Treatment of Acoustic Neuroma with Hydrocephalus
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Treatment of Acoustic Neuroma with Hydrocephalus
Treatment of Acoustic Neuroma with Hydrocephalus. Acoustic neuroma is a common cranial tumor mainly originating from Schwann cells of the vestibular nerve sheath, accounting for 80% to 90% of tumors in the cerebellopontine angle area.
Large acoustic neuroma compresses the surrounding brain tissue and causes secondary hydrocephalus, which ultimately endangers the patient’s life.
At present, there are no standardized guidelines for the treatment of acoustic neuroma with hydrocephalus.
The Department of Neurosurgery of the First Affiliated Hospital of Soochow University admitted 21 cases of acoustic neuroma with hydrocephalus from January 2013 to June 2019.
The clinical data were retrospectively analyzed and the diagnosis and treatment strategy of acoustic neuroma with hydrocephalus was discussed. The report is as follows.
1. Object and method
1.1 Clinical data
Inclusion criteria: head MRI diagnosed as acoustic neuroma with hydrocephalus. Exclusion criteria: patients with other neurological diseases, severe underlying diseases that cannot tolerate surgery or refuse surgery. A total of 21 cases were enrolled, including 7 males and 14 females; aged from 16 to 74 years old, with an average of 52.5 years old. The duration of the disease is 1d~10 years.
1.2 Diagnosis and classification of acoustic neuroma
All patients underwent cranial MRI before operation, and measured the largest diameter of the acoustic neuroma in the cerebellopontine cistern at the axial MRI through the internal auditory canal level. According to the Koos classification of acoustic neuroma:
- Grade 1 tumor is confined to the internal auditory canal;
- Grade 2 tumor invades the cerebellopontine angle and has a diameter of ≤20mm;
- Grade 3 tumor occupies the cerebellopontine angle cistern without brainstem displacement and has a diameter of ≤30mm; 4 Large-scale tumors are accompanied by brainstem displacement, and the diameter is more than 30mm.
The largest diameter of acoustic neuroma in this group is 30-67mm, with an average of 42.9mm; Koos grade 3 cases in 3 cases, 4 cases in 18 cases; solid acoustic neuromas in 3 cases and cystic acoustic neuromas in 18 cases.
1.3 Diagnosis and classification of hydrocephalus
Head CT or MRI indicates that the ratio of the maximum distance between the anterior horns of the two sides of the lateral ventricle to the largest cranial cavity (Evan’s index)> 0.3 is diagnosed as hydrocephalus; imaging examinations suggest that the fourth ventricle dilatation is diagnosed as communicating cerebral cerebrovascular disease Water, undilated fourth ventricle is diagnosed as non-communicating hydrocephalus. In this group, there were 19 cases of non-communicating hydrocephalus and 2 cases of communicating hydrocephalus.
13 cases of hydrocephalus symptoms were admitted to the hospital, including 8 cases of unstable walking, 9 cases of dizziness and headache, 2 cases of defecation disorder, 1 case of cognitive impairment; the remaining 8 cases were admitted to the hospital due to hearing loss, facial paralysis and other acoustic neuroma symptoms. The examination diagnosed as secondary hydrocephalus.
1.4 Treatment plan
All patients underwent suboccipital sigmoid sinus resection of acoustic neuroma. A straight incision of 8~10cm behind the suboccipital sigmoid sinus was performed on the affected side. The oval bone window of the posterior cranial fossa was 5cm×3.5cm, up to the transverse sinus, and to the outside. At the root of the mastoid, expose the sigmoid sinus and the angle of the transverse sinus, cut the dura mater, open the cisternus magna, release the cerebrospinal fluid, pull the cerebellum, pay attention to protecting the petrosal vein, trigeminal nerve and superior abductor nerve, and remove the tumor in pieces. Routine lumbar cistern drainage after tumor resection.
When hydrocephalus symptoms are severe and brain herniation is formed, ventricular drainage should be performed first, and acoustic neuroma resection should be performed in the second stage. After acoustic neuroma resection, the symptoms of hydrocephalus have not been alleviated, and even worsened during the follow-up period, a second-stage ventricular-abdominal shunt should be considered. All patients were routinely followed up within 3 to 6 months after surgery, and rechecked with head MRI.
2. Results
2.1 Acoustic neuroma surgery results
Postoperative CT and MRI of the head showed that the tumors were completely removed. Facial nerve anatomy was preserved in 20 cases; postoperative facial nerve function classification: House-Brackman grade Ⅰ~Ⅱ 19 cases, Ⅲ~Ⅳ grade 2 cases.
2.2 Results of hydrocephalus surgery
One case was admitted to the hospital with symptoms of acute hydrocephalus and underwent emergency ventricular drainage.
Two cases of acute hydrocephalus within 3 days after tumor resection were treated with ventricular drainage; one of them underwent ventricular-abdominal shunt 1 month later. Three cases underwent ventricular-abdominal shunt 3 to 12 months after tumor resection.
The ventricle shrank in 10 cases immediately after acoustic neuroma resection, 5 cases shrank ventricle after acoustic neuroma resection plus hydrocephalus surgery, 3 cases shrank ventricle during follow-up, and 3 cases showed no significant ventricle shrunk.
One patient was admitted to the hospital with a diagnosis of non-communicating hydrocephalus. After the resection of the acoustic neuroma, a re-examination of the head CT revealed that the fourth ventricle was dilated, and the postoperative diagnosis was communicative hydrocephalus (Figure 1).
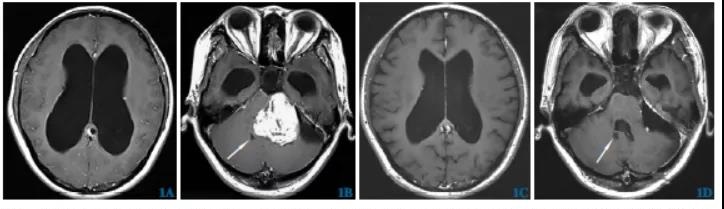
Of the 13 patients admitted to the hospital with symptoms of hydrocephalus, 10 cases were significantly relieved before discharge. During the follow-up, the symptoms of hydrocephalus improved to some extent in 3 cases; 8 cases of patients admitted to the hospital with symptoms of acoustic neuroma had symptoms during the follow-up process. Significant improvement.
2.3 Prognosis
During the follow-up period of 21 cases, MRI showed no tumor residual or recurrence, and the symptoms of acoustic neuroma and hydrocephalus were improved to some extent. One case of hemorrhage in the surgical area after acoustic neuroma resection, and one case of supratentorial hematoma after acoustic neuroma resection, all underwent emergency cerebral hematoma removal. After the symptoms improved, they were taken to a rehabilitation hospital for further treatment.
3 Discussion
The vast majority of patients with acoustic neuroma and hydrocephalus are non-communicating hydrocephalus. The reason is that when the acoustic neuroma is large, the tumor can block the circulation of cerebrospinal fluid and cause the ventricles to dilate.
Some patients were diagnosed as communicating hydrocephalus (2 cases in this group) on preoperative examination. The cause may be that acoustic neuroma leads to increased cerebrospinal fluid protein levels, causing brain tissue adhesion and subarachnoid inflammation, and finally causing cerebrospinal fluid absorption disorder .
1 patient in this group was diagnosed as non-communicating hydrocephalus before operation and after postoperative diagnosis as communicating hydrocephalus. The possible reasons for this phenomenon are presumed to be:
①The patient’s hydrocephalus has both circulatory obstruction and malabsorption factors. ; Therefore, when the tumor is removed and the circulatory obstruction is relieved, it appears as communicating hydrocephalus.
②The patient belongs to communicative hydrocephalus, because the fourth ventricle is not expanded due to direct compression of the tumor. When the tumor is removed, the fourth ventricle turns into an expanded state.
③The patient belongs to non-communicating hydrocephalus, due to the long-term expansion of the ventricle, which leads to decreased compliance. When the obstruction is relieved, the ventricle is difficult to recover, resulting in the expansion of the fourth ventricle after the operation. In principle, it is not recommended to actively treat hydrocephalus.
Most of the patients in this group did not undergo hydrocephalus surgery (15 cases), and the symptoms of hydrocephalus can be significantly improved, and the ventricle shrinks. Because surgical removal of the tumor body can relieve the cerebrospinal fluid circulation disorder, while reducing the level of cerebrospinal fluid protein, reducing the degree of cerebrospinal fluid absorption disorder, most secondary hydrocephalus can be relieved or even healed after the removal of acoustic neuroma.
When hydrocephalus affects the progress of acoustic neuroma surgery, hydrocephalus should be actively treated. In this group, the retrosigmoid approach was used for resection of acoustic neuroma.
After cutting the dura mater during the operation, the intracranial pressure of patients with hydrocephalus is usually high, which is not conducive to the exposure of the surgical field and the operation of the surgeon. Routine surgery to open up the cisternus magnum to release cerebrospinal fluid and reduce intracranial pressure.
In one patient, the intracranial pressure was extremely high during the operation, and the edge of the cerebellar hemisphere incarcerated bone window herniated. The occipital angle of the lateral ventricle was punctured, and about 30ml of cerebrospinal fluid was slowly released. The pressure in the posterior fossa decreased.
One patient was admitted to the hospital with symptoms of acute hydrocephalus, manifested as respiratory rhythm disturbance and progressive consciousness disturbance. He was immediately given emergency ventricular drainage. After the patient’s conditions permit, the second-stage surgical resection of the acoustic neuroma was performed. The ventricles of patients with hydrocephalus have a significant decrease in compliance due to long-term expansion.
The ventricles are difficult to recover quickly after the resection of the acoustic neuroma. At the same time, the intracranial pressure is lower than before the operation. The pathophysiological process is similar to that of normal pressure hydrocephalus. The curative effect of hydrocephalus surgery in such patients cannot be evaluated. Lumbar cistern drainage or lumbar puncture and drainage test should be performed first. If the symptoms have improved significantly, surgical treatment of hydrocephalus may be considered.
The symptoms of 3 patients in this group were significantly improved after lumbar cistern drainage, of which 2 patients underwent ventricular-abdominal shunt within 3 months after acoustic neuroma resection, 1 patient delayed surgery due to intracranial infection, and 1 year later due to hydrocephalus symptoms Repeated aggravation was performed by ventricular-abdominal shunt, and all 3 cases were cured.
Not all symptoms of hydrocephalus can be effectively relieved after acoustic neuroma resection, and some patients have even worsened symptoms. Two patients in this group developed acute hydrocephalus 3 days after resection of acoustic neuroma, and their condition improved after ventricular drainage in the emergency department. One patient underwent ventricular-abdominal shunt 1 month after surgery.
The reason for the aggravated hydrocephalus is speculated to be the traction and damage of the surrounding brain tissue by the surgical operation, which leads to postoperative brain tissue edema, and the circulation of cerebrospinal fluid is further restricted than before the operation, which eventually leads to acute hydrocephalus. For patients with aggravated hydrocephalus, ventricular drainage is used to relieve symptoms in the acute phase.
The conventional drainage tube is placed for 3 to 7 days and then removed. According to the patient’s symptoms, it is determined whether to receive lumbar cistern drainage or lumbar puncture. Regularly after the peak period of cerebral edema Follow up the degree of hydrocephalus, and carefully choose surgical treatment of hydrocephalus flow. In this group, 2 cases of acoustic neuroma had intracranial hematoma after resection of acoustic neuroma, and 1 case had hemorrhage in the operation area on the second day after operation. This case of acoustic neuroma has a large tumor body and long-term pressure on the surrounding brain tissue. The separation and traction injury during operation It is the main cause of postoperative bleeding.
A postoperative head CT showed a subdural hematoma in the frontotemporal area of the affected side. He was treated with cerebral hematoma in emergency. During the operation, two veins of the left frontal pole and superior frontal gyrus returning to the sagittal sinus were clearly ruptured and bleeding. The reason was During the resection of acoustic neuroma, the intracranial pressure decreased rapidly, and the brain tissue shifted, which caused the vein to tear and hemorrhage.
Therefore, during the surgical treatment of acoustic neuroma, you should not excessively pursue the reduction of intracranial pressure, especially when opening the cisternoma occipitalis, ventricular puncture, postoperative lumbar puncture, and lumbar cistern drainage, you must slowly release the cerebrospinal fluid, and gradually reduce the intracranial pressure.
(source:internet, reference only)
Disclaimer of medicaltrend.org



