Coronary stent: Cobalt-chromium alloy or stainless steel?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Coronary stent: Cobalt-chromium alloy or stainless steel?
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- What is the difference between Atorvastatin and Rosuvastatin?
- How long can the patient live after heart stent surgery?
Coronary stent: Cobalt-chromium alloy or stainless steel? Coronary stent thin or thick? In the era of bare metal stents, the thickness of the stent beams is very important.

The European Journal of Intervention published a study that compared the latest generation of thin-layer cobalt-chromium alloy stents with the previous generation of stainless steel thick steel beam stents for a 1-year clinical follow-up comparison. The research is a forward-looking, multi-center, non-inferior design.
After 9 months of follow-up, quantitative coronary angiography (QCA) was used to determine the late intra-stent lumen loss (LLL), which was used as the primary endpoint, and the clinical evaluation was performed after 1 year of follow-up. In 8 centers, a total of 200 patients were randomly (1:1) receiving BF-CoCr (thin) or BF-SS (stainless steel) stent implantation.
The baseline clinical and pathological characteristics of the two groups of patients were similar, with an average age of 66 years, 23% women, and an average of 1.5 stents implanted per patient.
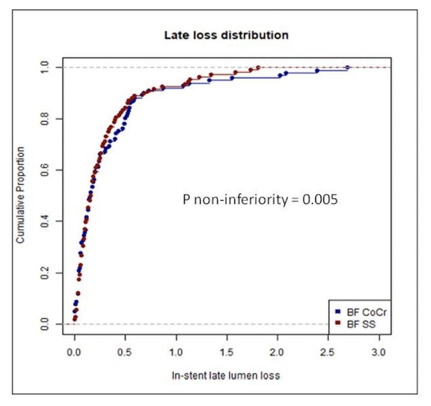
At the 9-month follow-up, the average in-stent LLL in the BF-CoCr group was 0.34±0.49mm, and the average in the BF-SS group was 0.29±0.37mm, and the non-inferiority P=0.005.
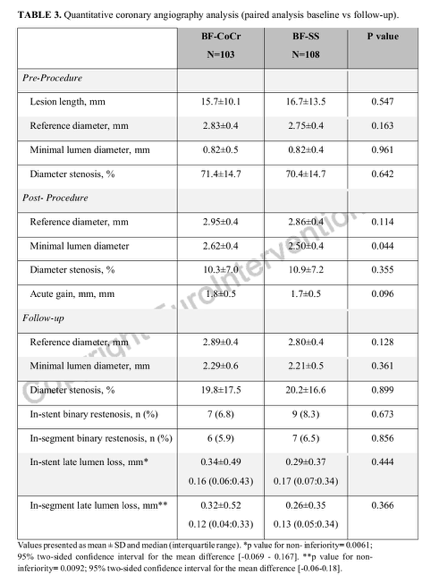
At 1 year, the target lesion failure rate was 7.3% in the BF-CoCr group and 9.3% in the BF-SS group, P=0.60.
In the era of bare metal stents, the thickness of the stent beams is very important. Kastrati et al. compared bare metal stents with different thicknesses (50 µm vs 140 µm) produced by the same manufacturer.
The restenosis rate of the angiographic review and the thinness of the LLL steel beam were better (15% vs 25.6%; p=0.003; 0.94±0.74) mm vs 1.17±0.78 mm, p=0.001).
In the DES era, the correlation between the thickness of steel beams and the incidence of restenosis is not too great.
Cypher stent steel beam thickness is 140um, LLL is close to 0mm; Taxus stent is 96um, LLL is about 0.4mm; ENDEAVOR stent is 91um, LLL is 0.6mm. However, if the stent beam is thick and rectangular, it may be related to restenosis and thrombosis.
Because of the low shear force caused by this structure, the blood recirculation area will be created, the concentration of locally activated platelets will increase, endothelialization will be delayed, and the production of natural anticoagulants will be reduced.
After the first-generation DES was applied to the clinic, the design of the stent has also undergone many innovations, such as thinner steel beams, polymer degradability, and the use of cobalt-chromium alloy materials instead of stainless steel.
The purpose is to speed up the coverage of the stent intima, reduce polymer stimulation, and shorten the treatment time of double antiplatelet.
With the adoption of new materials such as alloys, stent steel beams can be made thinner, which is also conducive to the delivery of stents under complex lesions, and the long-term benefits of reduced restenosis and mortality may also be related.
BF-CoCr is a new generation of DES made of ultra-thin cobalt-chromium alloy materials, with a steel beam of 84-88um; BF-SS is a DES based on a stainless steel platform with a steel beam of 120um, both of which are products of BioFreedom.
The two stents are not only different in materials, no polymer design, but also different in the types of anti-proliferative drugs, release kinetics, and concentration into the tissues.
The result of the study is that the new generation of stents is non-inferior. Isn’t that disappointing? We think its significance may be in the improvement of materials and the better delivery performance of the stent after ultra-thin steel beams, and it may also have advantages in shortening the dual antiplatelet therapy.
The main purpose of this study should be to evaluate the LLL of thin stents not inferior to thick stents.
In addition, the study also has some shortcomings, such as the small sample size and the lack of intracoronary imaging to evaluate the healing of the stent endothelium.
In addition, the follow-up time is not long enough. How safe is the new stent after stopping the dual antibodies?
There is also no question of whether the new stent doubles against platelets in one month.
Coronary stent: Cobalt-chromium alloy or stainless steel?
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



