The mechanism by which vitamin D inhibits the spread of ovarian cancer
- Why Botulinum Toxin Reigns as One of the Deadliest Poisons?
- FDA Approves Pfizer’s One-Time Gene Therapy for Hemophilia B: $3.5 Million per Dose
- Aspirin: Study Finds Greater Benefits for These Colorectal Cancer Patients
- Cancer Can Occur Without Genetic Mutations?
- Statins Lower Blood Lipids: How Long is a Course?
- Warning: Smartwatch Blood Sugar Measurement Deemed Dangerous
The mechanism by which vitamin D inhibits the spread of ovarian cancer
- Red Yeast Rice Scare Grips Japan: Over 114 Hospitalized and 5 Deaths
- Long COVID Brain Fog: Blood-Brain Barrier Damage and Persistent Inflammation
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
The mechanism by which vitamin D inhibits the spread of ovarian cancer.
Among various gynecological tumors, ovarian cancer (OvCa) has the highest mortality rate, and most patients have peritoneal metastasis at the time of diagnosis [1].
Because the ovary is directly exposed to the abdominal cavity, unlike other tumors that tend to metastasize to solid organs such as the lung and liver, OvCa rarely metastasizes through the blood and is more prone to peritoneal metastasis [2].
OvCa can be widely metastasized in the peritoneum, omentum, mesentery, etc. It is difficult to completely remove the tumor by surgery, and the treatment effect is poor.
At present, the clinic mainly relies on chemotherapy drugs such as cisplatin and paclitaxel to achieve complete remission.
Although the development of targeted drugs in recent years has prolonged the progression-free survival of patients, these drugs are still limited in prolonging the overall survival [3-4], and there is still a lack of effective treatments for peritoneal metastasis of OvCa.
Recently, a research team from Nagoya University published an important study in Matrix Biology .
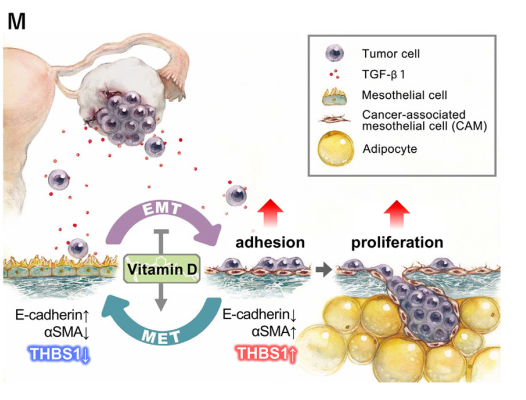
They found that vitamin D can promote the normalization of THBS1 expression, inhibit the mesenchymal transition of mesothelial cells, and reverse the mesenchymal state of tumor-associated mesothelial cells, thereby Inhibits peritoneal metastasis of OvCa [5].

Article cover
Some studies suggest that the control of peritoneal metastasis may play an important role in improving OvCa refractory.
After OvCa cells shed from the primary site, they are carried to the peritoneal wall, mesentery, and omentum through ascites.
Normally, these sites are covered by mesothelial cells (MCs), which protect the internal organs from pathogens and tumor cells [6].
Many studies have shown that MCs have cancer-promoting effects in the tumor microenvironment .
Cancer-associated fibroblasts (CAFs) are the most numerous cells in the tumor stroma and can produce various factors that promote tumor cell growth.
In a previous study, the research team confirmed the presence of many MC-derived CAFs in the OvCa matrix of peritoneal metastases [7].
During peritoneal metastasis, TGF-β1 secreted by tumor cells induces epithelial-mesenchymal transition (EMT) process, and MCs are transformed into mesenchymal state.
EMT-induced MCs, termed tumor-associated mesothelial cells (CAMs), are an important component of the tumor microenvironment that promotes OvCa progression .
Combined with the results of previous studies, the research team believes that inhibiting the activity of CAMs may contribute to the treatment of OvCa.
Studies have shown that vitamin D can not only play a key role in maintaining calcium homeostasis and regulating bone metabolism, but also inhibit cell cycle, proliferation and angiogenesis [ 8].
Furthermore, overexpression of vitamin D receptor (VDR) has been shown to be associated with good prognosis in various cancers [9].
Therefore, the research team speculates that vitamin D can play an anti-tumor effect by inhibiting the EMT of MCs, thereby treating peritoneal metastasis.
Mechanism diagram
First, the research team performed immunohistochemistry (IHC) on calretinin, a specific marker of MCs , to determine the location of MCs metastases in the greater omentum.
The results showed that MCs were present on the peritoneal surface and in areas where tumor cells infiltrated the greater omentum, but no MCs were observed in the primary site of OvCa .
The research team then examined the expression of VDR in MCs and CAMs, and found that MCs arranged on the surface of the greater omentum were VDR-positive, and there were also many VDR-positive MCs in the stroma of omental metastases .
Multiple immunohistochemistry further verified this conclusion.
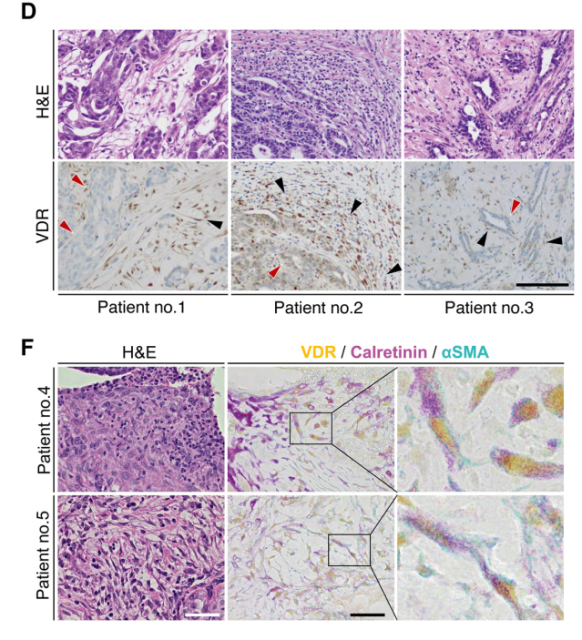
VDR-positive fibroblasts from MCs are seen in OvCa peritoneal metastases
The research team also analyzed data in public databases and found that VDR expression level is an important prognostic factor for OvCa.
Among patients with OvCa of all stages and III, patients with high VDR expression had a 25% lower risk of death (P=0.0013, P=0.0056).
Furthermore, VDR expression was higher in stromal cells than in tumor cells.
These results suggest that high expression of VDR in the stroma, including MCs, is associated with better prognosis in patients with OvCa .
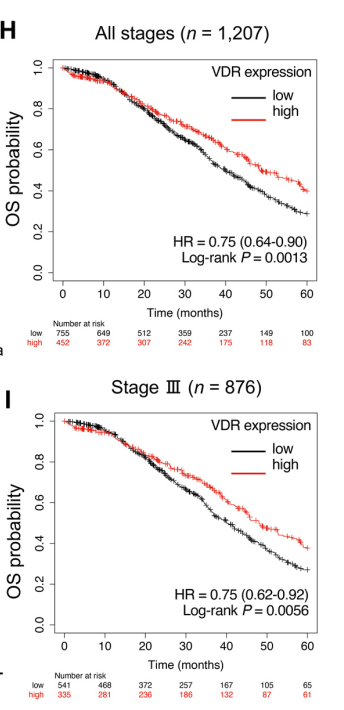
High VDR expression is associated with better prognosis in OvCa patients
Previous studies have demonstrated that malignant ascites of OvCa induces EMT in MCs, and TGF-β1 is the main inducer of EMT in MCs.
The research team explored the effect of vitamin D on MCs in the presence of TGF-β1.
The results showed that vitamin D could correct the morphological changes of MCs induced by TGF-β1 stimulation, the decreased expression of E-cadherin, and the increased expression of aSMA.
Vitamin D can also alleviate TGF-β1 stimulation-induced increases in the levels of EMT markers, such as N-cadherin, MMP2, MMP9, and Slug, among others.
These data suggest that vitamin D attenuates TGF-β1-induced EMT in MCs by altering the morphology and expression of EMT markers .
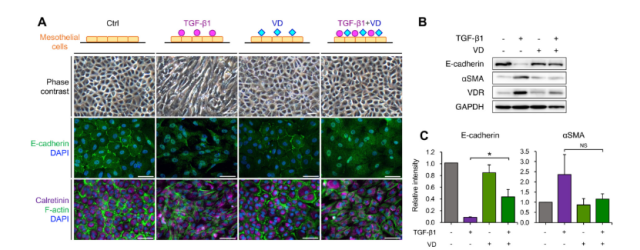
Vitamin D inhibits TGF-β1-induced epithelial-mesenchymal transition
Next, the research team performed RNA sequencing and analysis of MCs in different treatment groups, and found that vitamin D intervention downregulated the expression of EMT and focal adhesion-related genes .
Among them , the expression of thrombospondin -1 (THBS1) was highly up-regulated (27.90-fold) in TGF-β1-stimulated MCs, and was most significantly down-regulated after vitamin D intervention.
After verifying the changes in THBS1 expression by qRT-PCR, the research team analyzed the expression of THBS1 in OvCa patients.
Data from public databases indicated that high THBS1 expression was a poor prognostic factor (HR, 1.35; P=0.0018).
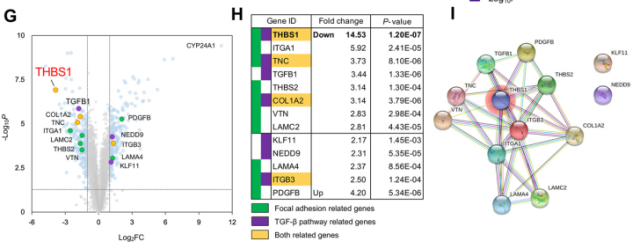
Vitamin D downregulates the expression of THBS1, a key protein that promotes adhesion
Therefore , the research team speculated that the increased expression of THBS1 in MCs may promote the adhesion of OvCa cells to the peritoneum, and vitamin D inhibits the adhesion of MCs by inhibiting the expression of THBS1 .
To test this hypothesis, the team further investigated the effect of vitamin D on the adhesion of OvCa cells to MCs.
The experimental results showed that the expression of THBS1 in MCs stimulated by TGF-β1 was significantly up-regulated (79-fold, P<0.0001), while vitamin D supplementation significantly inhibited the expression of THBS1.
In addition, vitamin D inhibited the adhesion and proliferation of OvCa cells in a concentration-dependent manner, and THBS1 enhanced the adhesion of OvCa cells in a concentration-dependent manner.
Next, the research team investigated the molecular mechanism of vitamin D-mediated inhibition of THBS1.
Previous studies have demonstrated that TGF-β signaling is initiated by ligand binding to a complex of TGF-β receptors expressed on the cell surface, followed by phosphorylation to activate Smad2 and Smad3, and activated Smad2/3 to form a complex with Smad4, and then to activate Smad2/3 in a complex with Smad4.
DNA binding acts as a transcription factor to regulate the expression of target genes. Vitamin D can inhibit the phosphorylation of Smad3 through VDR.
This study shows that VDR competes with Smad3 for binding to the promoter region of THBS1 and inhibits the transcription of THBS1.
Therefore, vitamin D inhibited the expression of THBS1 in MCs by inhibiting the binding of Smad3 to DNA, thereby inhibiting the adhesion of OvCa cells to MCs .
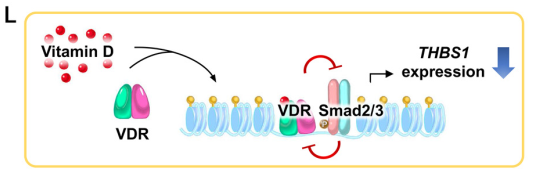
Vitamin D inhibits OvCa cell adhesion and proliferation through VDR/Smad3 competitive inhibition of THBS1
The research team also found that after vitamin D treatment, MCs returned to an epithelioid state, with increased levels of E-cadherin and decreased expression of actin stress fibers and THBS1 in MCs.
In the mesenchymal state, MET cannot be induced to an epithelioid state spontaneously or independently, and the experimental results suggest that vitamin D can partially reverse the mesenchymal state of stable MCs .
Even in the presence of tumor cells, vitamin D induced MET in MCs in the mesenchymal state and normalized THBS1 expression .
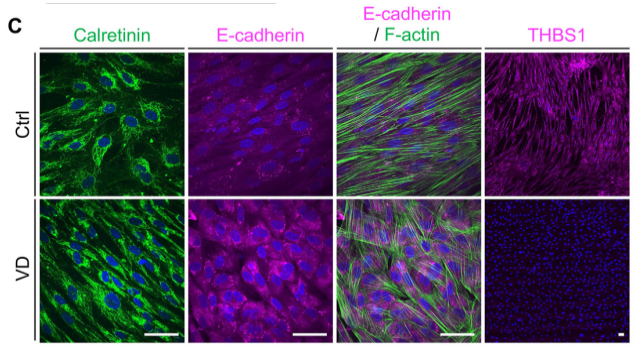
Vitamin D restores TGF-β1-induced EMT and THBS1 expression in MCs
Finally, the research team validated these results in a mouse model.
In vivo experiments showed that the expression level of THBS1 on the peritoneal surface of mice treated with vitamin D, the area of defective microvilli, and the peritoneal dissemination rate of OvCa cells were lower than those of mice treated with TGF-β1 alone.
Western blot and immunofluorescence analysis showed that THBS1 expression on the peritoneal surface was restored following MET induction after vitamin D treatment.
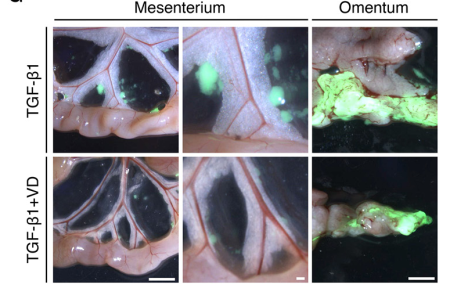
Vitamin D protects peritoneal microvilli in vivo and reduces peritoneal dissemination
In conclusion, this study highlights that MCs are a key determinant of OvCa peritoneal metastasis, finds that vitamin D suppresses tumorigenesis by inhibiting the mesenchymal state of MCs induced by TGF-β1, and establishes that THBS1 is critical for enhancing OvCa cell adhesion and proliferation protein .
In vivo, vitamin D inhibits peritoneal dissemination by inhibiting Smad-dependent TGF-β signaling through VDR/Smad3 competition.
References:
[1] Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021 [published correction appears in CA Cancer J Clin. 2021 Jul;71(4):359]. CA Cancer J Clin. 2021;71(1):7-33. doi:10.3322/caac.21654
[2] Lengyel E. Ovarian cancer development and metastasis. Am J Pathol. 2010;177(3):1053-1064. doi:10.2353/ajpath.2010.100105
[3] Oza AM, Cook AD, Pfisterer J, et al. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): overall survival results of a phase 3 randomised trial. Lancet Oncol. 2015;16(8):928-936. doi:10.1016/S1470-2045(15)00086-8
[4] González-Martín A, Pothuri B, Vergote I, et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N Engl J Med. 2019;381(25):2391-2402. doi:10.1056/NEJMoa1910962
[5] Kitami K, Yoshihara M, Tamauchi S, et al. Peritoneal restoration by repurposing vitamin D inhibits ovarian cancer dissemination via blockade of the TGF-β1/thrombospondin-1 axis. Matrix Biol. 2022;109:70-90. doi:10.1016/j.matbio.2022.03.003
[6] Mutsaers SE. The mesothelial cell. Int J Biochem Cell Biol. 2004;36(1):9-16. doi:10.1016/s1357-2725(03)00242-5
[7] Kenny HA, Chiang CY, White EA, et al. Mesothelial cells promote early ovarian cancer metastasis through fibronectin secretion. J Clin Invest. 2014;124(10):4614-4628. doi:10.1172/JCI74778
[8] Feldman D, Krishnan AV, Swami S, Giovannucci E, Feldman BJ. The role of vitamin D in reducing cancer risk and progression. Nat Rev Cancer. 2014;14(5):342-357. doi:10.1038/nrc3691
[9] Ferrer-Mayorga G, Gómez-López G, Barbáchano A, et al. Vitamin D receptor expression and associated gene signature in tumour stromal fibroblasts predict clinical outcome in colorectal cancer. Gut. 2017;66(8):1449-1462. doi:10.1136/gutjnl-2015-310977
The mechanism by which vitamin D inhibits the spread of ovarian cancer
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



