How many neutralizing antibodies are effective to prevent COVID-19?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
[Nature Sub-Journal] Answered for the first time: How many neutralizing antibodies are effective to prevent COVID-19?
How many neutralizing antibodies are effective to prevent COVID-19? To achieve herd immunity through vaccination is the most important way to liberate humans from the COVID-19 epidemic.
The release of vaccine clinical trial data has shown us that the protective power of different vaccines varies greatly; at the same time, as the number of vaccinated people increases, it has also been reported that more and more people are still infected with COVID-19 after full vaccination. These cases are called breakthrough infections, which are even very serious in some countries and regions.
After vaccination in the United States, there have been more than 9,000 breakthrough infections. Is it okay to not wear a mask indoors?
The vaccination rate is much higher than that of Israel, but the number of new cases is the highest in the world; what truth is this country telling us?
These status quo have made us repeatedly ask a question:
What level of serum neutralizing antibody titers need to reach after COVID-19 vaccination to make the vaccine protective?
A Nature Med article online today is extremely important and highly recommended, because it answers the question of how many antibodies and vaccines are needed for protection for the first time.
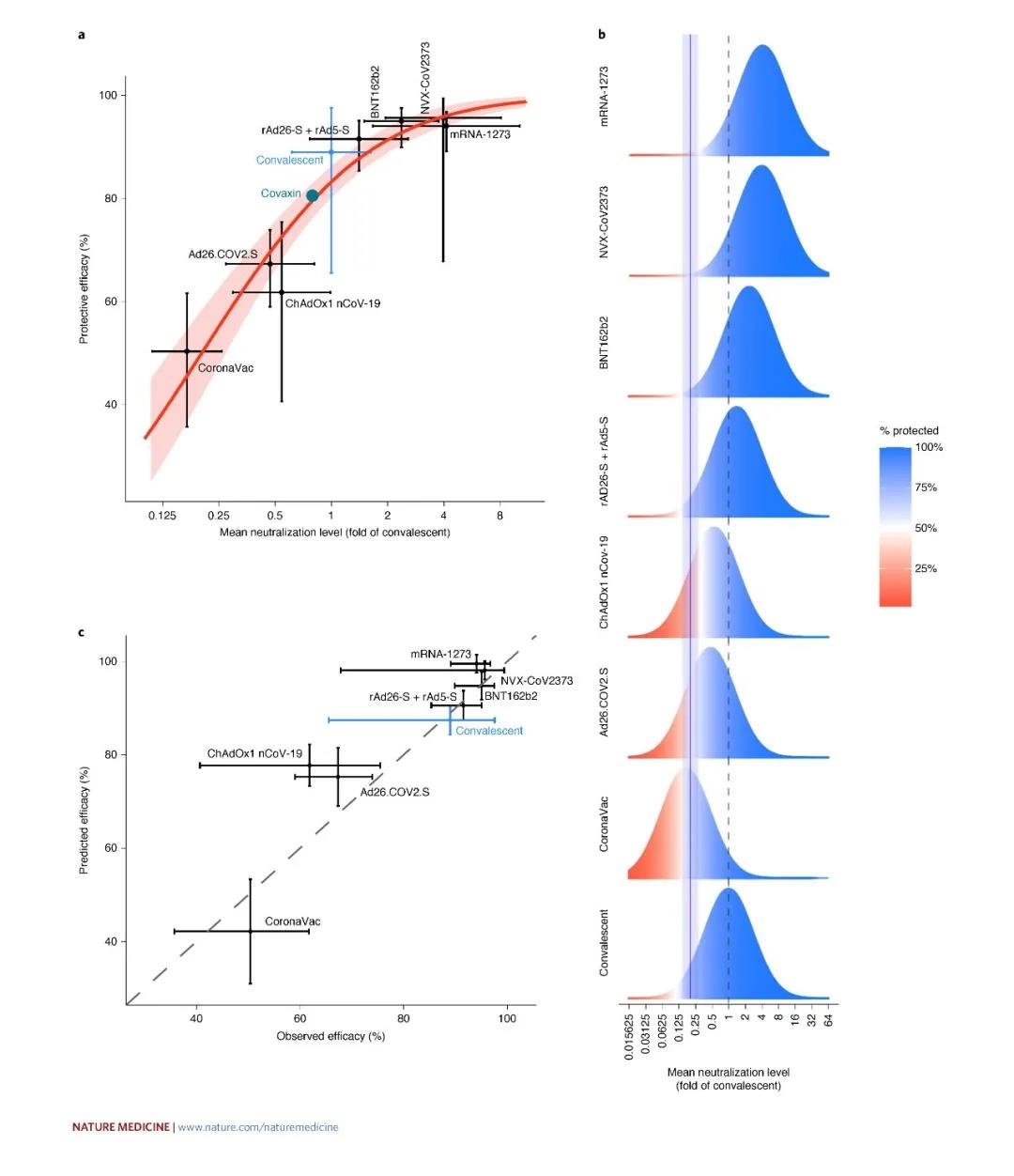
This study found that as long as the serum neutralization activity reaches 20.2% of the average recovery person’s serum neutralization activity, it can have more than 50% protection. This study also compared the serum neutralization activities induced by the current seven major vaccines in the world. Therefore, all current vaccines can meet this requirement.
This important discovery by the University of New South Wales in Australia proves that neutralizing antibodies after a patient is immunized (infection or single vaccination) can predict the severity of symptomatic infection.
The seven vaccines analyzed are: mRNA vaccine mRNA-1273 from Moderna, USA, mRNA vaccine BNT162b2 from Pfizer/Germany BioNTech, protein vaccine NVX-CoV2373 from Novavax, Russia, and adenovirus vector vaccine rAd26-S+rAd5-S from Russia , AstraZeneca’s adenovirus vector vaccine ChAdOx1 nCoV-19, AstraZeneca’s adenovirus vector vaccine Ad26.COV2.S made in India, and the inactivated virus vaccine CoronaVac of China Kexing Company.
This study found that as long as the serum neutralization activity reaches 20.2% of the average recovery person’s serum neutralization activity, it has a protective effect of more than 50% (95% CI = 14.4-28.4%).
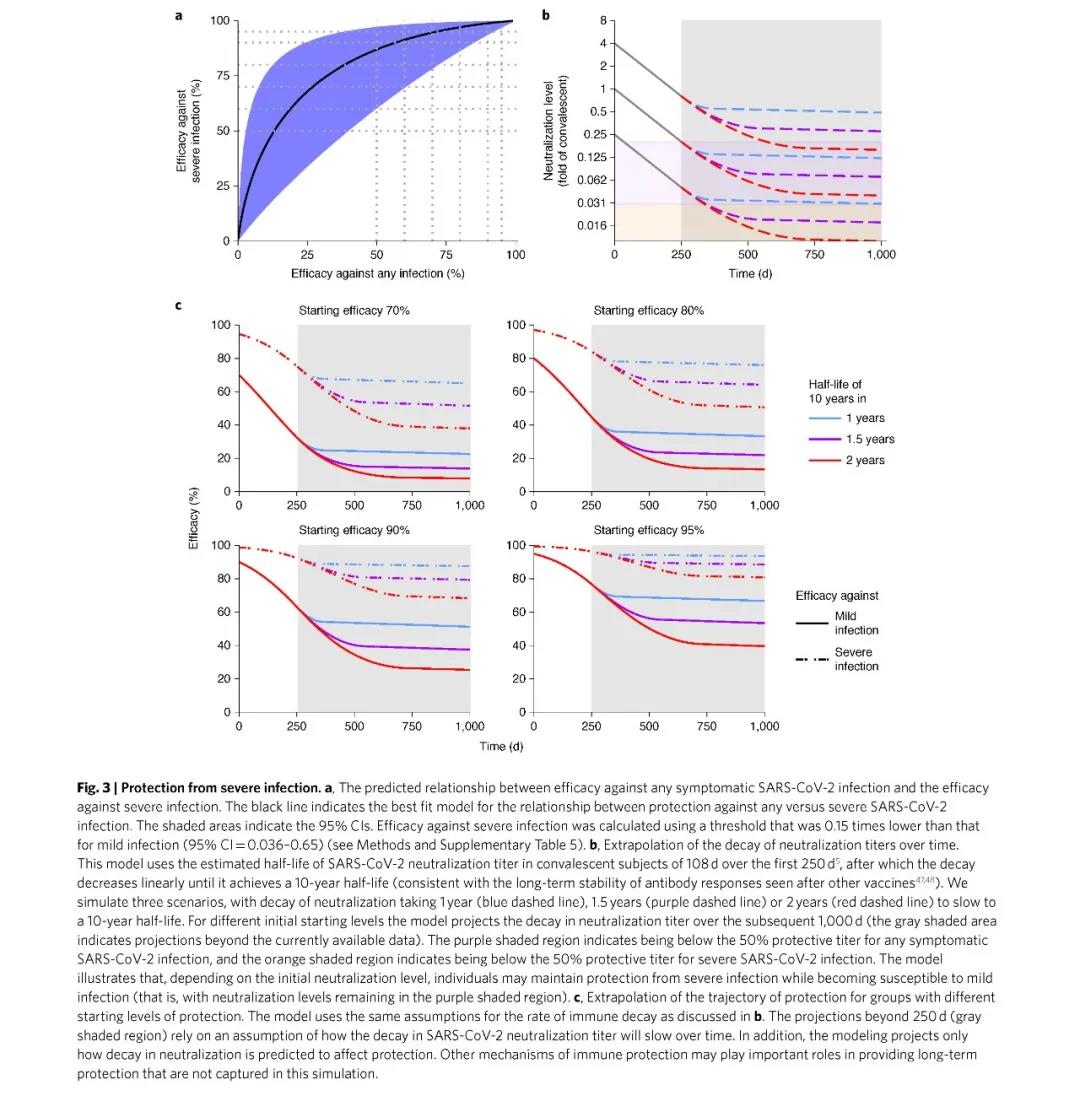
And if 50% probability of preventing severe illness will not occur, the neutralizing activity can reach 3% of the average serum neutralizing activity of the recovered patient (95% CI = 0.7–13%, P = 0.0004).
Studies have found that the serum neutralization titer will be significantly attenuated 250 days after immunization, thereby reducing the protection against repeated infections, but the effect of preventing severe COVID-19 is very long-lasting.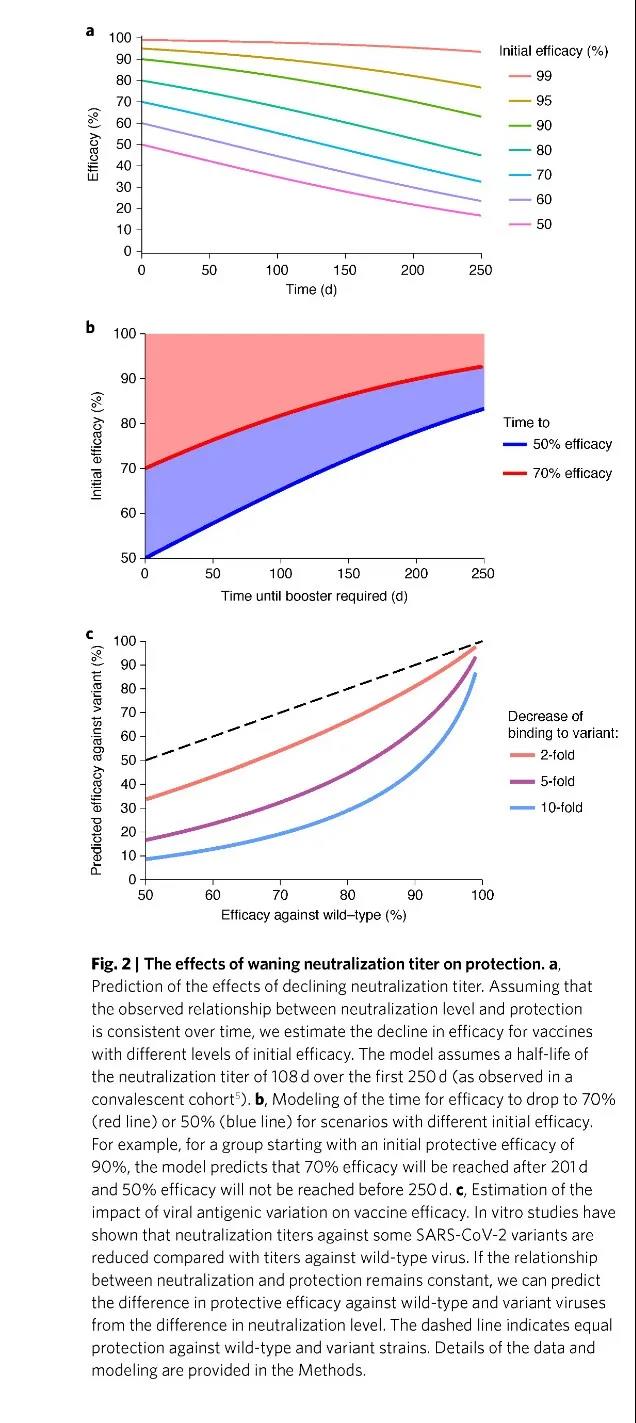
The study found that mRNA-1273, NVX-CoV2373 and BNT162b2 induced the highest neutralizing activity through analysis of vaccines with neutralization data in current phase I/II clinical trials. The neutralizing activity and protective power of Covaxin, Ad26.COV2.S, AZD1222 and CoronaVac were lower than those of the recovered serum itself.
Therefore, studies believe that the serum neutralization activity induced by the vaccine is directly related to the protective power, as well as the protective power against the mutant strain. This research is very important, it helps to develop vaccine strategies to control the pandemic.
Editor’s note:
Seeing this paper, our editorial team smiled at each other.
Because, we reported a long time ago that the level of neutralizing antibodies in early clinical trials is directly positively correlated with the protection of symptomatic COVID-19 shown in late clinical trials.
This is easy to understand, because the anti-coronavirus relies on neutralizing antibodies.
One member was also troubled by this yesterday (May 17). Because yesterday was the U.S. tax filing day, the stocks of vaccine research and development companies that he bought in advance based on this predictive indicator showed super high growth; the direct result is: very, very large taxes need to be paid!
It can be seen that in the early clinical trials of Pfizer/BioNTech of Germany, Moderna of the United States, and Novava of the United States, neutralizing antibodies were all about 4 times higher than those of recovered patients, and all three vaccines reached about 95% protection. And it has been tested in the real world. The most typical example is that Israel exchanged data disclosure for the priority supply of Pfizer’s mRNA vaccine, which was the first to achieve epidemic control. Although the current vaccination rate is only 60%, epidemic control has been the best result among the countries in the world that rely on vaccine control.
Then, as the neutralizing antibody decreases, the protection against symptomatic COVID-19 gradually decreases.
- The neutralizing antibody induced by the Russian satellite V is 1.49 times that of the recovered, and the protective power is 91%;
- British AstraZeneca vaccine is 1.12 times and the protection is 70%;
- The vaccine of SINOPHARM Beijing Institute is 0.6 times, and the protection is 79%;
- Kexing (SINOVAC) Group vaccine is 0.19 times, and the protection is 50.38% (Brazilian data).
The Nature Medicine article shows that the vaccine-induced neutralizing antibody reaches 20% of the recovered patients to achieve a 50% protective effect; the greater value is to reveal that if a 50% probability of preventing severe illness does not occur, the neutralizing activity It is enough to reach 3% of the average serum neutralization activity of the recovered patients.
This shows that even 250 days after vaccination, the serum neutralization titer will be significantly attenuated, thereby reducing the protection against repeated infections, but the effect of preventing severe COVID-19 is very long-lasting.
This is a great comfort to human beings who have already done a good job of living with the new coronavirus until at least 2025. Because even if unfortunately infected, people who have been vaccinated still have a higher probability of being protected from severe infections.
This result has also been confirmed in Seychelles, the country with the highest rate of new infections in the past month.
COVID-19 vaccine is useless even its vaccination rate higher than Israel?
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



