Chinese COVID-19 drugs are expected to be launched soon
- Paternal Microbiome Perturbations Impact Offspring Fitness
- New Report Casts Doubt on Maradona’s Cause of Death and Rocks Manslaughter Case
- Chinese academician unable to provide the exact source of liver transplants
- Early Biomarker for Multiple Sclerosis Development Identified Years in Advance
- Aspirin Found Ineffective in Improving Recurrence Risk or Survival Rate of Breast Cancer Patients
- Child Products from Aliexpess and Temu Contain Carcinogens 3026x Over Limit
Chinese COVID-19 drugs are expected to be launched soon.
- AstraZeneca Admits for the First Time that its COVID Vaccine Has Blood Clot Side Effects
- Was COVID virus leaked from the Chinese WIV lab?
- HIV Cure Research: New Study Links Viral DNA Levels to Spontaneous Control
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Chinese COVID-19 drugs are expected to be launched soon.
The latest news shows that China’s COVID-19 specific drugs finally have a “timetable”, and one of the antibody combination drugs is expected to be approved for conditional listing before the end of the year at the earliest.
According to statistics, there are currently six specific drugs for COVID-19 developed in China. Among them, two effective neutralizing antibody drugs BRII-196 and BRII-198 jointly developed and screened by Tsinghua University, Shenzhen Third People’s Hospital and Tengsheng Huachuang have participated in the treatment of more than 700 patients in China.
The R&D team has submitted the conditional listing application materials to the State Food and Drug Administration on October 9, and is expected to be approved for conditional listing before the end of December.

Professor Zhang Linqi of Tsinghua University: Hundreds of antibodies isolated from the blood of convalescent patients with COVID-19 have screened two antibodies with the highest activity and strong complementarity. In clinical trials conducted at home and abroad, they have shown that they can reduce severe illness and mortality by 78 % Excellent results.
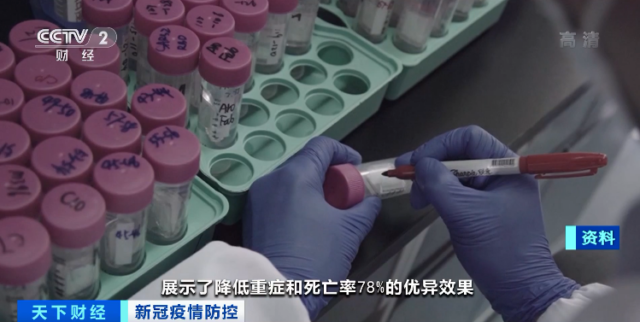
At the same time, the neutralizing antibody DXP604, jointly developed by Peking University’s Xie Xiaoliang team and Danxu Biotechnology, was used as a “sympathetic drug” in Beijing Ditan Hospital when no other drugs were available. After the drug, some patients have recovered and discharged.
In addition, the new generation of androgen receptor antagonist “Prokalutamide” under development by the pioneering pharmaceutical industry has been authorized for emergency use in Paraguay. Three clinical trials conducted in Brazil initially showed that “procrudamide” can Reduce the risk of death for severely ill patients by 78%.
According to the relevant person in charge, the current research and development of effective drugs for the new coronavirus is mainly focused on three technical routes of blocking the virus from entering cells, inhibiting virus replication, and regulating the human immune system. China has already deployed these technical routes.
Chinese COVID-19 drugs are expected to be launched soon.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



