Harvard: PD-1 inhibitors can promote the normalization of tumor blood vessels
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Harvard: PD-1 inhibitors can promote the normalization of tumor blood vessels
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Harvard: PD-1 inhibitors can promote the normalization of tumor blood vessels.
JNCI: Harvard team found that PD-1 inhibitors can promote the normalization of tumor blood vessels, opening up new ideas for liver cancer treatment.
Recently, the National Cancer Center released the latest national cancer statistics. The report shows that liver cancer is one of the five cancers with the highest incidence in some countries, and its mortality rate is also at the forefront. Among them, hepatocellular carcinoma (HCC) is the main histological subtype of liver cancer, accounting for 75%-85% of primary liver cancer [1]. How to better treat HCC has always been the direction of researchers’ efforts.
Sorafenib has anti-vascular endothelial growth factor receptor (VEGFR1-3) activity and is a multi-kinase inhibitor for the treatment of unresectable or distant metastatic hepatocellular carcinoma, and is the first treatment for patients with advanced HCC. systemic therapy. However, as an anti-angiogenesis targeted drug, a problem that cannot be ignored is drug resistance.
Recently, Dan G Duda’s team from Harvard Massachusetts General Hospital published important research results in the Journal of the National Cancer Institute (JNCI) [2], providing a feasible solution to the above problems.
They found that treatment of HCC with PD-1 inhibitors followed by continued treatment with the anti-angiogenic drug sorafenib enhanced HCC response to sorafenib, promoted normalization of tumor blood vessels, and induced an effective Anti-tumor immunity, this sequential combination therapy can achieve effective control of tumor growth and improve mouse survival .
This opens up new ideas for improving the efficacy of immune checkpoint inhibitors and anti-angiogenic drugs, providing a translatable strategy for clinical treatment of hepatocellular carcinoma (HCC).

Immune checkpoint inhibitors have been available for many years, and many tumor patients have benefited from them, but their effects in the treatment of hepatocellular carcinoma are not satisfactory. However, the combination of anti-angiogenic drug bevacizumab and immune checkpoint inhibitor PD-L1 inhibitor has achieved good therapeutic effect [3]. Based on this, researchers combined multi-kinase inhibitors with immune checkpoint inhibitors, hoping to overcome drug resistance and achieve synergistic treatment effects in a reasonable order of administration.
Next, let’s take a look at how this research is carried out.
The researchers first conducted a retrospective analysis of 25 patients with symptoms of cirrhosis who developed hepatocellular carcinoma (HCC) who received sorafenib after at least one dose of a PD-1 inhibitor. . Of these, 17 patients (68%) received nivolumab, 3 patients (12%) received pembrolizumab, and 5 received other anti-PD-1 antibodies.
Surprisingly, these HCC patients had an overall response rate of 12% to sorafenib and a median overall survival of 12.1 months . In clinical trials conducted in patients with advanced HCC in Europe and Asia Pacific, the response rate to sorafenib was only 2%-3%, and the median overall survival in the two studies was 6.5 months and 6.5 months, respectively. 10.7 months [4-5]. In contrast, the sequential treatment regimen showed certain advantages.
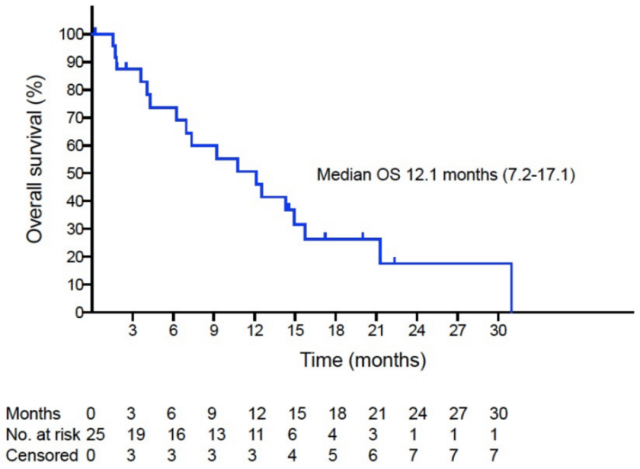
PD-1 inhibitor combined with sorafenib sequential therapy improves patient survival
Therefore, the researchers verified the priming effect of PD-1 inhibitors in this sequential therapy in animal models, and explored the mechanism of action of sequential combination therapy.
To demonstrate that PD-1 inhibitors can enhance patient response to standard sorafenib therapy (high dose, 40 mg/kg), the researchers used a PD-1 inhibitor – sensitive murine hepatocellular carcinoma cell line (RIL-175), established a mouse orthotopic liver cancer model. Tumor-bearing mice continued to receive sorafenib (40 mg/kg, orally daily) after receiving three PD-1 inhibitor treatments over a week.
As expected, sequential combination therapy (PS group) inhibited tumor growth and significantly prolonged the mice’s median progression-free survival, with 22% of the mice experiencing complete remission .
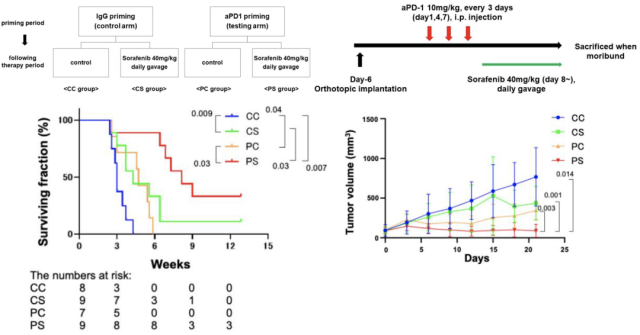
Sequential therapy inhibits HCC tumor growth in mice
Both immune checkpoint inhibitors and anti-angiogenic drugs affect immune cells in the tumor microenvironment, which in turn affects the efficacy of treatment. The researchers also repeated the sequential combination therapy in tumor-bearing mice and harvested tumor tissue from the mice to assess changes in CD8+ and CD4+ T lymphocytes.
The results showed that after sequential treatment, compared with the PD-1 inhibitor treatment group, the intratumoral infiltrating CD8+ T lymphocytes were significantly increased, while the intratumoral infiltrating CD4+ T lymphocytes were not significantly changed .
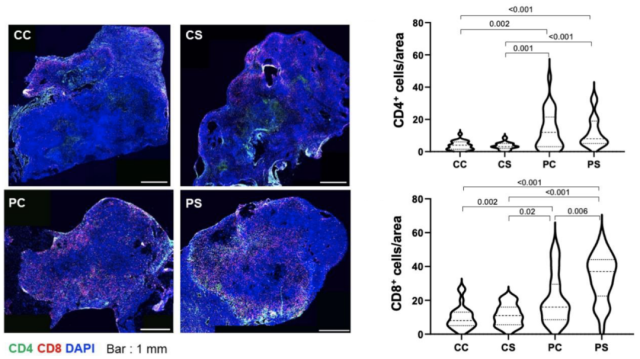
Sequential combination therapy increases infiltration of CD8+ T lymphocytes
In the tumor microenvironment, in addition to immune cells, there are also intricate vascular systems. These tumor blood vessels are different from normal blood vessels, with incomplete structures and abnormal functions.
Studies have confirmed that the combination of immune checkpoint inhibitors and anti-VEGFR2 anti-angiogenic drugs can promote the normalization of tumor blood vessels in HCC, which is conducive to tumor control [6]. Therefore, the researchers also focused on the effect of this sequential treatment on the intratumoral blood vessels of HCC.
The researchers assessed CD31+ microvessel density (CD31 is a marker of endothelial cells) in tumor samples , as well as pericyte coverage of these tumor microvessels. In general, treatment with sorafenib can effectively inhibit tumor angiogenesis.
Surprisingly , the sequential therapy with PD-1 inhibitor followed by sorafenib did not reduce the density of intratumoral microvessels, but induced more angiogenesis , which is worth mentioning Interestingly , these newly formed tumor vessels also had higher pericyte coverage, implying a normalization of the structure of these vessels .
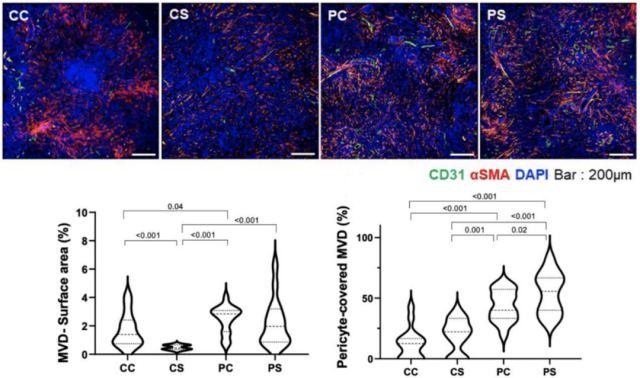
Sequential therapy improves tumor vascular structure
Here, the researchers confirmed improvements in the intratumoral microenvironment—increased T lymphocytes (especially CD8+ T lymphocytes) , and normalization of tumor vascular architecture. So, is the efficacy of the combination of PD-1 inhibitors and sorafenib mediated by CD8+ T cells? The researchers continued to explore.
It is still a familiar formula. First, the HCC tumor-bearing mice are given the leading PD-1 inhibitor treatment, and then the sorafenib treatment is given.
The difference is that a group of tumor-bearing mice was given sorafenib and also treated with Antibodies that deplete CD8+ T lymphocytes were received.
It can be seen that compared with the control group, sequential therapy (PS group) effectively improved the survival of tumor-bearing mice, and this benefit disappeared when CD8+ T lymphocytes (PDS group) were removed . But interestingly, when CD8+ T lymphocytes were reduced, the rate of tumor growth was still delayed.
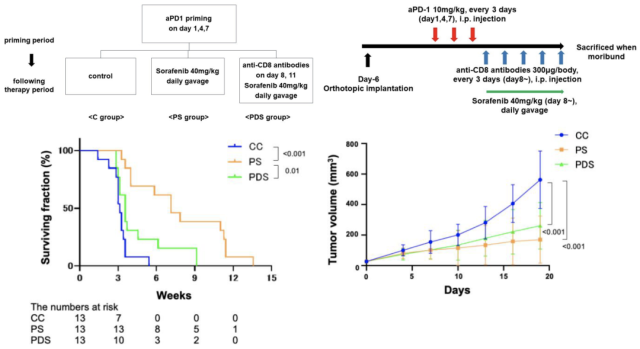
Depletion of CD8+ T lymphocytes affects the survival of tumor-bearing mice after sequential therapy
In 2017, Tian et al. demonstrated that in a breast cancer tumor model, effective anti-tumor immunity interacts with tumor vascular normalization, and this regulation is mainly achieved by CD4+ T lymphocytes [7].
Zheng et al believed that the positive feedback loop between anti-tumor immunity and tumor vascular normalization relies on CD8+ T lymphocytes in the treatment of breast cancer with immune checkpoint inhibitors [8]. So, which subsets mediate antitumor immunity and vascular normalization in HCC tumor models?
When anti-CD8 antibody and sorafenib were used at the same time, the intratumoral CD8+ T lymphocytes almost completely disappeared. At the same time, compared with the PS group, the intratumoral CD4+ T lymphocytes in the PDS group also decreased.
Not only that, but with the removal of CD8+ T lymphocytes, sequential therapy-mediated increases in microvascular and pericyte coverage disappeared.
That is to say, CD8+ T lymphocytes are the “main force” in this sequential therapy, affecting the changes of CD4+ T cells and mediating effective anti-tumor immunity and normalization of tumor blood vessels .
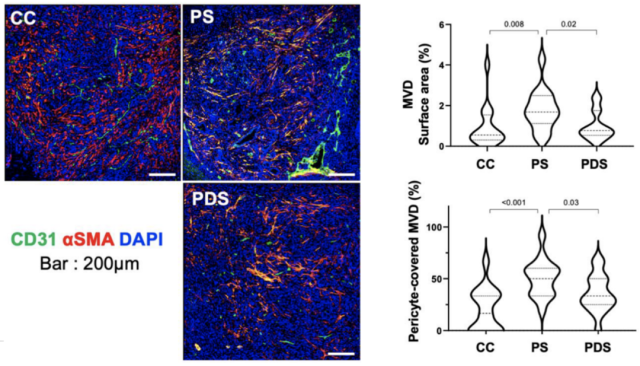
Depletion of CD8+ T lymphocytes affects tumor vascular structure
In conclusion, lead therapy with PD-1 inhibitor increased the infiltration of CD4+ and CD8+ T lymphocytes in HCC tumors, and sequential use of sorafenib after PD-1 inhibitor could protect tumor blood vessels and promote tumor growth.
Vascular normalization. In this sequential treatment regimen, sorafenib is more like an immune stimulator, promoting the infiltration of CD8+ T lymphocytes, promoting a positive feedback loop of anti-tumor immunity and tumor vascular normalization.
However, the mechanism by which CD8+ T lymphocytes protect tumor blood vessels and improve vascular structure remains to be explored.
Professor Rakesh K Jain, one of the authors of this paper, and his research team have been devoted to the study of tumor blood vessels for many years. In 2005, Professor Jain first proposed the concept of “tumor blood vessel normalization”: reducing the tortuosity and permeability of tumor blood vessels and reducing the interstitial space hydraulic pressure, thereby improving tissue hypoxia and promoting the delivery of drugs and immune cells to tumor tissue [9].
So far, some studies have confirmed that the rational use of anti-angiogenic drugs, combined with immune checkpoint inhibitors, can induce the normalization of tumor blood vessels, improve the tumor microenvironment, and generate effective anti-tumor immunity.
The PD-1 inhibitor was given first , and then sorafenib was given. This sequential treatment plan showed a certain therapeutic effect. If it can be further promoted in clinical treatment, it will bring good news to more HCC patients.
References:
[1] Brozzetti S, Tancredi M, Bini S, et al. HCC in the Era of Direct-Acting Antiviral Agents (DAAs): Surgical and Other Curative or Palliative Strategies in the Elderly. Cancers (Basel). 2021;13(12 ): 3025. Published 2021 Jun 17. doi: 10.3390/cancers13123025
[2] Kikuchi H, Matsui A, Morita S, et al. Increased CD8+ T-Cell Infiltration and Efficacy for Multikinase Inhibitors after PD-1 Blockade in Hepatocellular Carcinoma [published online ahead of print, 2022 Mar 15]. J Natl Cancer Inst .2022;djac051.doi:10.1093/jnci/djac051
[3] Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382(20):1894-1905. doi:10.1056/NEJMoa1915745
[4] Cheng AL, Kang YK, Chen Z, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10(1):25-34. doi:10.1016/S1470-2045(08)70285-7
[5] Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378-390. doi:10.1056/NEJMoa0708857
[6] Shigeta K, Datta M, Hato T, et al. Dual Programmed Death Receptor-1 and Vascular Endothelial Growth Factor Receptor-2 Blockade Promotes Vascular Normalization and Enhances Antitumor Immune Responses in Hepatocellular Carcinoma. Hepatology. 2020;71(4) :1247-1261. doi:10.1002/hep.30889
[7] Tian L, Goldstein A, Wang H, et al. Mutual regulation of tumour vessel normalization and immunostimulatory reprogramming. Nature. 2017;544(7649):250-254. doi:10.1038/nature21724
[8] Zheng X, Fang Z, Liu X, et al. Increased vessel perfusion predicts the efficacy of immune checkpoint blockade. J Clin Invest. 2018;128(5):2104-2115. doi:10.1172/JCI96582
[9] Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58-62. doi:10.1126/science.11 04819
Harvard: PD-1 inhibitors can promote the normalization of tumor blood vessels
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



