Israel’s magical drugs can “cure” COVID-19 pneumonia in five days?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Israel’s magical drugs can “cure” COVID-19 pneumonia in five days?
Israel’s magical drugs can “cure” COVID-19 pneumonia in five days? “Israel Embassy in China” announced that Israel has discovered a miraculous drug that can cure the COVID-19 pneumonia, with a cure rate of over 95%!

Recently, the headlines and Weibo accounts of the “Israel Embassy in China” announced that Israel has discovered a miraculous drug that can cure the COVID-19 pneumonia, with a cure rate of over 95%!
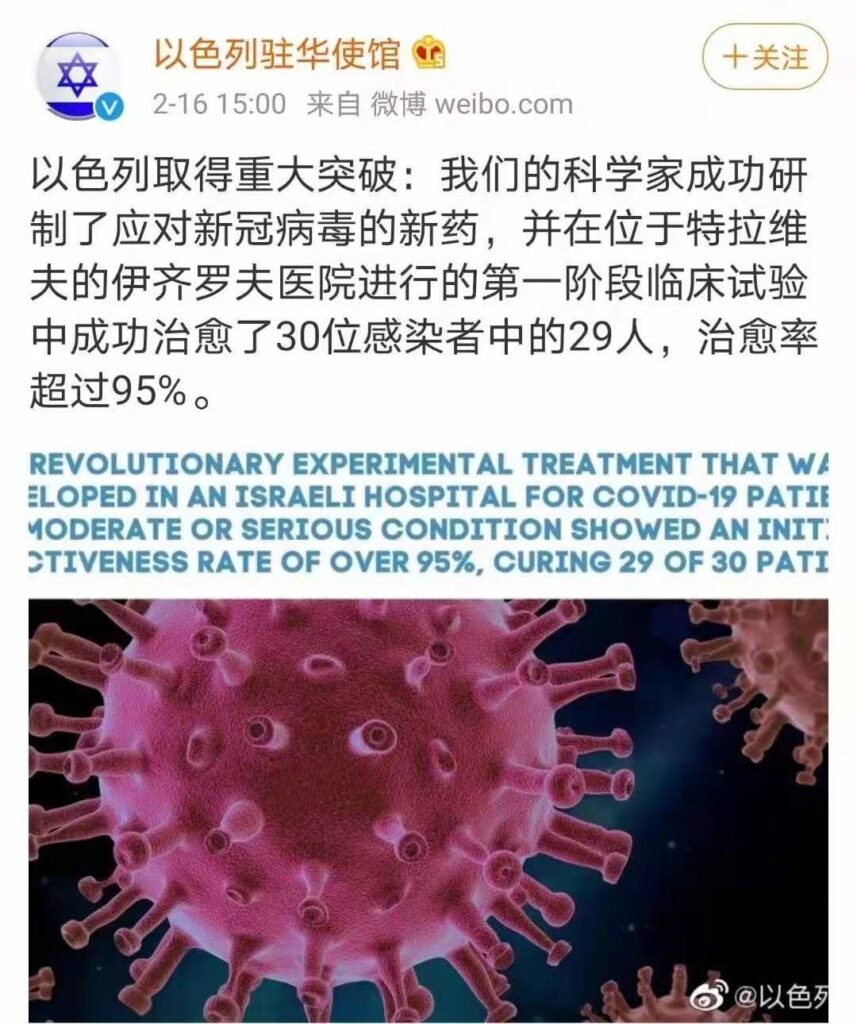
The original text reads as follows: “Israel has made a major breakthrough: Our scientists have successfully developed a new drug against the new coronavirus and successfully cured 29 of the 30 infected people in the first phase of the clinical trial conducted at the Izirov Hospital in Tel Aviv. , The cure rate exceeds 95%.”
According to a report by the internationally renowned technology media BGR, the new drug is called EXO-C24. It will not eliminate the virus, but is an immunomodulatory agent. The human immune system can eliminate the virus, and many people can recover from COVID-19 even without treatment. However, the most terrifying danger of the new coronavirus is that it may activate the immune system overreaction in the body, and cause disease deterioration in high-risk groups, that is, cytokine storm, causing multiple organ failure and death. EXO-C24 can regulate the cytokine storm to prevent the immune system from overreacting.
According to Professor Nadir Arber, head of the drug laboratory of Izirov Hospital in Tel Aviv, EXO-C24 uses a tiny carrier to deliver CD24 protein to the lungs. This is an inhaled preparation, once a day, each time lasts about a few minutes, a total of five days of treatment. This preparation can directly protect the heart and lungs that are most affected by the cytokine storm. Different from other preparations, it is a preparation for topical administration with a wide range of effects and no side effects.
Although there are currently many new coronavirus vaccines on the market, they cannot treat infected patients, nor can they ensure that the vaccine can effectively deal with all the mutations that have not yet appeared. Therefore, the invention of a drug for the treatment of new coronavirus pneumonia will greatly help fight the development of the disease. The drug will enter the next phase of research to obtain more efficacy and safety data.
The media pointed out in reports that there are still too many unknowns about the future. It is too early to call it a “miracle therapy”. If the test results can be verified in subsequent studies, it may become a breakthrough therapy. However, the hospital officials have called it a “game changer” against the COVID-19 virus.
Is it really so amazing?
Zhang Hongtao, an associate professor of research in the Department of Pathology and Experimental Medicine at the University of Pennsylvania School of Medicine (pennamed “a section of ginger”), commented: People with a discerning eye will know at a glance that this has only completed a phase 1 clinical trial. In the past year, there have been too many COVID-19 drugs that have completed phase 1 clinical trials. If they are all expected to be specific drugs, then there will be great hopes and disappointments. I have not completed the Phase 3 clinical trial, and dare not say “successfully developed a new drug to deal with the new coronavirus”, at most I can only say that the Phase 1 clinical trial has been successfully completed.
In the phase 1 clinical trial, 30 patients with moderate and severe new coronavirus pneumonia were treated with the drug inhaled once a day for five minutes each time. Twenty-nine people recovered within 3 to 5 days after treatment, that is, “the cure rate exceeds 95%”. It can be seen from this phase 1 trial that there is no control group in the trial, so it is impossible to judge the effectiveness of the trial drug at all. However, if patients with moderate to severe new coronavirus pneumonia can be cured 95% within 3-5 days, the effect is still good.
Is it really possible to do this? Only wait for follow-up phase 3 clinical trials to confirm. So, what can you do if the Phase 1 clinical trial is successful? You can continue to experiment. As for “no side effects”, it is impossible to tell from 30 people in the phase 1 trial. At most, they can only say “no obvious side effects”. This is like saying “no side effects” after the COVID-19 vaccine has done a phase 1 trial. If you still believe it, then 2020 is over.
Searching the Internet for the news of “Israel + Cure New coronavirus Pneumonia”, I found that starting from April 2020, Israel has invented the miraculous medicine to cure the new coronavirus pneumonia. What is the result now? This magic drug only cured 29 patients in the Phase 1 clinical trial. You need to observe it for a few months and wait until the Phase 3 trial.
(source:internet, reference only)
Disclaimer of medicaltrend.org



