New breakthrough: CAR-T therapy can treat all blood cancers
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
New breakthrough: CAR-T therapy can treat all blood cancers
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
New breakthrough: CAR-T therapy can treat all blood cancers.
CAR-T cell therapy is made from each patient’s own T cells that are genetically engineered. T cells are collected from the patient’s blood, modified and expanded in the laboratory, and then re-infused into the patient’s body as a “living cell drug”.
Professor Carl June of the University of Pennsylvania led the research that led to the approval of the world’s first CAR-T cell therapy – Kymriah – in 2017. So far, the FDA has approved six CAR-T cell therapies, which are designed for specific target antigens of certain types of blood cancer cells. Therefore, each one has a relatively narrow range of treatment.
For example, Kymriah therapy targets CD19 on the surface of B cells and is used to treat relapsed/refractory acute B lymphoblastic leukemia (r/r B-ALL). To date, no cell therapy targeting a single antigen has been developed that can work in all different types of blood and bone marrow cancers.
On August 31, 2023, researchers from the Perelman School of Medicine at the University of Pennsylvania and Harvard University published a research paper titled: Epitope base editing CD45 in hematopoietic cells enables universal blood cancer immune therapy in the Science sub-journal Science Translational Medicine.
CD45 is present on the surface of almost all blood cells (including blood cancer cells), and this study used an adenine base editor (ABE) to develop a new “epitope editing” strategy that allows CD45 to function normally in the blood system, while not being recognized by CAR-T cells.
Using this “epitope editing” strategy to modify hematopoietic stem cells and CAR-T cells, after entering the body, these CAR-T cells targeting CD45 will not kill each other, nor will they kill the input epitope-edited hematopoietic stem cells, but they can kill blood cancer cells expressing normal CD45.

The co-corresponding author of the paper, Professor Carl June, director of the Center for Cellular Immunotherapy at the University of Pennsylvania, said that one of the drawbacks of CAR-T cell therapy is that each therapy must be developed separately based on the target of different cancer types. This study lays the foundation for a more universal approach that could potentially expand CAR-T cell therapy to all blood cancers.
CD45 is present on the surface of almost all blood cells and is usually highly expressed on blood tumor cells. If a CAR-T cell therapy targeting CD45 is developed directly, it can indeed eliminate blood tumor cells, but it will also eliminate other healthy blood cells of the patient, including red blood cells, platelets, and even bone marrow stem cells that produce new blood cells. In addition, T cells are also blood cells and usually express CD45, so CAR-T cell therapy targeting CD45 will self-destruct before entering the patient’s body.
To overcome this challenge, the research team used base editing technology to develop a new epitope editing strategy that involves gene editing of CAR-T cells and hematopoietic stem cells (HSCs) to change the “epitope” that binds CAR-T cells to CD45 protein. The base-edited CD45 is still functional, but differs greatly from normal CD45, so CAR-T cells targeting CD45 cannot recognize and attack them.
Therefore, this is essentially a combination of hematopoietic stem cell transplantation and CAR-T cell therapy. When these edited CAR-T cells targeting CD45 are inputted, they can kill blood cancer cells carrying normal CD45, including patient-derived acute myeloid leukemia, B-cell lymphoma and acute T-cell leukemia. But they will not kill each other or attack the transplanted epitope-edited hematopoietic stem cells. Unlike hematopoietic stem cells with direct gene knockout of CD45, epitope-edited hematopoietic stem cells can engraft, persist and differentiate to produce new blood cells.
The research team conducted a large number of experiments in cell and mouse models to test the feasibility of this epitope editing strategy. The results showed that this new method not only prevents CAR-T cells targeting CD45 from self-destruction or attacking epitope-edited hematopoietic stem cells, but also quickly destroys blood cancer cells. In mouse experiments, CAR-T cells targeting CD45 eliminated leukemia cells within three weeks after inputting and persisted for more than two months and were able to continuously kill leukemia cells.
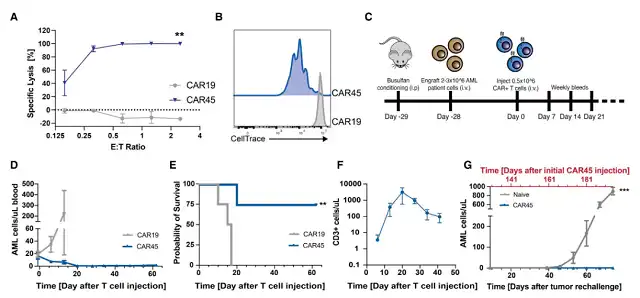
It is reported that the research team is conducting further toxicology experiments and validating in other models to prepare for entering human clinical trials.
New breakthrough: CAR-T therapy can treat all blood cancers
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



