than virus surface protein. The binding site of nucleoside antiviral drugs is the active area of polymerase, so nucleoside antiviral drugs are relatively resistant to resistance. Slow, this is also the reason why Redecive and Malnupravir still have high inhibitory activity against a variety of mutant strains including Delta, Gamma and Mu.
Compared with Remdesivir (Remdesivir), another important advantage of Malnupravir is that its structure is simpler, there is no complicated phospholipid prodrug part (Protide), and the synthetic route is simpler. It can be passed through cheap cytosine. Simple amino conversion is obtained, which is easy for rapid mass production, and the production cost is also lower.
In view of the possible continuous mutation of the COVID-19 in the future and the possible emergence of new epidemics caused by new RNA viruses, it is an important strategy to deploy broad-spectrum anti-RNA virus drugs in advance for potential RNA viruses.
Due to the structural similarity of the RNA virus replication key polymerase (RdRp), the development of broad-spectrum anti-RNA virus drugs is extremely feasible.
The correct clinical research protocol is the key to the success of Molnupiravir’s phase III
From the online information, the special highlight of the Molnupiravir clinical trial design is: the entry standard is laboratory-confirmed mild to moderate COVID-19, and there is at least one risk factor related to the severity of the disease (such as obesity, age> 60). Age, diabetes, and heart disease), and symptoms appeared within 5 days before randomization.
The main efficacy indicator is the percentage of hospitalizations and/or deaths within 29 days after randomization. This is the real clinical benefit.
1. Review of Remdesivir research
Looking back at the clinical study of Remdesivir in China a year ago, the study was divided into two groups: mild and severe. Compared with the clinical study of Molnupiravir, the selection criteria for the critical group of the clinical study of Redecivir is the main evaluation index of the phase III clinical study of Molnupiravir.
From a clinical point of view, in the severe stage, the use of antiviral drugs is already too late. The main evaluation index of the Redecive mild group is the clinical recovery time. However, the length of the clinical recovery time is not important. Whether it becomes severe or causes hospitalization is the key.
According to the information obtained on the Internet, pre-clinical studies have shown that Molnupiravir is more active against the new coronavirus in vitro than remdesivir, and it is still effective against the remdesivir-resistant coronavirus. Its IC50 for the new coronavirus SARS-CoV-2 in the Vero cell line is 0.3uM, which is stronger than the 0.77uM of Remdesivir.
Although Molnupiravir is more active than remdesivir, it does not mean that remdesivir is ineffective. If safety permits, it is enough to increase the dose of remdesivir.
The purpose of clinical research is to reflect the efficacy of the product under the premise of subject safety through rational design.
Merck completely got rid of the influence of the previous Remdesivir program in the design of the Molnupiravir program, which is also the key to the success of the Molnupiravir research.
2. Factor analysis of the success of Molnupiravir Phase III clinical trial
First, the Molnupiravir study selected the right subjects. For viral infections such as COVID-19 pneumonia, antiviral treatment must be timely. This is similar to the antiviral treatment of influenza.
Molnupiravir selected subjects who had symptoms within 5 days. Once the initial antiviral window is missed, the symptoms of COVID-19 pneumonia are mainly caused by the body’s immune response, and it does not matter whether the patient has the virus in the body.
At the same time, due to the low hospitalization and mortality caused by the COVID-19 virus among healthy young people, subjects with advanced age, obesity, and other underlying diseases were selected.
Among these subjects, the hospitalization rate and mortality rate after the COVID-19 infection are higher.
Choosing such subjects can reduce the sample size. From the results of the Phase III study, of the 377 patients in the placebo group, 45 were hospitalized and 8 died, with a mortality rate of 2.12%. This mortality rate is also higher than that of healthy young people.
Secondly, the Molnupiravir study is also very cautious in the selection of the main evaluation indicators. First of all, hospitalization and mortality are clinical benefits and direct evidence of the efficacy of Molnupiravir.
At the same time, the Molnupiravir study did not use mortality alone as the main evaluation index. Because the mortality rate is low, the sample size calculated by the mortality rate will be relatively large.
For example, if the mortality rate of the control group is estimated at 2.12%, it is necessary to control the mortality rate of the test group at 0.3% to obtain a sample size of 1550. This is undoubtedly a challenge. Moreover, this study was designed according to the method of Adaptive Design.
Due to the consumption of a value, the mortality rate needs to be controlled at a lower level. Judging from the results of the trial, there were 385 patients in the experimental group and 0 deaths; in the placebo group, there were 377 patients and 8 deaths. This result is very lucky.
If 1 of the 385 patients in the experimental group died, there would still be a significant difference between the two treatment groups, but once there were 2 deaths, the P value was greater than 0.05, and the interim analysis would fail.
Therefore, the combined calculation of the hospitalization rate and the mortality rate increases the overall assurance of the research.
According to the data of the interim analysis, 28 of the 385 patients in the experimental group were hospitalized and died, and 53 of the 377 patients in the placebo group were hospitalized and died.
Even if the trial group adds 8 more hospitalized patients, that is to 36 cases, a significant difference can still be obtained. Therefore, this design is a relatively safe design.
Third, the method of Adaptive Design was adopted, and the results of the interim analysis were used for reporting. The interim analysis of Adaptive Design itself consumes a value. If there is no significant difference in the results of the interim analysis, the final analysis will face greater challenges.
Moreover, as mentioned earlier, Adaptive Design itself will also lead to an increase in the total sample size. This is the risk of Adaptive Design. However, Merck’s R&D personnel made the right decision through risk analysis, which greatly advanced the approval of the drug.
Molnupiravir compound patent or until 2038
Similar to Remdesivir, Molnupiravir is also based on a known nucleoside antiviral compound. Emory University discovered that EIDD1931 compounds can be used as broad-spectrum antiviral drugs to prevent the replication of a variety of RNA viruses, including influenza viruses, SARS and MERS viruses, Ebola viruses, and respiratory syncytial viruses.
However, unlike Remdesivir, although Gilead applied for compound patents, the use patents for inhibiting the COVID-19 virus were first listed by Wuhan University. Emory University has already low-keyed the priority application in 2020, and in 2021 The PCT international patent application was filed in February.
Patent for the use of Molnupiravir to inhibit the new coronavirus
The latest application of Molnupiravir to inhibit new coronavirus patent publication number: WO2021159044A1, title of invention: N4-hydroxycytidine and derivatives and Anti-viral uses related thereto (N4-hydroxycytidine and derivatives and anti-viral uses related thereto) A pharmaceutical composition for treating COVID-19 pneumonia infection, wherein the active ingredient is a compound of formula XXI
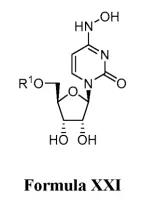
The examples verify the activity of EIDD-1931 and its isobutyrate prodrug EIDD-2801 (ie, Molnupiravir compound) in inhibiting the new coronavirus.
The filing date of this patent is February 7, 2021. Based on three US priority patents, they were filed on February 7, March 11, and March 25, 2020.
Why Molnupiravir brings new hope for development of COVID-19 drugs?
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



