Immunotherapy to treat tumors
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Immunotherapy to treat tumors
Immunotherapy to treat tumors. In the annual ranking of the top ten scientific breakthroughs by Science magazine in 2013, immunotherapy ranked first. In recent years, immunotherapy has always been a hot spot in the field of tumor treatment research at home and abroad, which can be called a revolutionary breakthrough.
In the annual ranking of the top ten scientific breakthroughs by Science magazine in 2013, immunotherapy ranked first. In recent years, immunotherapy has always been a hot spot in the field of tumor treatment research at home and abroad, which can be called a revolutionary breakthrough.
Cancer immunotherapy can make the patient’s own immune system fight cancer. Each type of cancer is different, and each patient is unique. How can cancer treatment be targeted at one person? Medical researchers have conducted a large number of clinical trials on this and found that adoptive cell therapy or cellular immunotherapy (using patient T cells) can be used to combat specific cancers.
Among them, adoptive cell therapy, also known as cellular immunotherapy, is a type of immunotherapy, by directly separating our own immune cells and simply expanding their numbers, or genetically modifying our immune cells (through gene therapy) To enhance its anti-cancer ability. The global medical community’s new research, new breakthroughs, and new understandings of adoptive cellular immunotherapy have gradually transformed its status from “non-mainstream therapy” to “adjuvant therapy to standard therapy”.
The following focuses on the four types of adoptive cell therapy that have made significant research progress in the world.
- Chimeric antigen receptor (CAR) T cell therapy
- Tumor infiltrating lymphocyte (TIL) treatment
- Natural Killer (NK) Cell Therapy
- Engineered T cell receptor (TCR) therapy
Chimeric antigen receptor (CAR) T cell therapy
Speaking of the hottest cancer therapy in recent years, CAR-T in immunotherapy is definitely one of the focuses.
Especially in terms of hematological tumors, the FDA successively approved the only two CAR-T cell therapy commercial products in the world in 2017: Novartis’ Kymriah (lymphoma) and Gilead’s Yescarta (leukemia), which has completely changed The landscape of cancer treatment.
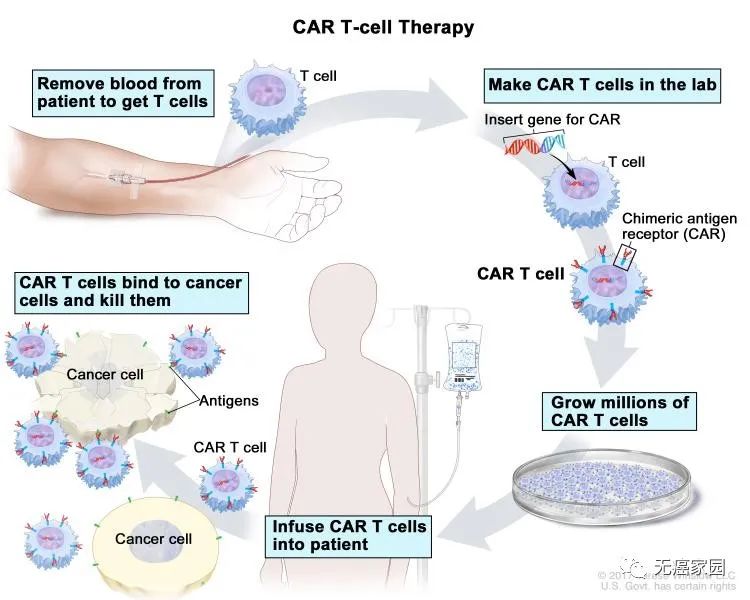
However, research on CAR-T treatment of solid tumors is very limited. In response to this urgent problem, medical researchers at home and abroad have made various modifications to CAR-T to treat a variety of solid tumors.
The effective rate of anti-cancer is 72%, CAR-T technology breaks the solid tumor barrier
At the 2019 American Association for Cancer Research (AACR) annual meeting, a new type of CAR-T cell technology targeting mesothelin has attracted the attention of everyone, and its efficacy data can be described as impressive. This CAR-T cell therapy called LcasM28z is dedicated to the research team of Prasad Adusumilli, deputy director of thoracic surgery at Memorial Sloan Kettering Cancer Center.
【Research Background】
This clinical trial included 21 patients with relapsed and refractory mesothelin-positive advanced solid tumors who had failed other treatments in the past, including 19 cases of advanced malignant mesothelioma, 1 case of advanced lung cancer, and 1 case of advanced breast cancer. . All patients received LcasM28z combined with PD-1 antibody treatment.
LcasM28z was developed by Adusumilli and his team using the second generation of activated CD28 all-human mesothelin CAR T cells with Icaspase-9 safety gene.Moreover, most patients have received high-dose chemotherapy before treatment with myeloablative pretreatment.It should be noted that this operation must be carried out in a very experienced top hospital and requires a high degree of infection prevention.
【Research results】
After 38 weeks of treatment, persistent CAR-T cells could still be detected in the peripheral blood of 13 of 21 patients. 11 patients successfully completed CAR-T injection and at least 3 courses of PD-1 treatment. After at least 3 months of follow-up, 8 of 11 patients had tumor shrinkage, and even 2 patients showed complete disappearance of tumor by PET-CT, and the overall remission rate was as high as 72%.
(source:chinanet, reference only)
Disclaimer of medicaltrend.org



