COVID-19 Vaccine: Mass Production in China Soon
COVID-19 Vaccine: Mass Production in China Soon
COVID-19 Vaccine: Mass Production in China Soon. The COVID-19 epidemic has caused nearly 1.49 million deaths worldwide and 64.3 million confirmed cases. The epidemic is still ongoing, and vaccines have become a key word. Sun Chunlan, Vice Premier of the State Council: Get ready for mass production! Some people used it urgently this year.
On December 2, the United Kingdom approved the use of the COVID-19 vaccine from Pfizer and BioNTech. The vaccine will be available throughout the UK starting next week.
In the United States, according to the latest documents obtained by CNN, Pfizer’s first batch of COVID-19 vaccines will be delivered on December 15th, and then Moderna’s first batch of vaccines will also be delivered on December 22nd. The document also estimates that Pfizer will produce 22.5 million doses of vaccine and 18 million doses of Moderna vaccine this month.
When the United Kingdom and the United States successively approved vaccine delivery, how did China’s local vaccine progress?
According to Xinhua News Agency, Vice Premier Sun Chunlan and State Councilor Wang Yong were in Beijing on the 2nd to investigate the research and development and production preparations of the COVID-19 virus vaccine. Understand the new coronavirus vaccine scientific research, production workshop construction, preparation for batch issuance, etc., and fully affirm the achievements of the vaccine work.
Sun Chunlan pointed out that since the outbreak of the COVID-19 pneumonia, the State Council’s joint prevention and control mechanism research team for vaccine research and development and relevant parties have thoroughly implemented the spirit of General Secretary Xi Jinping’s important instructions, implemented the decisions and deployments of the Party Central Committee and the State Council, and promoted vaccines in an urgent wartime state. In all aspects of work, scientific researchers are racing against time and struggling to tackle key problems. Departments and localities take the initiative to serve the front line. Manufacturers take the overall situation into consideration and dare to take responsibility to promote important progress in vaccine work. At present, 14 vaccines under 5 technical routes have entered clinical trials, of which 5 vaccines are undergoing phase III clinical trials in accordance with the procedures and standards, and the emergency use and production preparations are proceeding in an orderly manner.
Sun Chunlan pointed out that in the next step, we must continue to advance the vaccine phase III clinical trials scientifically and rigorously, and conduct review and approval in strict accordance with laws, regulations and internationally recognized technical standards to ensure that the vaccine is safe, effective, and can withstand all aspects of inspection. We must be prepared for large-scale production, in strict accordance with laws, procedures and requirements such as quality supervision and biosafety, improve the traceability system of the entire vaccine process, severely punish violations of laws and regulations in accordance with the law, and create a good market environment.
It is necessary to study and formulate the vaccination distribution plan after the vaccine is on the market, clarify the population range, vaccination sequence and time considerations, and organize the training of personnel of disease control agencies and grassroots vaccination units in advance. According to the needs of winter epidemic prevention and control, emergency use of high-risk groups such as port-related employees and front-line supervisors will be completed this year. Vaccines are related to people’s life safety. All relevant departments must strictly enforce management systems for vaccine research and development, production, circulation, and use, strengthen batch issuance and supervision capacity building, help R&D units and manufacturers solve practical problems, and carry out publicity and education of vaccine knowledge, The people have a scientific and rational understanding of vaccines, and actively carry out preventive vaccination and personal protection.
The five new coronavirus vaccines in China that Sun Chunlan called into Phase III clinical trials are:
- Inactivated vaccine from Wuhan Institute of Biology, a subsidiary of Sinopharm Group;
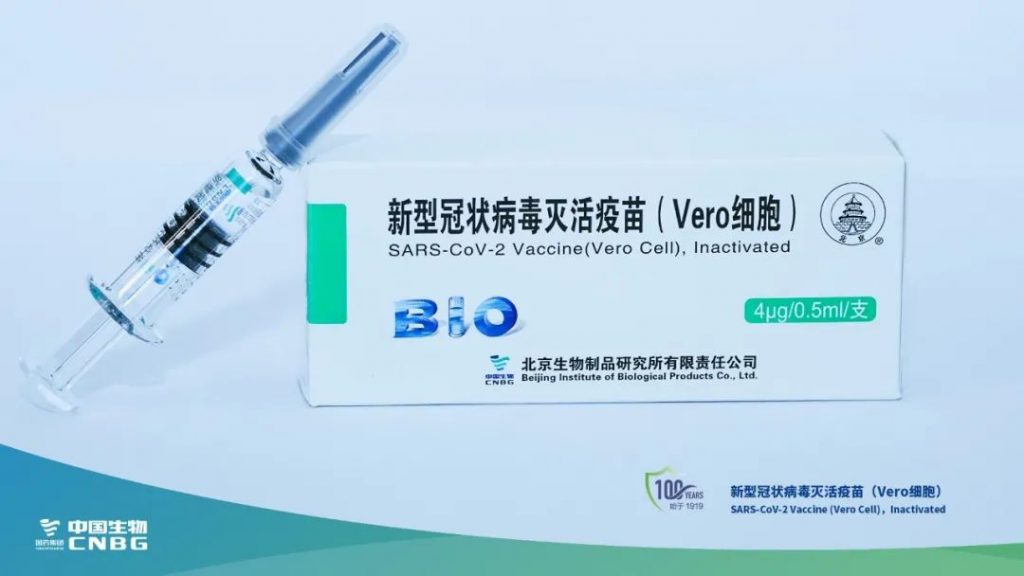
- Inactivated vaccine from Beijing Institute of Biology, a subsidiary of Sinopharm Group;
- The adenovirus vector vaccine developed by the Academy of Military Sciences in conjunction with Cansino;
- Recombinant protein vaccine of Anhui Zhifeilongkoma Biopharmaceutical Co., Ltd.;
- Inactivated vaccine from Beijing Kexing Zhongwei Biotechnology Co., Ltd.
How is China’s vaccine progress?
SINOPHARM
On November 25, Shi Shengyi, deputy general manager of Sinopharm Group, stated that Sinopharm Group had submitted a COVID-19 vaccine application to the State Food and Drug Administration. Once the news was released, Sinopharm hit its daily limit at the close.
However, the Sinopharm Group has not yet released relevant announcements on this matter.
Previously, on November 6, the 3rd Hongqiao International Health Technology Innovation Forum was held in the 3rd CIIE Pavilion. At the forum, Liu Jingzhen, Chairman of Sinopharm Group, said when talking about the topic of COVID-19 vaccine, there are now dozens of Ten thousand people were vaccinated urgently with two inactivated COVID-19 vaccines under the Sinopharm Group. There was no serious adverse reaction. The number of people who left the country after vaccination reached 56,000 and none of them have been infected.
According to Liu Jingzhen’s statement, the above-mentioned personnel leaving Beijing involved China Petroleum, Petrochemical, Electronic Technology, International Group and other units. Liu Jingzhen also shared a case of a company’s Mexico office. According to him, a company has offices in more than 180 countries and regions around the world. There are 99 people in the Mexico office who work in one room, and 81 of them have received the COVID-19 vaccine. , 18 were not vaccinated. After the office outbreak, 10 people were infected, all from 18 unvaccinated people.
Liu Jingzhen said that whether it is from scientific research test data or use around the world, the effectiveness of the COVID-19 vaccine has been proved.
Regarding the future production capacity of vaccines, Liu Jingzhen said that by the end of this year, the production capacity of Sinopharm’s COVID-19 vaccine will probably reach about 100 million doses. According to national requirements, the production capacity will exceed 1 billion doses next year. “It can be used by the Chinese people in particular next year.”
CanSino Bio
On November 7, CanSino Bio issued an announcement stating that the international phase III clinical trial of the recombinant new coronavirus vaccine (adenovirus vector) jointly developed by the company and the Academy of Military Sciences has been launched in Mexico and has already been vaccinated. Earlier, CanSino Bio announced that it will provide Mexico with 35 million doses of the COVID-19 vaccine from the end of 2020 to 2021.
It is reported that the cooperative vaccine was developed by the team of Academician Chen Wei of the Academy of Military Medicine of the Academy of Military Sciences. It is the first vaccine in the world to enter the clinical phase I. As early as June 25, it obtained the military special-needed drug approval issued by the Central Military Commission Health Bureau ( Valid for 1 year).
On November 28, at the 2020 First China Health Technology Innovation and Development Conference, Yin Weidong, chairman of Kexing Holding Biotechnology Co., Ltd., said that the COVID-19 vaccine developed by Beijing Kexing Zhongwei Biotechnology Co., Ltd. was launched in Brazil. The phase III clinical study is expected to complete the interim analysis in early December.
The inactivated vaccine developed by Kexing Zhongwei is currently undergoing Phase III clinical trials in Brazil, Indonesia and other countries. Previously, media reported that the vaccine was decided to stop testing on November 10 by the Brazilian health supervision agency, the National Health Supervision Agency. Since then, the company issued a statement that it has been re-authorized to resume clinical trials of the COVID-19 vaccine.
Kexing
The vaccine was also approved for emergency use in China’s COVID-19 vaccine on July 22. In mid-October, Zhejiang initiated emergency vaccination of the COVID-19 vaccine, which was used. Judging from the official WeChat content of the Jiaxing City Center for Disease Control and Prevention in Zhejiang Province, the COVID-19 vaccine for emergency vaccination in this area will be ordered by the provincial department from Beijing Kexing Biologics. The price of the vaccine is 200 yuan/bottle (bottle) and 2 doses. A total of 400 yuan.
Kexing Zhongwei stated that the production of inactivated COVID-19 vaccines was fully launched in August. According to the previous production results, the production capacity is about 300 million doses per year. By the end of this year, about 100 million doses of inactivated COVID-19 vaccines (including Semi-finished products).
Zhifei
The COVID-19 vaccine developed by Anhui Zhifei Longkoma Biopharmaceutical Co., Ltd., a subsidiary of Zhifei Biology, is jointly developed by the company and the Institute of Microbiology of the Chinese Academy of Sciences. It uses a technical route similar to the hepatitis B vaccine, hepatitis E vaccine, and cervical cancer vaccine. Subunit vaccine.
The vaccine started a phase III clinical trial in China in Xiangtan County, Hunan Province on November 18. Foreign countries will start a phase III clinical trial in Uzbekistan in the near future. It is reported that this vaccine has been shipped to Uzbekistan, and several Chinese experts have also been in place. They will be responsible for monitoring volunteers and training Uzbekistan doctors. In addition, Indonesia, Pakistan, and Ecuador will also start phase III clinical trials.
It is understood that the company has obtained the drug production license change in September and added the reorganization of the new coronavirus vaccine. The new coronavirus vaccine workshop was put into use that month, and the trial production was smooth, with an annual output of more than 300 million doses. As for the price, no news has been revealed.
What is the price and supply of the vaccine?
According to the Securities Times, at the first China Health Technology Innovation and Development Conference held on November 27, Zhong Nanshan, an academician of the Chinese Academy of Engineering, said that the three-month clinical trial results of Pfizer and Moderna were far from enough. Vaccine protection rate is not the only indicator. (Note: The Phase III clinical trial of Pfizer vaccine began on July 27, and on November 18, Pfizer and BioNTech announced the end of the Phase III clinical trial of their COVID-19 vaccine candidate)
In response to the most concerned vaccines, Zhong Nanshan pointed out that natural immunization is unrealistic, unscientific and inhumane. Among the 14 new coronavirus vaccines currently in clinical trials, China’s vaccines are moving forward steadily.
“We will not lag behind the foreign vaccines that have already announced some Phase III results. Currently, the results published by Moderna and Pfizer have data on effective and protective efficiency of more than 90%, but this data is only one of the indicators. Good vaccine’, we have to consider other factors.”
In addition, the public is also concerned about how the vaccine is priced. According to the National Health Commission, the online rumors are all false reports, and most of them are taken out of context. The pricing of the COVID-19 vaccine is not based on supply and demand, but on cost. The pricing of the COVID-19 vaccine must be within the range of public acceptance.
On October 20th, Zheng Zhongwei, director of the Medical Science and Technology Development Research Center of the Chinese Health Commission, said that by the end of this year, the annual production capacity of China’s COVID vaccine will reach 610 million doses. Next year, the annual production capacity of China’s COVID-19 vaccine will be effectively expanded on this basis. , To effectively ensure the demand for China’s COVID-19 vaccine in China and other countries around the world.
(source:xinlang)



