This therapy dramatically reduces breast cancer recurrence and death
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
100,000 women participated in the trial: This therapy dramatically reduces breast cancer recurrence and death
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
100,000 women participated in the trial: This therapy dramatically reduces breast cancer recurrence and death.
Introduction: Chemotherapy reduces recurrence and mortality in patients with early operable breast cancer.
Compared with patients without chemotherapy, anthracyclines plus taxanes reduce mortality by approximately 40% within the first 10 years after diagnosis, regardless of patient age, tumor characteristics (including tumor size, grade, lymph node involvement) , hormone receptor status, HER2 expression, etc.).
Recently, “The Lancet” published an article entitled “Anthracycline-containing and taxane-containing chemotherapy for early-stage operable breast cancer: a patient-level meta-analysis of 100 000 women from 86 randomized trials” A meta-analysis aimed at a more comprehensive understanding of the benefits and risks of different anthracycline and taxane chemotherapy regimens for patients with early breast cancer.
The results show that for women with early breast cancer who are at high risk of recurrence and who can receive chemotherapy, the benefit of an anthracycline plus taxane regimen is superior to that of other monotherapy.
 https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(23)00285-4/fulltext
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(23)00285-4/fulltext
Research Background
Cytotoxic chemotherapy for operable early-stage breast cancer substantially reduces the risk of recurrence and death. Cyclophosphamide plus methotrexate plus fluorouracil or four cycles of anthracycline plus cyclophosphamide reduced breast cancer mortality by 2.5% compared with no chemotherapy, meta-analysis from the Early Breast Cancer Trials Collaborative Group (EBCTCG) showed .
The ratio is reduced by 20-25%. Adding a taxane (docetaxel or paclitaxel) to anthracycline-containing chemotherapy, or substantially increasing the cumulative dose of anthracyclines, further reduced breast cancer mortality by 10–15% . Additional benefits can be gained by increasing the dose intensity of chemotherapy.
The results of these meta-analyses suggest that compared with no chemotherapy, anthracycline plus taxane chemotherapy reduces breast cancer mortality by approximately 40% within the first decade after diagnosis , regardless of patient age or tumor characteristics ( Including size, grade, lymph node involvement, hormone receptor status and HER2 expression), there was a similar reduction in proportion.
However, the optimal chemotherapy regimen is uncertain. Anthracyclines increase the long-term risk of cardiovascular disease and acute myeloid leukemia, and taxanes cause dose-dependent peripheral neuropathy.
Therefore, the risks of anthracycline plus taxane chemotherapy may outweigh the benefits, especially for women with low-risk tumors receiving optimal surgery or radiotherapy (and endocrine or anti-HER2 therapy as appropriate).
Research results
The researchers identified 28 taxane trial protocols with or without anthracyclines, of which 23 were considered eligible and 15 provided data on 18,103 women.
Across all 15 trials for which individual data were available, there was an average 14% reduction in relapse compared with a taxane-free anthracycline regimen (RR 0·86, 95% CI 0·79–0·93; p=0·0004).
There was no increase in non-breast cancer deaths, but there was 1 additional case of acute myeloid leukemia for every 700 women treated.
Compared with docetaxel plus cyclophosphamide at the same dose, the reduction in relapse rate was most pronounced when docetaxel plus cyclophosphamide was added to an anthracycline (10-year risk of relapse 12.3% vs 21. 0%; risk difference 8·7% , 95% CI 4·5–12·9; RR 0·58, 0·47–0·73; p<0·0001). The 10-year breast cancer mortality rate in this group was reduced by 4·2% (0·4–8·1; p=0·0034).
Sequential taxane plus anthracycline regimens did not significantly reduce the risk of relapse compared with docetaxel plus cyclophosphamide (RR 0·94, 0·83–1·06; p=0·30).
Thirty-five trials (n=52 976) provided individual patient data for the analysis of taxane-containing and taxane-free anthracycline regimens.
When the cumulative dose of anthracyclines in each group was the same (RR 0·87, 0·82–0·93; p<0·0001; n=11167), compared with the control group non-taxanes (main are anthracyclines) the cumulative dose was two times higher than the taxane group (RR 0·96, 0·90–1·03; p=0·27; n=14 620).
A direct comparison between anthracycline and taxane regimens showed that regimens with higher cumulative doses and higher dose intensities were more effective.
The proportional reduction in relapse rate with taxane plus anthracycline was similar in estrogen receptor-positive and estrogen receptor-negative disease and did not differ by age, nodal status, tumor size, or grade.
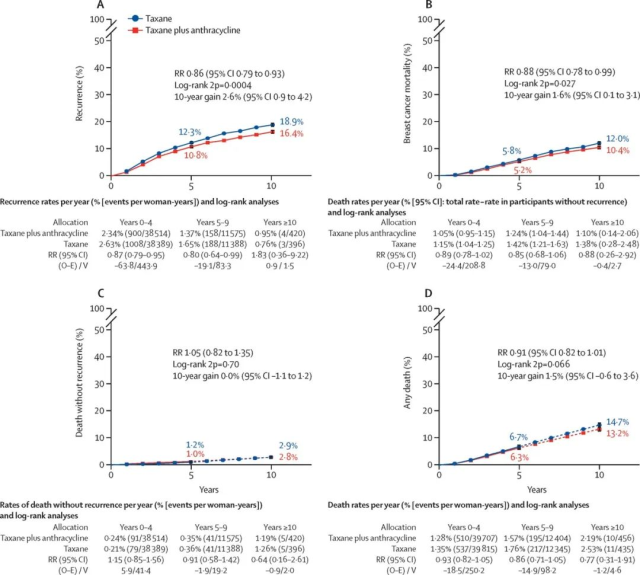 10-Year Cumulative Risk Analysis of Taxanes Plus Anthracyclines vs. Taxanes Without Anthracyclines
10-Year Cumulative Risk Analysis of Taxanes Plus Anthracyclines vs. Taxanes Without Anthracyclines
Significance
This meta-analysis suggests that adding anthracyclines to a taxane-based regimen yields greater benefit than a taxane-free regimen. Anthracycline and taxane regimens with higher cumulative doses conferred the greatest benefit.
This informs patient treatment decisions, clinical guidelines, and the design of future clinical trials.
Considering that the absolute benefit that patients get from chemotherapy increases with the increase of recurrence risk, it is recommended that when deciding to take endocrine therapy or use anti-HER2 positive drugs, the risk of recurrence should be fully scored and taken as a key consideration for chemotherapy.
References:
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(23)00285-4/fulltext
100,000 women participated in the trial: This therapy dramatically reduces breast cancer recurrence and death.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



