Minimally invasive surgery for the treatment of ventricular cast hematoma
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Minimally invasive surgery for the treatment of ventricular cast hematoma
Minimally invasive surgery for the treatment of ventricular cast hematoma. Research progress of ventricular cast hematoma and its minimally invasive surgical treatment.
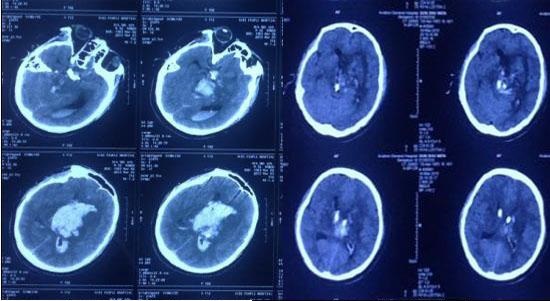
Ventricle cast hematoma is common in primary ventricular hemorrhage and hypertensive intracerebral hemorrhage and breaks into the ventricle. It is a common critical illness in neurosurgery. It has a rapid onset, severe disease, poor prognosis, and extremely high mortality and disability rates. In the world, more than 1 million people die from cerebral hemorrhage every year. In China, 60 to 80 people per 100,000 people have a history of cerebral hemorrhage, and the mortality rate of cerebral hemorrhage is as high as 50% to 70%.
The pathogenesis and pathophysiological changes of ventricular cast hematoma are closely related to the patient’s prognosis. At present, minimally invasive surgery such as lateral ventricle puncture and external drainage and neuroendoscopic surgery are often used to treat ventricular cast hematoma. This article reviews the pathogenesis, pathophysiological changes and the status of minimally invasive surgical treatment of ventricular cast hematoma.
1. The pathogenesis of ventricular cast hematoma
The formation of ventricular cast hematoma is not caused by a single factor, but often combines multiple factors. At present, most scholars believe that the theory of microaneurysm rupture in cerebrovascular disease is the basic mechanism of ventricular cast hematoma formation.
1.1 Theory of microaneurysm rupture
When the body is in a state of hypertension for a long time, it will cause increased shear stress on the blood vessel wall (that is, increased friction between the blood and the vascular intima), and increased oscillating shear index (that is, the intensity and direction of blood flow in the blood vessel is unstable, resulting in more (Reflux and vortex), increased wall shear stress gradient (indicating decreased vascular compliance), faster blood flow, and wider impact on the vascular intima. Affected by hemodynamic changes, the tension of blood vessels in the brain, especially the walls of small arteries in the brain, changes, which is the basis for the formation of aneurysms. Aneurysms are common in the weakened parts of small arteries. Multiple miliary microaneurysms are more common in perforating arteries in the brain, which are saccular and usually about 500μm in diameter.
When the blood pressure suddenly rises due to emotional excitement, strenuous exercise, or irregular use of antihypertensive drugs, multiple miliary microaneurysms are very easy to rupture and hemorrhage. When rupture and hemorrhage occurs in the basal ganglia or ventricle, it will cause ventricular casting. Formation of type hematoma.
1.2 Lipid hyaline degeneration and hyaloid degeneration of blood vessels in the brain
When the blood vessels in the brain, especially the small arteries in the brain, are under continuous high pressure for a long time, the blood vessel wall will undergo lipid hyaline degeneration and lipid hyaline degeneration. The proliferation of fibroblasts under the vascular intima replaces the original vascular smooth muscle cells, which makes the blood vessels hard and elastic. Decrease, and a large number of macrophages adhere to the inner wall of the blood vessel after phagocytosis of lipids, causing necrosis of the blood vessel wall and narrowing of the vascular lumen, leading to further increase in intravascular pressure, especially for perforating small arteries with a diameter of 100 to 300 μm. serious. When the pressure in the blood vessel exceeds the load pressure of the blood vessel or the blood pressure fluctuates greatly, the blood vessel in the brain is very easy to rupture and hemorrhage and eventually lead to the formation of intraventricular hematoma.
1.3 Atherosclerosis in the brain
Atherosclerosis mainly involves the middle cerebral artery and the basilar artery, followed by the deep perforating small arteries; atherosclerotic lesions have different stages:
- (1) Early lesions, also called striatal stage, where a large number of foam cells gather in the arterial intima of the lesions, forming yellow needle-like spots and striped lesions, the size of the spots varies, the length of the stripes varies, and the direction of the stripes Parallel to the long axis of the blood vessel.
- (2) Fibrous plaque stage, the characteristic manifestation of this stage is the formation of a fibrous cap in the vascular intima of the lesion, which is formed by the compensatory hyperplasia of intimal fibers. Hyalid degeneration occurs in the vascular intima, lipids are deposited in the degeneration, and yellow plaques gradually evolve into slightly shiny, wax drop-like gray-white plaques.
- (3) At the atheroma stage, the main manifestations are fibrous cap degeneration and necrosis, the plaques continue to grow larger, and the adjacent plaques merge with each other and bulge, and the localized narrowing of the vascular lumen is more obvious. Under the microscope, it can be seen that a thin layer of fibrous connective tissue is formed on the surface of the lesion, a large amount of cholesterol crystals and red-stained necrotic materials are formed in the deep part, a large number of granulation tissue proliferation and foam cells infiltrate the base and edge of the plaque.
- (4) The formation of atherosclerotic plaque will cause secondary damage. The plaque will cause the vascular intima to be uneven, easily form thrombus, cause stenosis of the lumen, and deposit calcium in the plaque, decrease the elasticity of the blood vessel wall and increase the fragility , Can cause smooth muscle atrophy and thinning of the lesion, local tumor-like expansion, and the formation of aneurysms, which are affected by blood pressure on the basis of the formation of aneurysm, and then increase the risk of vascular rupture. Therefore, blood vessel rupture and bleeding is a joint effect of blood pressure and vascular disease.
2. Pathophysiological changes of ventricular cast hematoma
The main pathophysiological changes of ventricular cast hematoma are the spongiform degeneration of the ventricle wall and surrounding tissues 30 minutes after the formation of the ventricular cast hematoma; the necrotic layer, hemorrhage layer, and hemorrhage layer appear in the brain parenchyma around the ventricle wall after 6-8 hours. Spongiform degeneration and edema zone.
2.1 Space effect
Cerebrospinal fluid is a colorless and transparent liquid that is secreted by the choroid plexus of each ventricle, mainly from the choroid plexus of the lateral ventricle; the pressure of normal cerebrospinal fluid is maintained between 686-1765Pa (70-180mmH2O). The channels for circulating cerebrospinal fluid such as the interventricular foramen, the third ventricle, the midbrain aqueduct, and the fourth ventricle are relatively narrow. When blood enters the cerebrospinal fluid circulation system, it is easy to be blocked in the stenosis. Ventricular cast hematoma increases the contents of the cranial cavity, leading to the destruction of the stability of the cranial cavity; and the hematoma has a space-occupying effect, which can cause obstruction of stenosis in the cerebrospinal fluid circulation system, induce acute obstructive hydrocephalus, and cause rapid intracranial pressure Increased, compressing the surrounding normal brain tissue, causing a rapid decrease in cerebral perfusion pressure, which can further aggravate the ischemic and hypoxic damage of brain cells.
The intracranial pressure above the obstruction is significantly increased. When the pressure difference between the two sides of the obstruction exceeds a certain limit, the tentorium notch hernia and/or the foramen magnum hernia can be induced. Therefore, the most common direct cause of death in patients with ventricular cast hematoma is brain herniation.
2.2 Secondary damage
(1) The red blood cells in the ventricular cast hematoma quickly die and lyse, and release a large amount of oxygen free radicals, magnesium ions, vasoactive substances, endothelin, etc., which change the vascular permeability, cause cerebral vasospasm or expansion, and make the surrounding hematoma normal Hypoxic-ischemic changes in brain tissue.
(2) After the formation of ventricular cast hematoma, the complement system can be activated, a large number of white blood cells are activated, and inflammatory mediators are activated and expressed; after the decomposition of the hematoma, a large number of inflammatory cells gather around the hematoma, causing inflammatory reactions, destroying the blood-brain barrier, and leading to brain edema Further aggravated.
(3) After the formation of ventricular cast hematoma, especially in the subacute phase (3-7 days after cerebral hemorrhage), the red blood cells in the hematoma cavity lose the protective effect of blood vessels, the red blood cells begin to lyse, and a large amount of hemoglobin and Fe3+ are released. In the formation and development of hydrocephalus after cerebral hemorrhage, Fe3+ and hemoglobin play a key role.
Studies have shown that direct injection of hemoglobin and Fe3+ into the ventricle greatly increases the probability of hydrocephalus. After hemoglobin enters the ventricular system, the complement is activated, and the expression of lipocalin 2 (LCN2) increases, which significantly increases the incidence of ventricular dilatation. A large amount of Fe3+ deposited in the ventricle after cerebral hemorrhage can activate the nuclear factor E2-related factor 2/hemooxygenase-1 (nuclear factor erythroid 2-related factor 2/hemooxygenase 1, Nrf/HO-1) pathway, resulting in iron content Increased protein expression. The study found that injection of a large amount of Fe3+ into the cerebral ventricle of rats can induce acute ventricular dilation and hydrocephalus. After 24 hours of injection of deferoxamine into the cerebral ventricle, the ventricle collapsed and the hydrocephalus was significantly improved.
Studies have shown that after injection of thrombin into the cerebral ventricle of rats, the expression of transforming growth factor β1 (TGF-β1) increases, the subarachnoid space appears progressive fibrosis, the blood clots in the ventricle increase significantly, the ventricle dilates significantly, and the brain The degree of hydrocephalus was significantly increased; and after the injection of TGF-β1 inhibitor into the cerebral ventricle, the degree of ventricular expansion was inhibited and hydrocephalus was significantly improved.
(4) After the formation of ventricular cast hematoma, in order to drain the blood cerebrospinal fluid and solve the obstructive hydrocephalus, thrombolysis as soon as possible to reduce the time of blood retention in the ventricular system, tissue plasminogen activator (tissue plasminogen) is often used after surgery. activator, TPA) is perfused into the lateral ventricle of the hematoma cavity. TPA can accelerate thrombolysis, inhibit thrombosis, and inhibit ventricular expansion. However, some scholars believe that after the hematoma cavity is perfused with TPA, the hematoma dissolves faster, but it also produces a large amount of metabolites, which can cause severe inflammation.
3. Minimally invasive surgical treatment of ventricular cast hematoma
After the formation of ventricular cast hematoma, the space-occupying effect of blood clots can induce acute obstructive hydrocephalus, and there are risks of intracranial hypertension, brain tissue compression, and brain herniation. The effect of conservative medical treatment is not ideal, and surgical treatment is the reason. The main treatment method. Because the removal of ventricular hematoma by craniotomy can cause serious damage to patients and have many postoperative complications, this surgical treatment is not recommended clinically. At present, minimally invasive surgery is mainly used to treat ventricular cast hematoma, including lateral ventricle puncture and external drainage and neuroendoscope-assisted removal of ventricular cast hematoma.
3.1 Extraventricular puncture drainage
Take 2.5cm in front of the coronal suture and 2.5cm next to the midline (side ventricle of hematoma) as the puncture point. Cut the first skin longitudinally, about 4cm in length; drill a cranial drill to make a bone window with a diameter of about 2cm, with a “cross” Cut the dura mater. Hold the ventricle puncture needle perpendicular to the direction parallel to the midline and perpendicular to the line of the external ear canal. The depth of the needle is about 5-6cm. After the hollow is felt, the needle core is pulled out, and the bloody cerebrospinal fluid flows out. Remove the puncture needle. Into the drainage tube and lead out from the side hole.
After the re-examination of the skull CT confirms that the tip of the drainage tube is located in the ventricle to ensure smooth drainage of the drainage tube and confirm that there is no progressive bleeding, inject urokinase into the hematoma cavity, 20,000 to 40,000 IU/time, 2 times/d. After urokinase is perfused, the drainage tube needs to be clamped for 2 to 3 hours before opening. During the treatment, pay attention to the color change of the cerebrospinal fluid, and regularly review the head CT; after confirming that the hematoma is basically removed, try to clamp the drainage tube for 1 to 2 days. If the patient has no obvious signs of hydrocephalus, the drainage tube can be removed (the drainage tube is indwelling The time should not exceed 7 days); if the drainage effect of hematoma is not satisfactory, patients who need to extend the drainage tube indwelling time can replace the drainage tube to reduce the risk of infection.
3.2 Neuroendoscope-assisted removal of ventricular cast hematoma
The bone window approach was adopted, and the bone window was drilled according to the method of lateral ventricle puncture and external drainage. Insert a cannula and an endoscope to the side of the intraventricular hematoma. When the endoscope enters the hematoma cavity, in order to ensure the expansion of the ventricle, the flushing channel needs to be opened, and the flushing pressure is maintained at 10-15cmH2O. Do not forcibly pull the blood clot attached to the ventricle wall To prevent secondary bleeding caused by vascular damage; if active bleeding is found in the hematoma cavity or the wall of the ventricle, bipolar coagulation can be used to stop the bleeding.
Try to remove the intraventricular hematoma without causing secondary damage. Don’t emphasize the complete removal of the hematoma at one time during the process of removing the hematoma. It can be removed multiple times and in small amounts to reduce intracranial hypertension and relieve obstructive cerebral hematoma. Water for the purpose. During the operation, care should be taken to insert the endoscope into the third ventricle under safe conditions and remove its hematoma, so as to maintain the ventricular system as unobstructed as possible. After the hematoma is removed, a drainage tube should be placed in the ventricle on the side of the hematoma, and the head CT should be reviewed within 24 hours after the operation to observe the removal of the hematoma. If there is still a large intracranial hematoma (>10mL), and may cause obstructive hydrocephalus, please refer to the method of lateral ventricle puncture and external drainage to perfuse urokinase into the hematoma cavity.
During treatment, observe the color change of the cerebrospinal fluid, and regularly review the head CT to confirm that the hematoma is basically cleared. After the cerebrospinal fluid circulates smoothly, try to clamp the drainage tube for 1 to 2 days. If the patient has no obvious signs of hydrocephalus, the drainage tube (drainage tube) can be removed. The indwelling time should not exceed 7 days); if the drainage effect of the hematoma is not satisfactory, the drainage tube can be replaced if the drainage tube needs to be extended.
4. Evaluation and prospect of surgical methods
Ventricle cast hematoma is a kind of cerebral hemorrhage disease with high fatality rate and disability rate. Its clinical manifestations are different. In mild cases, only meningeal irritation may be manifested, such as headache and vomiting, without unconsciousness and neurological loss; With the further deterioration of the disease, severe cases may experience coma, partial neurological loss, etc. In the late stage, severe complications such as brain herniation, decerebral rigidity, and complete loss of neurological function may be life-threatening.
Simple ventricle puncture and external drainage is one of the commonly used surgical methods for the treatment of ventricular cast hematoma. Although this surgery has the advantages of less trauma and easy operation, it also has certain limitations and blindness, and cannot effectively stop bleeding. The treatment effect of patients with progressive hemorrhage is not good, and the hematoma with space-occupying effect cannot be removed as soon as possible. The hematoma lasts for a long time, and the probability of hydrocephalus is significantly increased. At the same time, the postoperative drainage tube indwelling time is too long and repeated Infusion of TPA into the hematoma cavity leads to a relatively higher probability of postoperative intracranial infection, which seriously affects the prognosis of patients. Therefore, although ventricular puncture and external drainage has achieved certain effects in the treatment of ventricular cast hematoma, the fatality rate and disability rate of patients are still high.
How to quickly, safely and effectively remove ventricular hematoma and reduce postoperative complications has become the key to effectively reducing the disability and mortality of patients with ventricular cast hematoma. With the continuous advancement of neuroendoscopy technology and the continuous improvement of neuroendoscopy equipment, neuroendoscopy-assisted ventricular hematoma removal has become an important method for the treatment of ventricular cast hematoma. Neuroendoscopy can greatly reduce the blind area of the visual field during the operation, the visual field is clearer, and the scope of exposure of the hematoma is wider. It can accurately identify the ventricular wall, nerves, blood vessels, choroid plexus and other structures, and achieve the purpose of observing and removing the hematoma under close guidance. .
In addition, the placement of the drainage tube under the direct vision of the neuroendoscope can reduce the damage to the ventricular membrane due to blind placement of the tube, and can avoid the choroid plexus as much as possible, and avoid the blockage caused by the drainage tube being surrounded by the choroid plexus as much as possible. Intraoperative neuroendoscopy under direct vision is more precise and meticulous, which can avoid bleeding caused by blind puncture of lateral ventricle puncture and external drainage, and the intraoperative damage is significantly reduced, postoperative complications are significantly reduced, and the efficacy is significantly improved.
The study found that the neuroendoscope-assisted ventricular hematoma removal operation adopts a microbone window approach, which has small intraoperative damage, avoids the damage caused by large bone flap craniotomy, and has a short postoperative drainage tube indwelling time and absolute bed rest time. Short, fast recovery; and the endoscope tube has a long length and a small diameter, and it is less invasive to the brain tissue during the operation. It is suitable for intraventricular operation and can enter the contralateral ventricle through the interventricular foramen to remove hematoma.
Compared with lateral ventricle puncture and external drainage, neuroendoscopic surgery has many advantages, but due to the expensive equipment of neuroendoscopy and higher technical requirements for operators, it has not been popularized in the majority of primary hospitals, and neuroendoscopy The lateral perspective of the tube has certain limitations. There are still some blind areas of the visual field, which cannot achieve the panoramic observation of the ventricle. It is difficult to enter the third and fourth ventricles, so it is more difficult to effectively remove the hematoma. There are risks that may cause damage to the ventricle wall. Therefore, the operation skills of neuroendoscopy in the treatment of ventricular cast hematoma still need to be improved, and the equipment still needs to be further improved. The treatment of patients with ventricular cast hematoma still requires continuous in-depth research to optimize the treatment plan and improve surgical skills , And ultimately achieve the goal of improving patient survival rate and improving prognosis.
(source:shenjingqianyan, reference only)
Disclaimer of medicaltrend.org



