Celarevir potassium with sofosbuvir treats chronic hepatitis C infection
- Why Lecanemab’s Adoption Faces an Uphill Battle in US?
- Yogurt and High LDL Cholesterol: Can You Still Enjoy It?
- WHO Releases Global Influenza Vaccine Market Study in 2024
- HIV Infections Linked to Unlicensed Spa’s Vampire Facial Treatments
- A Single US$2.15-Million Injection to Block 90% of Cancer Cell Formation
- WIV: Prevention of New Disease X and Investigation of the Origin of COVID-19
Celarevir potassium with sofosbuvir treats chronic hepatitis C infection
Celarevir potassium with sofosbuvir treats chronic hepatitis C infection. J Gastroenterol Hepatol: The effectiveness and safety of celarevir potassium tablets combined with sofosbuvir tablets in the treatment of chronic hepatitis C virus infection.
Most of the adverse events that occurred in the study were mild or moderate, and they were cured or relieved without clinical intervention.

This study is a multi-center, single-arm, open phase III clinical trial. To evaluate the efficacy and safety of HCV nonstructural protein 3/4A inhibitor celarevir potassium tablets combined with sofosbuvir tablets in Chinese patients with chronic hepatitis C and non-cirrhosis.
Cerarevir potassium tablets, 100 mg, 2 times a day; Sofosbuvir, 400 mg, once a day, combined administration for 12 or 24 weeks for the treatment of chronic Chinese patients with non-cirrhosis, initial treatment or interferon treatment Patients with hepatitis C. The primary endpoint is the sustained virological response (SVR12) rate at 12 weeks after discontinuation.
The researchers successfully enrolled 205 subjects in 23 clinical research centers across the country, of which 202 subjects completed the study and 3 subjects withdrew early.
Of the 205 subjects, 27 (13.2%) were interferon-treated patients, and all subjects had HCV genotype 1.
The sustained virological response rate at 12 weeks after discontinuation was 98% (201/205) (95% confidence interval: 95.1%~99.5%), of which the HCV gene 1a subgroup was 100%, and the gene 1b subgroup It is 98.0%.
In another exploratory study, 21 subjects with HCV genotype 2, 7 subjects with genotype 3, and 8 subjects with genotype 6 were enrolled. The continuous virological response at 12 weeks after drug withdrawal The rate is 100%.
Most of the adverse events that occurred in the study were mild or moderate, and they were cured or relieved without clinical intervention.
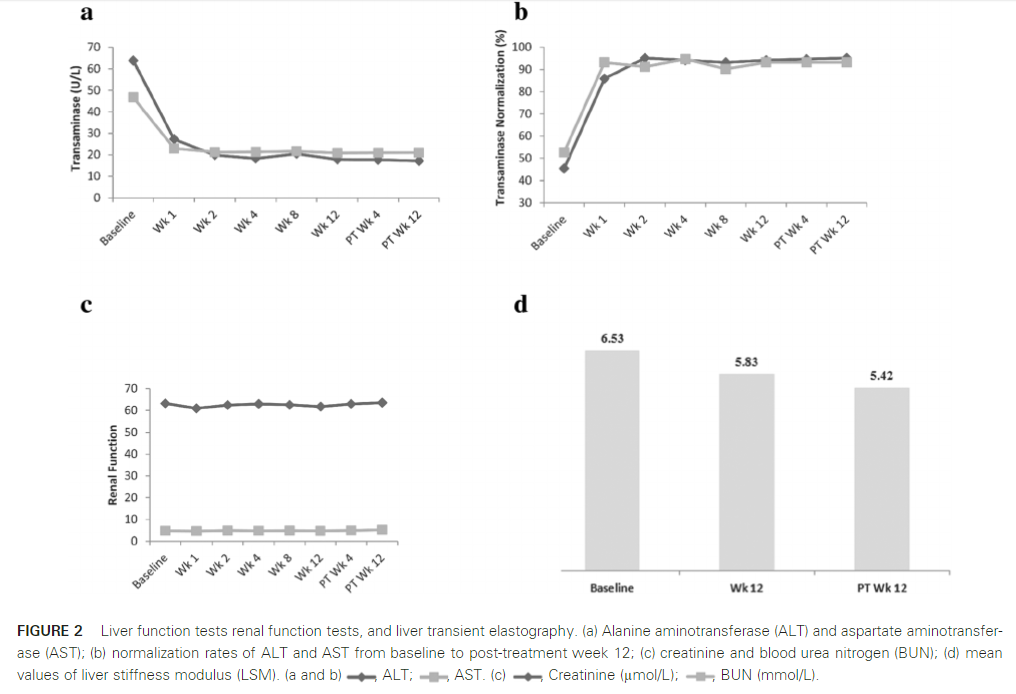
Studies have shown that celarevir potassium tablets combined with sofosbuvir tablets is a full oral treatment without ribavirin, which is good for patients with chronic hepatitis C who are not cirrhotic, newly treated or treated with interferon Efficacy and safety.
(source:internet, reference only)
Disclaimer of medicaltrend.org



