Medicine Development: Targeting the GPCR receptor complex
Medicine Development: Targeting the GPCR receptor complex
Medicine Development: Targeting the GPCR receptor complex. G protein-coupled receptors (GPCRs) are the most important drug targets and are the site of action for nearly 40% of approved drugs. GPCR is a complete membrane protein, once it binds to extracellular ligands, it triggers an intracellular signal cascade. Traditionally, the focus of drug discovery has been to promote or prevent receptor activation by directly targeting receptor proteins. However, there is a new dimension in drug discovery, because many receptor proteins exist in complexes with receptor-related proteins.
The functional properties, signaling pathways, and localization of these receptor complexes are usually different from isolated receptor proteins. Receptor complexes provide an opportunity to target and regulate specific signaling pathways, which cannot be achieved by traditional receptor protein targeting alone. In addition, due to the differential expression of receptor-related proteins, multi-protein receptor complexes are usually limited to specific tissues or cell types, which also provides opportunities for regulating specific receptor functions. This allows us to make more precise regulation of the disease mechanism, and it is possible to develop more effective drugs with fewer side effects.
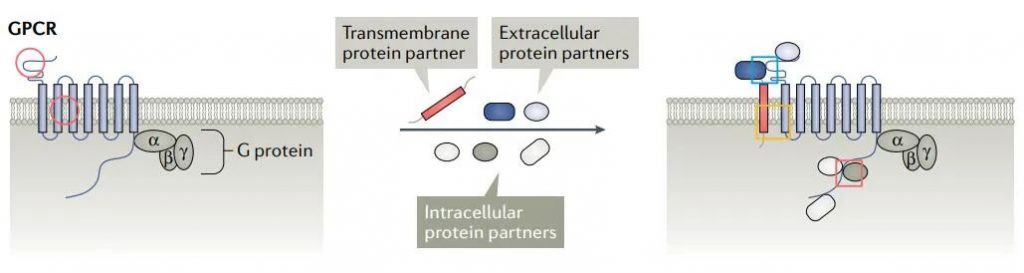
The composition of the receptor complex
GPCRs are membrane proteins that cross the membrane seven times. The intracellular G protein is the key to function. Post-translational modifications, including phosphorylation and palmitoylation, also have a profound impact on receptor availability and signal transmission. We need to realize that these receptors exist as complexes rather than isolated entities. In addition, phenotypic differences due to mutations in genes encoding proteins also affect the polymorphism of GPCRs.
The receptor complex is composed of a core receptor protein and one or more receptor-related proteins combined with receptor subunit proteins. Receptor-related proteins directly bind to receptor proteins and usually have a significant impact on the structure, function and/or localization of the receptor complex. In addition, the receptor complexes are not uniform, and their characteristics depend on the cell type, tissue location, developmental stage, and disease state. Traditional drug discovery is to directly target receptor proteins. Receptor complexes mediated by multiple protein interactions outside of cells, cells and cell membranes provide ample opportunities for the development of new drugs.
GPCR receptor complex
The first example of GPCR receptor complex biology is the regulation of MCR accessory proteins (MRAPs) on the melanocortin receptor (MCR) family: MRAP is important for the cell surface expression of MCR, but it also affects signal transduction and Internalization. MCR is involved in a variety of physiological functions, such as skin pigmentation and energy balance. Functional MCRs can only be expressed in adrenal cells, which indicates the presence of adrenal MRAP. In addition, mutations in genes encoding MCRs cannot fully explain certain familial glucocorticoid deficiencies, which indicates the existence of another related protein, which was later considered MRAP. MRAP2, a brain-specific homologue, was found to play an important role in energy balance, and its association with MCR reduces the obesity phenotype.
In GPCRs, another example of complexity is the GABAB receptor, because it contains two 7 transmembrane domain proteins: one is GABA-bound GABA
B1 subunit, one is the G protein binding GABAB2 subunit. In addition, it forms stable complexes with members of the K+ channel tetramerization domain (KCTD) protein family and several other proteins that regulate receptor signaling and localization. This provides many opportunities for fine-tuning the GABAB receptor complex, not just by directly targeting the core GABAB receptor subunits.
Strategies for targeting receptor complexes
Historically, many drugs targeting receptor complexes have been discovered in phenotypic screening. Recently, high-throughput screening (HTS) has been used for target drug development to determine the small molecule regulation of such complexes Agent. In addition, the lack of structural information of the receptor complex hinders the rational design of drugs and structure-based drug design, and recent advances, especially cryo-electron microscopy, have promoted our understanding of the structure of protein complexes, which enables the identification of GPCR monomers. The allosteric binding sites of homodimers and homodimers and heterodimers, and the development of small molecules that modulate subtype-specific receptor complexes.
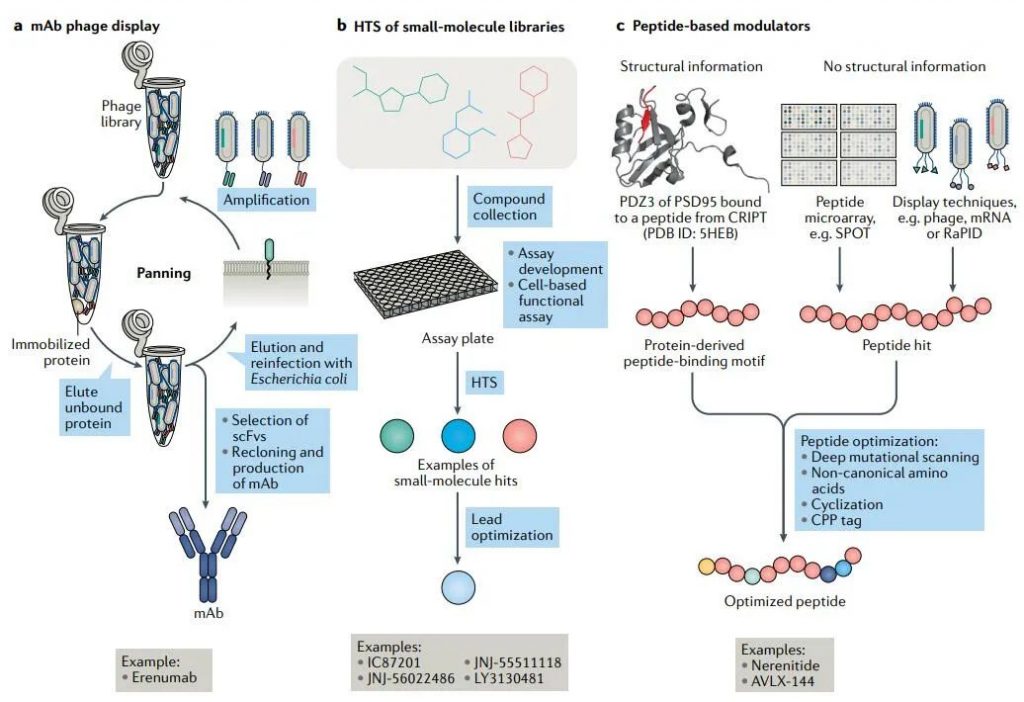
Antibodies: The main advantages of antibodies include high affinity and selectivity, as well as a long half-life. The recent approval of the monoclonal antibody erenumab is an example. The antibody targets a complex of calcitonin receptor-like receptor (CALCRL) and receptor activity modifying protein 1 (RAMP1) for the preventive treatment of migraine. Antibodies can also target a specific conformation of the receptor complex, allowing for stabilization of the active or inactive state of the receptor complex. Antibody phage display is an in vitro selection technology widely used in fully humanized monoclonal antibodies, which is based on the fusion of peptides and phage envelope proteins. Although it eliminates the need for immunity, antibody phage display does require suitable antigens, which may be the biggest challenge for membrane protein complexes.
Small molecules: Small molecules usually require the availability of high-quality, diverse structure libraries. In addition, the development of a well-designed and validated screening analysis is a prerequisite for any HTS activity. GPCR screening analysis usually includes cell-based functional analysis, rather than simple binding analysis. HTS challenges may include low hit rates, high false positive rates, and poor screening quality. Nevertheless, HTS and subsequent medicinal chemistry studies have provided a series of small molecule targeted receptor complexes, such as compounds targeting the AMPAR–TARPγ8 complex, which are currently under development.
Peptides: In cases where the regulation of receptor complexes requires complete replacement of two related proteins, small molecules may not be suitable. On the contrary, peptides can be a good starting point. However, the poor pharmacokinetic properties of peptides have brought great challenges, which must be resolved in subsequent optimization work. Several peptide display technologies, such as phage display, ribosome display, or mRNA display, can be used to identify and optimize peptide inhibitors.
Another major challenge of peptides is the lack of cell permeability. Various research strategies have solved this problem, including macrocyclization and linking cell-penetrating peptides. At present, the development of cell-penetrating peptides with excellent performance is an active area of research.
Drugs targeting the GPCR complex
CALCRL–RAMP1: Regulating calcitonin-related peptide (CGRP) receptor complex for the treatment of migraine is a significant example of the pharmacological targeting of GPCR complexes. The central component of the CGRP receptor complex is CALCRL, a type B GPCR involved in hormone homeostasis. In order to form a functional receptor, CALCRL must be associated with RAMP1, which transports CALCRL to the cell surface, where RAMP1 is still associated with CALCRL to form a functional CGRP receptor complex.
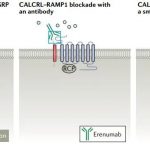
Erenumab was developed by Amgen for migraine prevention. It is a human monoclonal antibody targeting the CGRP receptor complex. Erenumab benefits from the ability of antibodies to span the large binding sites conferred by CALCRL and RAMP1, and effectively blocks the binding of CGRP, thereby activating the receptor. Due to its high affinity and specificity for the CGRP receptor complex over CALCRL–RAMP2/RAMP3 complex (5000 times), plus the prolonged serum half-life of the antibody, erenumab is very effective in the prevention of intermittent and chronic migraine. In addition to the first CGRP targeted therapy, erenumab is also the first FDA approved antibody against GPCR.
Several small molecule antagonists of CGRP receptor complexes have shown promising results in preclinical and clinical studies. Although their chemical structures are not related, these molecules are collectively referred to as “gepants.” Gepants share the same mode of operation: they use the CALCRL–RAMP1 interface to prevent CGRP binding. Some gepants, including olcegepant and telcagepant, alleviated migraine symptoms without cardiovascular side effects, but some gepant clinical programs were suspended due to liver toxicity. However, other programs are still continuing, including Allergan’s ubrogepant and atogepant. These two drugs have no hepatotoxicity and still have a strong effect on the treatment of acute migraine. Ubrogepant was approved by the FDA in December 2019, and rimegepant developed by Biohaven Pharmaceuticals was approved by the FDA for the treatment of acute migraine in February 2020.
GBRs and KCTD proteins: GBR is a heterodimeric class C GPCR composed of one GB1a subunit or one GB1b subunit and one GB2 subunit. KCTD proteins (KCTD8, KCTD12, KCTD12B and KCTD16) are cytoplasmic proteins that bind to the C-terminal tail of the GB2 subunit and the Gβ subunit of the G protein. KCTD12 and KCTD12B induce rapid desensitization of channel gating by isolating Gβγ, thereby uncoupling effector Ca2+ and K+ channels.
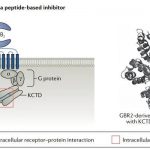
GABA, the main inhibitory neurotransmitter in the brain, regulates synaptic transmission by activating presynaptic and postsynaptic G protein-coupled GABAB receptors (GBRs). GBRs are expressed in the central nervous system, and their dysfunction is related to neurological and neuropsychiatric diseases, including epilepsy, pain, convulsions, autism spectrum disorders, depression, schizophrenia, anxiety and addiction. Currently, there are only two drugs targeting GBRs on the market-baclofen and gamma-hydroxybutyric acid.
Outlook
Receptor complexes present challenges and exciting new opportunities for drug discovery. We are in the initial stages of rationally targeting receptor complexes to identify more effective, selective and safer drugs. Targeted receptor complexes need to reconsider screening strategies, especially when heterologously expressed receptors often lack key receptor-related proteins. Along these lines of thought, it is important to reveal the complete composition of related proteins in the natural receptor. Only with this understanding can we develop analytical methods that faithfully reproduce biological targets, and then develop more effective drugs with fewer side effects.
(source:chinanet)
Disclaimer of medicaltrend.org



