Cryo-EM Structures Reveal the Molecular Basis of Receptor-Initiated Coxsackievirus Uncoating
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Cryo-EM Structures Reveal the Molecular Basis of Receptor-Initiated Coxsackievirus Uncoating
Cryo-EM Structures Reveal the Molecular Basis ofReceptor-Initiated Coxsackievirus Uncoating. Recently, the team of Professor Xia Ningshao of Xiamen University published an online study titled “Cryo-EM Structures Reveal the Molecular Basis of Receptor-Initiated Coxsackievirus Uncoating” in the sub-Journal of “Cell” (Cell Host & Microbe) paper.
This study clarified the interaction between Coxsackievirus B (CVB) and its highly dependent functional receptor Coxsackievirus and Adenovirus Receptor (CAR) and the fine features of the allosteric process At the molecular level, it revealed the mechanism and key sites of CAR-mediated high-efficiency decapsidation of CVB virus, and guided the discovery of a unique high-efficiency therapeutic antibody that can mimic the receptor action characteristics and induce the instability and disintegration of mature CVB virus particles .
This is what Professor Xia Ningshao’s team has done in recent years in “Nature Communications” (Nature Communications, 2017), “Science Advances” (Science Advances, 2018), “Nature Microbiology” (Nature Microbiology, 2019) and “Cell• Another important research result after the publication of research papers on important enterovirus pathogens CVA6, CVA10, EVD68 and CVA16 on Cell Host & Microbe (2020), can provide a key basis for the research of CVB vaccines and antiviral drugs. It enriches the understanding of the interaction mechanism between the virus and the receptor, and provides an important reference and theoretical basis for the development of high-efficiency broad-spectrum antiviral drugs targeting the binding domain of the virus receptor.
Coxsackie group B virus (CVB) is an enterovirus pathogen that can cause a variety of serious diseases. It can be transmitted through feces or mouth. It is highly contagious and can infect infants, adolescents, and adults. It is widespread worldwide. There are 6 serotypes of CVB (CVB1~6), which can cause important diseases, including fever, hand, foot and mouth disease, diarrhea, encephalitis, meningitis, myocarditis, pancreatitis, acute flaccid paralysis, and even death.
Studies have found that CVB infection is closely related to viral pancreatitis and viral myocarditis that have been increasing in incidence in recent years. It is one of the main causes of type I diabetes and adolescent cardiogenic death, and it has serious harm. The importance and public health significance of CVB prevention and treatment has been highly valued, but there are currently no specific treatment drugs and preventive vaccines for CVB. Related drugs are an important direction of current anti-enteric virus research.
However, there is still a lack of clear understanding of the basic virology of CVB, especially the potential high-efficiency antiviral targets and the mechanism of effector molecules, which severely restricts the effective development of related research.
CAR is a functional cell receptor that is highly dependent on CVB infection, and is also known as uncoating receptor. CAR binding can induce the irreversible conformation of CVB mature virus particles and promote the release of nucleic acid, which plays a key role in CVB infection. The CVB-CAR binding domain is an ideal target for discovering broad-spectrum antiviral targets.
However, there is still a lack of clear understanding of the precise characteristics and detailed mechanism of the interaction between CVB and CAR, and the precise information and targeted neutralizing antibodies for the potential and highly effective antiviral epitopes of the CAR binding domain of CVB have not been discovered and accurately identified. It is a core scientific problem that urgently needs to be solved in the development of new vaccines or antiviral drugs.
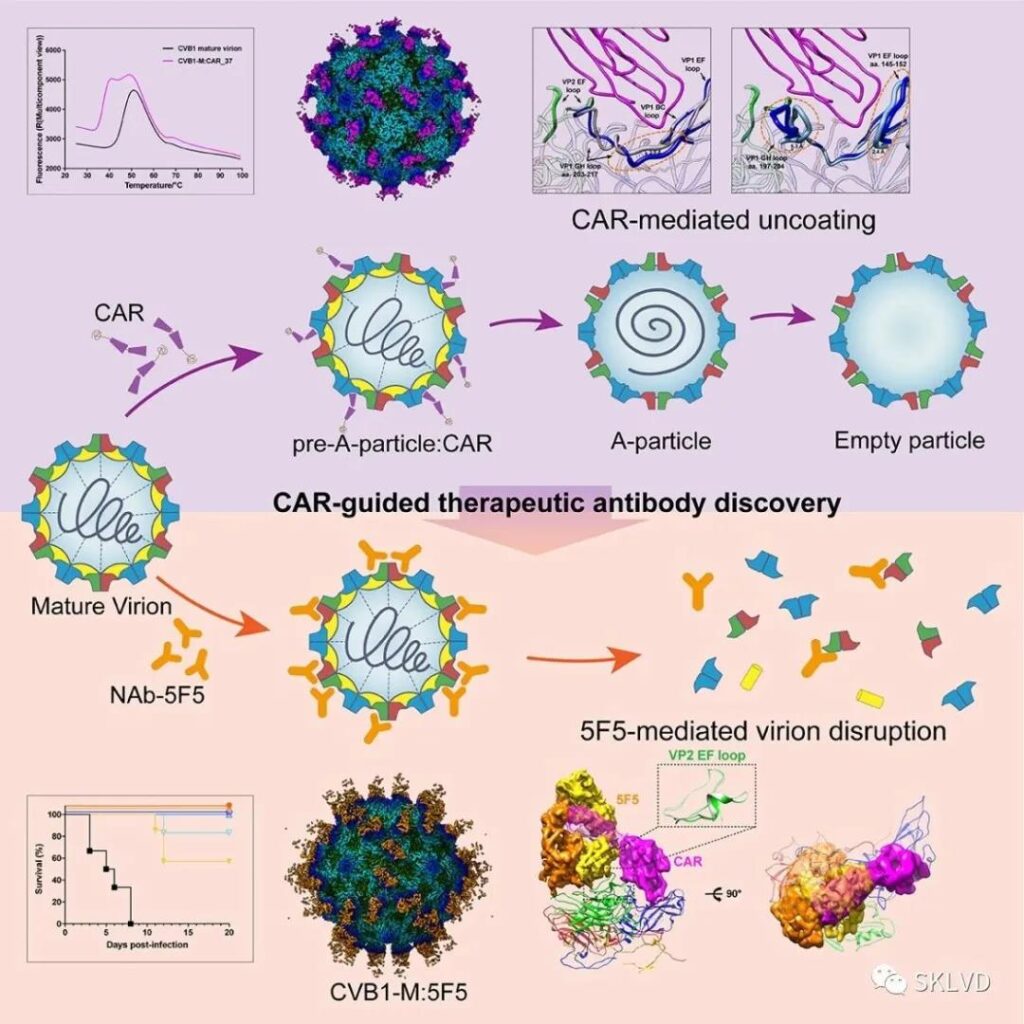
In this study, through the construction and analysis of the high-resolution structure of CVB virus particles and the CAR receptor complex, a series of near-atomic resolution structures representing different stages of virus infection under neutral pH conditions were obtained, including CVB1 mature particles (Mature virion), CAR-combined CVB1 precursor decapsid intermediate state (Pre-A-particle), decapsid intermediate state (A-particle) and hollow particle (Emptyparticle) combined with CAR at low temperature and physiological temperature, and capture the decapsidation of CVB mature virus particles Many different intermediate states of the process and a series of fine conformational changes in the CAR receptor interaction domain.
Studies have shown that CAR binds to the “canyon” area around the five-fold axis formed by the capsid protein VP1 on the surface of the virus. Compared with other known enterovirus uncapsid receptors, CAR binds deeper into the canyon groove. In part, the VP1 hydrophobic “pocket” inside the valley can be induced to collapse under physiologically neutral pH conditions, so that the lipid molecules (also known as “pocket factor”) that maintain the stability of enterovirus particles inside the hydrophobic pocket ) Release, thereby initiating the decapsidation process of mature virus particles, promoting the further transformation of the virus into the intermediate decapsidization state and hollow particles, and completing the complete decapsidation and genome release process of the virus.
The study explained the molecular mechanism of CAR binding driving CVB infection, revealed the precise interaction mode, structural domain characteristics and key amino acid sites between CVB and CAR, and found that the receptor binding domain can be used as a potential high-efficiency antiviral target Domains and key sites. Based on the guidelines of the above findings, this study screened a unique and highly effective neutralizing antibody 5F5 that can mimic the receptor action characteristics and induce the instability and disintegration of CVB mature virus particles.
Studies have found that 5F5 can block or compete for the interaction between CAR and CVB through steric hindrance. It can not only simulate the interaction between the receptor and virus particles, but also induce the key target VP2 EF-loop of the receptor CAR binding domain. The conformational change of the virus destroys the stability of the virus capsid, and finally leads to the lysis of mature virus particles to exert an efficient neutralization effect.
This research enriches the basic theories of enterovirus infection and the interaction mechanism with receptors, discovers precise information on key antiviral targets and effector antibody molecules of receptor binding domains, and develops rational design of new vaccine immunogens and antiviral drugs And application development has important value and significance.
(source:internet, reference only)
Disclaimer of medicaltrend.org



