Genetic engineering to transform red blood cells to develop anti-cancer therapy
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Nature: Genetic engineering to transform red blood cells to develop anti-cancer therapy
Genetic engineering to transform red blood cells to develop anti-cancer therapy. T cells specifically recognize tumor antigens through T cell receptors (TCR) and play an important role in anti-tumor immunity.
In recent years, methods that modulate or simulate T cell responses (such as immune checkpoint inhibitors and CAR-T therapy) have become an important method of anti-cancer treatment. However, the fatal side effects, the generation of drug resistance, and the expensive production process of autologous CAR-T cells have prompted people to continue to study alternative methods to stimulate T cell-mediated anti-tumor responses.
Red blood cells (RBC) are used for blood transfusion due to their inherent biocompatibility. In addition, red blood cells are involved in a variety of immune privilege mechanisms, protecting them from adverse host reactions. From a manufacturing point of view, the cell culture and differentiation characteristics of hematopoietic progenitor cells allow for massive expansion of red blood cells. In addition, genetic modification can also produce engineered human red blood cells expressing biotherapeutic proteins for use in red blood cell therapy.
As we all know, erythroid precursor cells lose their nucleus during the process of forming mature red blood cells. Therefore, after genetic modification of erythroid precursor cells, the modified protein still expresses, but the genetic material does not exist. There is no rejection problem, and there is no need to worry about the potential negative effects of modified genes on the human body. Moreover, this method can carry out large-scale cultivation and expansion, and the cost will be greatly reduced compared with CAR-T therapy.
Recently, the research team of the American company Rubius Therapeutics published a research paper titled: Engineered red blood cells as an off-the-shelf allogeneic anti-tumor therapeutic in Nature Communications.
This research transforms red blood cells into artificial antigen-presenting cells, enhances the anti-HPV-positive tumor efficacy of T cells, and is expected to realize anti-HPV-positive tumor treatment through autologous red blood cells.

First, the research team infected human CD34+ hematopoietic progenitor cells with lentivirus, and then expanded and differentiated into enucleated engineered red blood cells to produce RCT-aAPC. This red blood cell is composed of tumor-specific peptides that bind to MHC class I, costimulatory ligand (4-1BBL), and interleukin 12 (IL-12). These signals are all necessary to activate T cells. They found that RCT-aAPC can promote antigen-specific T cell expansion, memory formation and cytotoxicity to tumor cells in vitro. It shows that RCT-aAPC can promote the differentiation of T cells into functional CD8+ T cells.
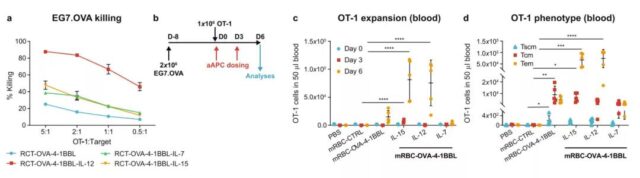
RCT-aAPC cannot be evaluated using the mouse model, because human red blood cells are quickly cleared in the mouse body. The research team produced mRBC-aAPC by using chain chemistry and found that mRBC-aAPC is involved in homologous interactions with antigen-specific T cells, and indicated that the spleen may be one of the main sites of this effect in the body.

In vivo experiments found that mice injected with mRBC-aAPC, compared with mRBC-CTL, tumor tissue growth slowed down, and can form long-term memory to prevent tumor recurrence. It shows that the antigen present on aAPC leads to a more powerful anti-tumor effect. Importantly, tumor control is related to the development of long-term memory and the spread of epitopes, thereby producing therapeutic effects on tumors that do not express the original target antigen but are otherwise identical.

In order to treat HPV16-positive cancer patients, the research team developed RTX-321 based on engineered red blood cells, which express human leukocyte antigen HLA-A and human HPV peptides, 4-1BBL and IL-12. They found that RTX-321 can induce the activation of HPV antigen-specific primary human T cells, and these three signals are sufficient to promote effector function and the differentiation of effector memory cells.

In general, the research has developed a platform for the treatment of cancer with aAPC based on genetically engineered red blood cells. Red blood cells have been used in blood transfusion medicine for decades. Genetically engineered red blood cells can be infused into patients and produced in large quantities without the need for complex and expensive production equipment like TCR-T therapy and CAR-T therapy. The RCT-aAPC platform exhibits a wide range of anti-cancer properties and can be used as a potential cancer immunotherapy method.
About Rubius Therapeutics

As the world’s first company to transform hematopoietic stem cells into special “therapeutic” red blood cells, Rubius is also the only company that has entered the clinical stage. It is expected to become an emerging force in the field of cell therapy. The following article will introduce the company in detail.
Rubius Therapeutics was established in 2014 and is located in Massachusetts, USA, and was incubated by the famous Flagship venture capital laboratory. It is the first company in the world that uses genetic engineering to transform hematopoietic stem cells for cell therapy, and is currently the only red blood cell therapy company that has entered clinical trials.
In 2018, Rubius Therapeutics was listed on the Nasdaq with a market value of US$1.8 billion, and its current market value is US$2.3 billion.
Currently, Rubius Therapeutics has four R&D pipelines undergoing clinical trials for the treatment of solid tumors, acute myeloid leukemia and HPV-16 positive cancers.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



