How to treat patients with brain metastases from HER2-positive breast cancer?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
How to treat patients with brain metastases from HER2-positive breast cancer?
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
How to treat patients with brain metastases from HER2-positive breast cancer? ESMO official paper for you
Interpretation of MDT-guided treatment strategies for brain metastases from HER2-positive breast cancer.
Breast cancer is prone to brain metastasis, and the proportion of HER2-positive breast cancer patients with central nervous system (CNS) metastasis is 30% to 55% [1] .
Although anti-HER2 targeted therapy prolongs the survival of breast cancer patients and improves the control rate of extracranial lesions, the existence of the blood-brain barrier greatly weakens the killing effect of most systemic drugs on brain tumors, making breast cancer brain Metastasis has become a difficult problem in clinical treatment.
Once HER2-positive breast cancer develops brain metastases, its prognosis is poor, and the effective management of such patients has become a major clinical challenge [2] .
Recently, ESMO Open , the official journal of the European Society for Medical Oncology (ESMO), published a review titled “How we treat HER2-positive brain metastases” [3] , which outlined the treatment methods for HER2-positive breast cancer brain metastases. The relevant content is now interpreted as follows for readers.
Characteristics and course development of brain metastases from HER2-positive breast cancer
The incidence of brain metastases varies by breast cancer subtype and stage, and especially in patients with advanced breast cancer, HER2 overexpression is associated with an increased risk of brain metastases. On the one hand, overexpression or abnormal amplification of HER2 makes breast cancer cells more aggressive and thus more prone to metastasize to the brain.
On the other hand, in the era of anti-HER2-targeted therapy, the application of anti-HER2 therapy in HER2-positive breast cancer patients has greatly improved the survival of patients, and the CNS may become a HER2-overexpressing breast cancer due to its biological specificity (blood-brain barrier) . “Refuge” of cancer cells [3] .
For patients with HER2-positive early-stage breast cancer receiving breast-conserving therapy and systemic therapy, the 10-year risk of developing brain metastases is approximately 12%. For patients with HER2-positive advanced breast cancer, about 50% develop brain metastases during the disease period.
In the HERA trial, after a median follow-up of 4 years, the risk of a CNS event as the initial site of recurrence was low (2% for both trastuzumab and observation, P=0.55) .
Among patients who died in the HERA trial, there was a trend toward a reduction in CNS events in patients initially treated with trastuzumab (47% vs. 57%) , but this difference remained non-significant (P=0.06) , and The incidence of CNS events in both groups was very high among these deaths.
The findings suggest that the incidence of brain metastases in patients with metastatic HER2-positive breast cancer may also increase as the disease progresses.
This finding also led to thinking about the risk of brain metastases under anti-HER2 therapy for HER2-positive breast cancer [3] .
To date, no adjuvant therapy (including trastuzumab, pertuzumab, lapatinib, or T-DM1) has been shown to prevent CNS recurrence. For example, in the ALLTO trial, no difference in the incidence of the CNS as the first site of recurrence was observed (2% in both the lapatinib plus trastuzumab and mAb groups) .
Similarly, in the KATHERINE trial, 1486 patients with residual disease after neoadjuvant anti-HER2 therapy were randomized to continue trastuzumab or switch to T-DM1 therapy.
Although there was a significant reduction in metastatic events in the T-DM1 group, there was no reduction in the risk of the CNS as the first site of recurrence, occurring in 5% of patients in both groups, and brain metastases accounted for more than half of distant recurrences in the T-DM1 group.
In the T-DM1 group, the CNS was the only site of recurrence in 4.8% of patients compared with 2.8% in the trastuzumab group.
In the ExteNET trial, 8-year follow-up data for adjuvant neratinib in early-stage HER2-positive breast cancer showed that the incidence of CNS metastases was 1.3% in the neratinib group and 1.8% in the placebo group. These findings together demonstrate that anti-HER2 adjuvant therapy does not prevent the occurrence of brain metastases[3]。
Treatment of brain metastases from HER2-positive breast cancer
The treatment of brain metastases from breast cancer should implement a multidisciplinary diagnosis and treatment model.
The purpose of treatment is to control the metastatic lesions, improve the symptoms of patients, improve the quality of life, and maximize the survival time of patients.
Breast cancer brain metastases are treated with surgery, radiotherapy, drug therapy, and symptomatic and supportive care.
The overall treatment principle is to give priority to surgery and/or radiotherapy for brain metastases under the premise of full assessment of the systemic situation, and to consider systemic treatment reasonably [4] .
At present, local therapy including surgery, stereotactic radiosurgery (SRS) and whole brain radiation therapy (WBRT) are still the main treatments for breast cancer.
Symptoms, number, size, distribution of metastases, and past treatment decisions. How these treatment options are selected and how they are deployed is a decision made by an experienced multidisciplinary team.
In general, surgery is suitable for patients with single-lesional brain metastases without extracranial lesions or with controlled extracranial lesions, and the brain lesions are large or involve enlargement, etc.
For patients without prior diagnosis of metastatic disease, surgery may also provide tissue from the lesion to confirm the status of metastatic disease.
In the perioperative period, radiation therapy to the surgical site is generally recommended to improve local control.
For single, small, and inaccessible brain lesions, first-line SRS is an alternative to surgery. For patients with multiple brain metastases, SRS is very suitable for patients with a limited number of lesions.
Data from controlled studies show that SRS is superior to WBRT in patients with brain metastases with up to 4 lesions.
In an ongoing randomized controlled study comparing the efficacy of SRS and WBRT in patients with 5-20 brain lesions, results are currently inconclusive.
In view of its acute and chronic toxicity, WBRT is generally considered to be avoided first, but in the case of multiple intracranial lesions and multiple large lesions, WBRT is usually required.
Systemic therapy may also be considered as first-line therapy in newly diagnosed HER2-positive patients with limited brain metastases and asymptomatic or mild symptoms.
Systemic therapy may also be considered if CNS progression develops after local therapy [3] .
When patients with brain metastases need to be considered for systemic therapy, the following treatment strategies are available depending on the clinical situation (Figure 1 and Table 1) .
Table 1. Systemic treatment options for HER2-positive brain metastases [3]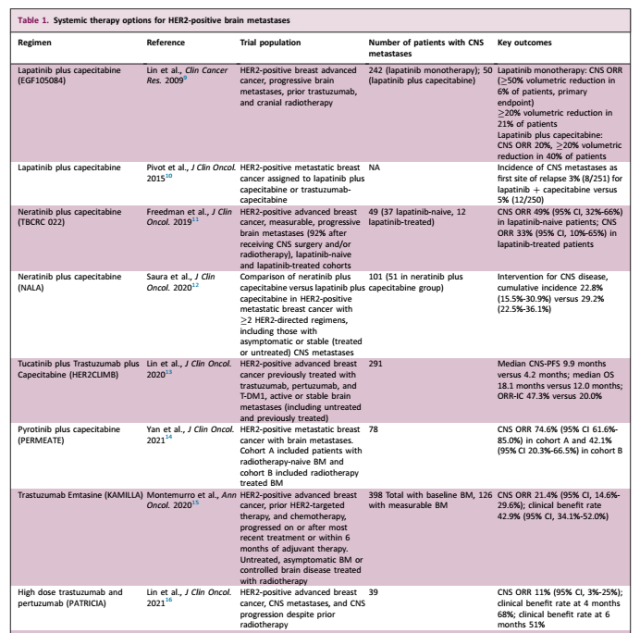
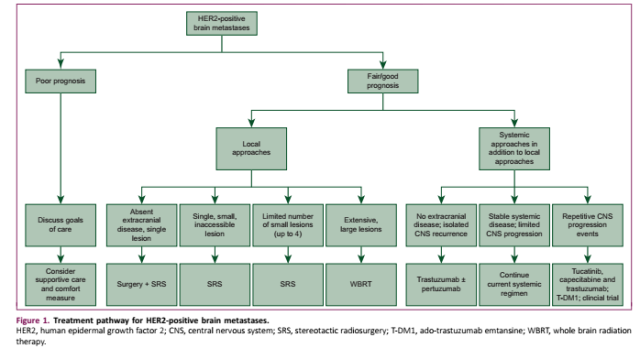 Figure 1. Therapeutic pathways for HER2-positive brain metastases [3]
Figure 1. Therapeutic pathways for HER2-positive brain metastases [3]
Patients without any symptoms of systemic disease but with CNS recurrence
For patients with CNS metastases without signs of extracranial disease, CNS progression is mainly treated with surgery and/or radiotherapy, and there is currently a lack of prospective studies to guide treatment options other than local therapy.
However, trastuzumab or trastuzumab + pertuzumab (and endocrine therapy in patients with estrogen receptor-positive disease) is usually used clinically after local treatment of CNS disease [3] .
Patients with stable systemic disease but localized CNS progression
For patients with stable systemic disease but local CNS progression, SRS is usually used to treat progressive CNS disease, and existing systemic therapy is continued. Among them, anti-HER2 monoclonal antibody can be continued throughout the course of radiotherapy.
If the patient is receiving chemotherapy, chemotherapy can be suspended and resumed 1-2 weeks after the completion of radiotherapy.
Patients with repeated CNS progression events within shorter time intervals tend to delay SRS and switch to systemic therapy, both to help control systemic lesions and to have potential efficacy in micrometastatic disease.
In general, this drug is recommended for patients who have not received T-DM1. Second, for patients who have progressed during T-DM1 treatment in the past, the use of tucatinib + capecitabine + trastuzumab triple therapy in patients with brain metastases tends to increase the patient’s CNS remission rate, no Progression survival (PFS) and overall survival (OS) , based on the 1-year PFS rate of 24.9% in the tucatinib triple therapy arm compared to placebo in 291 patients with brain metastases in the HER2CLIMB trial Only 0% (HR=0.48, 95%CI 0.34-0.69, P<0.001) .
And in an exploratory analysis of patients with brain metastases, the confirmed intracranial objective response rate (ORR) was 47% and 20% , respectively (P=0.03) , and the median intracranial response duration was 6.8 months and 3.0 months, respectively. , the median OS was 18.1 months and 12.0 months, respectively. If tucatinib is not available, lapatinib + capecitabine or neratinib + capecitabine can be used instead.
Based on the evidence of efficacy of tucatinib in patients with brain metastases from the HER2CLIMB trial, the ongoing COMPASS-RD trial (NCT03975647) aims to evaluate whether tucatinib plus T-DM1 can effectively improve the efficacy of adjuvant tucatinib plus T-DM1 in patients with high-risk residual disease. Invasive disease survival (IDFS) , but the study did not include patients previously treated with HER2-targeted tyrosine kinase inhibitors (TKIs) or ADCs.
For patients with CNS progression despite T-DM1 and HER2-targeted TKIs, other systemic therapies (eg, trastuzumab combined with carboplatin or liposomal doxorubicin, or high-dose trastuzumab) may be considered in combination with pertuzumab) , but the evidence is usually based on small nonrandomized studies or case reports.
Although both tucatinib and neratinib responded to treatment with HER2-targeted TKIs, overall, the efficacy of sequential HER2-targeted TKI therapy has not been fully validated [3] .
Patients with stable systemic disease but CNS progression ( eg, multiple CNS minimal lesions )
In patients with stable or remission of systemic disease but CNS progression with many small CNS lesions that are not candidates for SRS, WBRT (if not already given) is recommended with systemic therapy.
If the patient is not already receiving tucatinib + capecitabine + trastuzumab, the combination therapy should be considered with close follow-up to delay the onset of WBRT and its associated toxicity. For patients who have received WBRT, consider systemic therapy (if not already receiving tucatinib, clinical trials, or other regimens ) [3] .
Patients with extensive CNS progression
Although WBRT is clinically avoided as much as possible based on safety considerations, for patients with extensive CNS progression, WBRT can effectively control morbidity and mortality, and often has a good treatment response [3] .
Patients with concurrent CNS and extracranial progression
For patients with simultaneous CNS and systemic progression, it is recommended to follow the guideline-recommended treatment strategy for advanced HER2-positive breast cancer, but the regimen with CNS therapeutic activity is preferred [3] .
Other handling opinions
In view of the poor overall prognosis of patients with brain metastases in the past, the overall incidence of radiation brain necrosis reported in breast cancer brain metastases is low, especially symptomatic brain necrosis, the incidence rate is only 6%-11%.
However, under the background of comprehensive treatment, the survival of HER2-positive breast cancer has been significantly prolonged, and the reports of related radiation brain necrosis have gradually increased.
Clinicians should note that, in some cases, a condition that shows progressive CNS disease on radiology after SRS may be radiation necrosis. If radiation necrosis is suspected, clinical follow-up will be performed with interval imaging.
If symptomatic cerebral necrosis occurs, glucocorticoids, represented by dexamethasone, are the classic first-line treatment because they often have symptoms of intracranial hypertension.
In addition, anti-vascular endothelial growth factor drugs can theoretically prevent the destruction of the blood-brain barrier, thereby reducing vasogenic edema and related symptoms.
Some clinical studies have also shown that related drugs can improve the imaging manifestations of radiation brain necrosis [3] .
Clinical trial method
With the increased understanding of the biology of HER2-positive brain metastases, many clinical trials are currently exploring new approaches to the management of HER2-positive CNS metastases, and participation in such trials is encouraged if feasible.
Although the blood-brain barrier makes the delivery of some systemic therapeutics challenging, most drugs appear to penetrate the CNS when the barrier is disrupted, albeit sometimes at lower concentrations.
In addition, the genetic differences of breast cancer brain metastases and the differences in the CNS tumor microenvironment are also research directions worthy of exploration, which will help to explore more effective treatment options [3] .
Summary
(How to treat patients with brain metastases from HER2-positive breast cancer?)
The treatment of HER2-positive brain metastases is complex and requires the participation of an experienced multidisciplinary team.
A series of clinical studies have been conducted to explore the activity of existing treatment options in patients with brain metastases, and new drugs with therapeutic potential are being developed.
As the occurrence of breast cancer brain metastases becomes more and more common, the need to improve the prognosis of breast cancer brain metastases is more urgent.
It is particularly important to develop more effective treatment strategies to manage and ultimately prevent CNS diseases in the future [3] .
references:
[1]Lin NU, Amiri-Kordestani L, Palmieri D, et al. CNS metastases in breast cancer: old challenge, new frontiers[J]. Clin Cancer Res, 2013, 19(23): 6404-6418.
[2] Ma Li, Wang Hui, She Chunhua, et al. Research progress on brain metastases from breast cancer [J]. Chinese Journal of Breast Diseases: Electronic Edition, 2015, 9(3):6.
[3]Stavrou E, Winer EP, Lin NU. How we treat HER2-positive brain metastases. ESMO Open. 2021 Oct;6(5):100256.
[4] 2021 CSCO Breast Cancer Diagnosis and Treatment Guidelines
How to treat patients with brain metastases from HER2-positive breast cancer?
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



