mRNA Vaccine Targeting Bacteria to Prevent Lyme Disease from Tick Bites
- EB Virus Could Be Infected by Kiss: A Hidden Threat Linked to Cancer
- The Silent Threat: How Gas Stoves Pollute Our Homes and Impact Health
- Paternal Microbiome Perturbations Impact Offspring Fitness
- New Report Casts Doubt on Maradona’s Cause of Death and Rocks Manslaughter Case
- Chinese academician unable to provide the exact source of liver transplants
- Early Biomarker for Multiple Sclerosis Development Identified Years in Advance
mRNA Vaccine Targeting Bacteria to Prevent Lyme Disease from Tick Bites
- AstraZeneca Admits for the First Time that its COVID Vaccine Has Blood Clot Side Effects
- Was COVID virus leaked from the Chinese WIV lab?
- HIV Cure Research: New Study Links Viral DNA Levels to Spontaneous Control
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
mRNA Breaks New Ground: UPenn Team Develops mRNA Vaccine Targeting Bacteria to Prevent Lyme Disease from Tick Bites.
Lyme disease, a naturally occurring infectious disease caused by Borrelia burgdorferi bacteria, is typically transmitted to humans through the bites of black-legged ticks (I. scapularis). It can lead to symptoms such as fever, headaches, fatigue, and skin rashes. If left untreated, the infection can spread to the joints, heart, and nervous system.
While most Lyme disease patients can be successfully treated with antibiotics for several weeks, some develop Post-Treatment Lyme Disease Syndrome (PTLDS), which can result in severe joint pain and long-term cognitive issues. Currently, there are no approved Lyme disease vaccines for regular human use.
The rapid approval of mRNA vaccines during the COVID-19 pandemic highlighted the powerful potential of mRNA vaccines. Consequently, researchers began exploring the preventive potential of mRNA vaccines against tick-borne diseases.
Recently, the team led by Norbert Pardi at the University of Pennsylvania, in collaboration with mRNA vaccine pioneer Professor Drew Weissman, published a research paper titled “Development of an mRNA-lipid nanoparticle vaccine against Lyme disease” in the Molecular Therapy journal.
This study utilized a lipid nanoparticle-encased modified mRNA (mRNA-LNP) platform to develop an mRNA vaccine targeting Lyme disease. The vaccine employed mRNA encoding the antigen OspA from Borrelia burgdorferi, the pathogen responsible for Lyme disease, and demonstrated robust humoral and cellular immune responses in mice after a single dose.
These potent immune responses resulted in protection against Borrelia burgdorferi infection, suggesting that highly effective mRNA vaccines can be developed not only against viruses but also against bacteria.

As early as November 2021, a team led by Erol Fikrig at the Yale University School of Medicine, in collaboration with mRNA vaccine pioneer Professor Drew Weissman, published a cover paper in Science Translational Medicine.
Their research developed an mRNA vaccine targeting black-legged ticks using lipid nanoparticles (LNPs) to deliver mRNA encoding 19 proteins (19ISP) found in the saliva of these ticks. This approach triggers a rapid response in the skin upon tick bites, limiting the time ticks can feed on blood and infect hosts, while also allowing hosts to detect tick bites quickly and prevent the transmission of Lyme disease by black-legged ticks.
It’s important to note that this mRNA vaccine targets the black-legged ticks themselves rather than specific pathogens transmitted by them. Tick bites are often painless because ticks secrete substances that numb the skin. The mRNA vaccine developed in this study helps the immune system recognize ticks, preventing Lyme disease infection. Additionally, the vaccine causes rapid redness and itching at the site of tick bites, making them as noticeable as mosquito bites and aiding in the removal of ticks before they can transmit pathogens.

In this latest study published in the Molecular Therapy journal, the team at the University of Pennsylvania, led by Norbert Pardi, along with Professors Drew Weissman and Erol Fikrig, developed an mRNA vaccine directly targeting Borrelia burgdorferi, the pathogen responsible for Lyme disease.
Compared to viruses, Borrelia burgdorferi is a more complex bacterium, making the development of effective vaccines against them more challenging. The team identified a protein in Borrelia burgdorferi, Outer Surface Protein A (OspA), which elicits a robust immune response and is a conserved protein across multiple strains of the bacterium, making it an ideal target for preventing Borrelia burgdorferi infection and subsequent development into Lyme disease.
The researchers used the mRNA-LNP vaccine technology already validated in COVID-19 vaccines to deliver mRNA encoding OspA. Preclinical animal model experiments showed that the mRNA-LNP vaccine targeting OspA induced strong antigen-specific antibodies and T-cell responses after a single dose, preventing Borrelia burgdorferi infection. Importantly, the vaccine triggered a robust memory B-cell response that could be activated long after vaccination, helping to prevent future Borrelia burgdorferi infections.
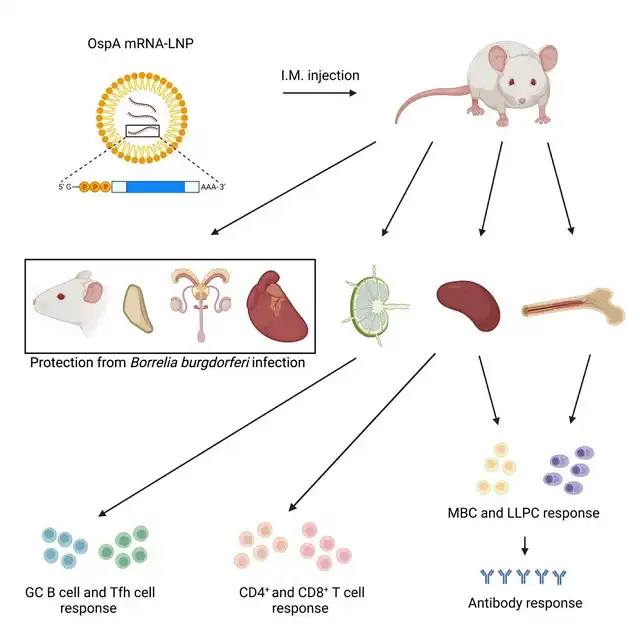
The research team stated that the experimental mRNA vaccine developed in this study provided effective protection in preclinical animal models against Borrelia burgdorferi infection.
This study suggests that mRNA vaccines could be a powerful tool in preventing Lyme disease, potentially reducing the number of Lyme disease cases and Post-Treatment Lyme Disease Syndrome (PTLDS).
mRNA Vaccine Targeting Bacteria to Prevent Lyme Disease from Tick Bites
Paper Links:
https://doi.org/10.1016/j.ymthe.2023.07.022
https://www.science.org/doi/10.1126/scitranslmed.abj9827
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



