The Critical Role of Immune Cell Triumvirates in Enhancing CD8+ T Cell Function
- Major Breakthrough in Infertility Research: This Stem Cell Could Be Key to IVF and Other Fertility Treatments
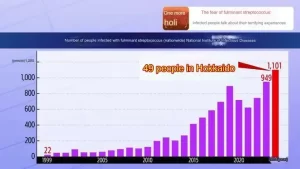
- Flesh-Eating Bacteria Infection Over 1000 Cases in Japan!
- CDC Recommends Updated COVID-19 Vaccines for 2024-2025 Season
- Will China and India produce cheaper “Miracle Weight Loss Drug”Semaglutide soon?
- Keto Diet Accelerates Aging and Promotes Cancer Metastasis
- The Critical Role of Immune Cell Triumvirates in Enhancing CD8+ T Cell Function
- Chinese-made Drug Enters Australia: Causing at Least 20 Deaths!
- How serious is Japan’s “flesh-eating bacteria” problem?
- Taiwan 6th wave of COVID outbreak: 623 confirmed cases in one week and 38 deaths
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
The Critical Role of Immune Cell Triumvirates in Enhancing CD8+ T Cell Function
A research team led by Andrea Schietinger at the Memorial Sloan Kettering Cancer Center (MSKCC) has published a groundbreaking study in the journal Cancer Cell. This research highlights the critical importance of the unique spatial positioning and interactions of immune cells within tumors, rather than just their quantity, as key determinants of the effectiveness of immunotherapy for solid tumors.
The team discovered that the effective activation and cytotoxic function of tumor-specific CD8+ T cells depend on their interaction with CD4+ T cells and antigen-presenting cells, forming a “triumvirate”. This combination leads to the reprogramming of CD8+ T cells within tumors, even reversing their dysfunction, allowing them to exert cytotoxic effects and eliminate tumors. Each component of this trio has a distinct role: antigen presentation, functional activation, and cell killing.
The combination of CD8+ T cells, CD4+ T cells, and antigen-presenting cells creates a powerful synergy. The tumor cells are overwhelmed by this cooperative attack, likened to a coordinated friendship charge.

Screenshot of the paper’s first page
In recent years, immunotherapy, which activates the patient’s own immune system to attack and eliminate tumor cells, has made significant progress in cancer treatment.
Despite the enhancement of CD8+ T cells to varying degrees, many tumors still evade immune surveillance and immunotherapy. This phenomenon is mainly due to the functional impairment of CD8+ T cells within the tumor. To address this challenge, Andrea Schietinger’s team focused on the potential role of CD4+ T cells in regulating and restoring the function of tumor-infiltrating CD8+ T cells.
Using a melanoma mouse model undergoing adoptive T cell therapy, they observed that, similar to clinical treatments for solid tumors, injecting only high-functioning, tumor-specific CD8+ T cells still allowed tumor progression.
However, when both tumor-specific CD4+ and CD8+ T cells were injected, the tumors were completely eradicated in mice, with a 100% long-term tumor-free survival rate.
Further transcriptomic and epigenetic analyses confirmed that CD4+ T cells could indeed help CD8+ T cells overcome their dysfunction. Through targeted depletion of specific cells and constructing tumor models with different antigens, researchers found that the synergistic effect of these two T cells requires the participation of another key member—the dendritic cell. This revealed the task distribution within the “Iron Triangle.”
CD8+ T cells are the main players in killing tumor cells but are prone to dysfunction.
Fortunately, CD4+ T cells are there to help. Researchers found that CD4+ T cells can prevent the terminal differentiation of CD8+ T cells by inducing reprogramming within the tumor, causing CD8+ T cells to express genes associated with a precursor T cell state while reducing the expression related to dysfunction and exhaustion, effectively combating the tumor.
During adoptive T cell therapy in mice, co-injection of tumor-specific CD4+ T cells could prevent CD8+ T cell dysfunction. If CD4+ T cells were injected after CD8+ T cell dysfunction, it effectively reversed the dysfunction, producing more effector molecules like IFN-γ and TNF-α, ultimately achieving tumor eradication.
However, CD4+ T cells don’t seek out CD8+ T cells for collaboration on their own; dendritic cells must bridge the gap. Specifically, dendritic cells, as antigen-presenting cells, capture and process tumor antigens and present them on their surface via MHC I and MHC II molecules. This allows dendritic cells to present tumor antigens to both CD8+ and CD4+ T cells and activate them, forming a triad that physically brings these two T cells together, driving CD4+ T cell regulation of CD8+ T cells.
How powerful is this combination?
Researchers found that when mice received adoptive T cell therapy or immune checkpoint blockade (ICB) treatment, tumors with similar numbers of infiltrating immune cells but lacking the triad structure could not completely inhibit growth. Tumors forming numerous triads, however, eventually regressed. (Clearly, mechanisms matter as much as the numbers.)
Finally, researchers evaluated the spatial positioning of immune cells in malignant pleural mesothelioma (MPM) patients before and after ICB treatment, checking whether CD8+ T cells, CD4+ T cells, and dendritic cells formed dual or triad structures.
The results showed that, unlike the dual structure (CD4+ T cells and dendritic cells), the presence of triads clearly distinguished responders from non-responders. CD8+ T cells in the triads expressed higher levels of IFN-γ and granzyme B, and tumors with the triad structure had significantly fewer immunosuppressive regulatory T cells (Tregs).
In other words, simply increasing the number of immune cells isn’t enough. The spatial structure is also a critical factor determining the success of immunotherapy!
In summary, this study reveals a previously overlooked phenomenon: within tumors, the unique interactions and spatial relationships between tumor-specific CD4+ T cells and CD8+ T cells can significantly enhance the ability of CD8+ T cells to eliminate solid tumors. Future research will explore the specific mechanisms of reprogramming and how clinical interventions can be developed to promote and maintain the formation of immune cell triads, enhancing the effectiveness of ICB therapy, adoptive T cell therapy, and cancer vaccines.
Additionally, consistent with these findings, a paper published in *Nature Medicine* last year showed that in early hepatocellular carcinoma patients treated with PD-1 inhibitors, the formation of triad structures of dendritic cells, CD4+ T cells, and CD8+ T cells in the tumor microenvironment helped CD8+ T cells differentiate towards higher anti-tumor activity, strengthening therapeutic effects.
The Critical Role of Immune Cell Triumvirates in Enhancing CD8+ T Cell Function
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.