Revolutionary Non-Opioid Analgesic Unveiled in Phase III Clinical Trials
- Global First: Bovine Avian Influenza Transmission to Humans Detected
- Potassium-Enriched Salt Substitutes: Reducing Blood Pressure Risk?
- Tritium Detection in Fukushima’s Seawater: Below Safety Standards
- What Reason Let AstraZeneca Withdraws COVID Vaccine From The Market?
- How Many Times Can a Cancer Patient Undergo Chemotherapy?
- What are the WHO recommendations for Japanese encephalitis vaccines?
Breaking Medical News: Revolutionary Non-Opioid Analgesic Unveiled in Phase III Clinical Trials
- AstraZeneca Admits for the First Time that its COVID Vaccine Has Blood Clot Side Effects
- Was COVID virus leaked from the Chinese WIV lab?
- HIV Cure Research: New Study Links Viral DNA Levels to Spontaneous Control
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Breaking Medical News: Revolutionary Non-Opioid Analgesic Unveiled in Phase III Clinical Trials
On January 30, 2024, Vertex made a groundbreaking announcement on its official website, revealing positive outcomes in the Phase III study of its selective NaV1.8 inhibitor, VX-548, in the treatment of moderate to severe acute pain.

Vertex Official Website Announcement
Interpretation by researchers from the National Institutes of Health in the United States:
In a significant development in the field of medicine, Vertex has introduced a novel non-opioid analgesic, the sodium ion channel NaV1.8 inhibitor: VX-548. This drug exhibited outstanding analgesic effects in Phase III randomized controlled trials (RCT). The birth of this new medication holds immense significance as it represents a non-addictive and highly effective analgesic, potentially heralding a revolution in pain medicine.
The clinical trial included 1,118 patients undergoing abdominal surgeries and 1,073 patients undergoing thumb arthritis surgeries, randomly assigned to different groups. The study found that VX-548 demonstrated excellent safety and patient tolerance.
Results indicated that VX-548 was more rapid and effective in relieving pain compared to the placebo. The primary endpoint of the study was SPID48, a weighted pain score (NPRS) of patients in the 48 hours post-surgery. VX-548 significantly improved post-surgical pain in both abdominal surgery by 48.4 (P<0.0001) and thumb arthritis surgery by 29.3 (P=0.0002) compared to the control group.
VX-548 is anticipated to be among the most awaited clinical trial results in the field of biomedicine in 2024. Vertex plans to apply for Breakthrough Therapy and Fast Track Designations for pain treatment from the FDA in the middle of this year.
Pain medicine and analgesic treatments play a crucial role in clinical disciplines, and the introduction of a non-addictive and efficient analgesic is poised to revolutionize this field, making pain control more accessible for patients and holding significant implications.
In modern drug development, each groundbreaking drug’s emergence stems from a meticulous understanding of a biological/physiological mechanism. Similar to PD-1 inhibitors and GLP-1R agonists, VX-548 reaffirms this point—NaV1.8 is the voltage-gated sodium channel (VGSC) discussed in university physiology textbooks; John Wood reported in 1996 in Nature that blocking this channel has analgesic effects, marking it as Nobel-worthy work.
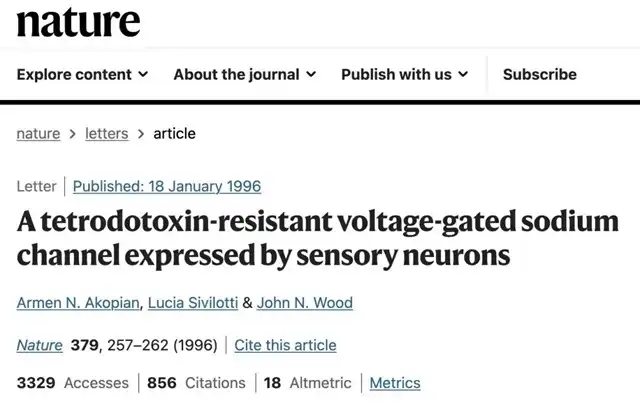
John Wood reported in Nature in 1996 that blocking NaV1.8 has an analgesic effect (Source: Nature )
Breaking Medical News: Revolutionary Non-Opioid Analgesic Unveiled in Phase III Clinical Trials
(source:internetI8nu8tIF965AspFTh8UEmg, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.