Auxiliary drugs to reduce the dosage of neuromuscular blockers
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Auxiliary drugs to reduce the dosage of neuromuscular blockers
Auxiliary drugs to reduce the dosage of neuromuscular blockers. Magnesium, dexmedetomidine, and clonidine can reduce the dose of NMBA required to maintain satisfactory neuromuscular block without affecting patient safety and recovery.
As the number of surgical operations increases year by year, there is an increasing shortage of intensive care and perioperative drugs worldwide. Insufficient supply of medicines not only imposes an additional cost burden on the health system, and may increase the occurrence of adverse events during the perioperative period.
Coupled with the 2019 COVID-19 pandemic, unprecedented manufacturing pressure has affected the global supply of medicines, including neuromuscular blockers (NMBA). Therefore, this study conducted a systematic review and meta-analysis of published literature to explore whether adjuvant drugs can effectively reduce the amount of NMBA in perioperative and critically ill patients. The results of the study will be published in the British Journal of Anaesthesia in March 2021.

Methods:
The researchers searched the Medline, Embase, Web of Science and Cochrane databases from January 1, 1970 to August 15, 2020. The main research goal is the total dosage of NMBA to achieve clinically satisfactory neuromuscular block depth. The secondary research objectives were adverse events, induction time, extubation time, and neuromuscular block residuals after the use of adjuvant drugs.
Results:
A total of 31 randomized controlled trials were included in this study (1962 patients). A total of 1085 patients received adjuvant drug treatment, while 877 patients received other drugs or saline as controls. Seven different adjuvant drugs in common use include magnesium sulfate, dexmedetomidine, clonidine, nicardipine, diltiazem, lidocaine, and dexamethasone.
Main research results:
① Magnesium is the most commonly used auxiliary drug.
Compared with the normal saline control group, 20 of the 21 studies reported a reduction in the NMBA dose after the use of magnesium (Figure 1) (SMD: -1.10 [-1.44~-0.76]; P<0.001; I2=85%).
② Dexmedetomidine:
Four of the five randomized controlled trials reported that a loading dose of 1ug/kg of dexmedetomidine and a maintenance dose of more than 0.2ug/kg/h can reduce the amount of NMBA. For dexmedetomidine (Figure 2), the dose of each NMBA was reduced (SMD: -0.89 [-1.55~-0.22]; P=0.009; I2=87%).
③Clonidine:
Clonidine (2-3 ug/kg) was given preoperatively in all 3 trials, and clonidine (1-2 ug/kg/h) was given intraoperatively in 2 trials. For clonidine (Figure 3), the dose of each NMBA was reduced (SMD: -0.67 [-1.13~-0.34]; P=0.004; I2=0%).
④ Lidocaine:
In the 3 randomized controlled trials included in the group, lidocaine was injected intravenously before operation (1.5-2 mg/kg) and during operation (1.5-2 mg/kg/h). Compared with the control group, lidocaine (Figure 3) had no effect on the total perioperative NMBA dosage (SMD: -0.46 [-1.01~-0.09]; P=0.10; I2=68%).
Due to the small number of studies, meta-analysis of nicardipine, diltiazem, and dexamethasone cannot be performed.
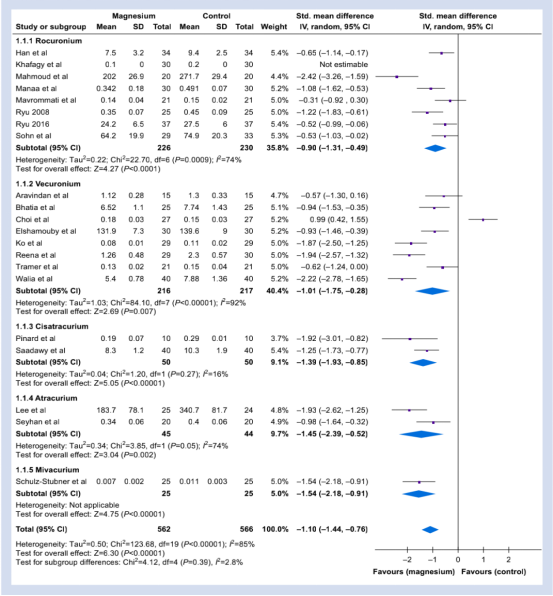
Figure 1: The NMBA dose required for the perioperative period of magnesium in each subgroup and the control group.
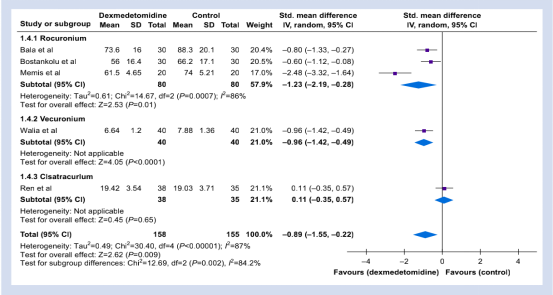
Figure 2: Dexmedetomidine in each subgroup and the control group required NMBA dose during the perioperative period.
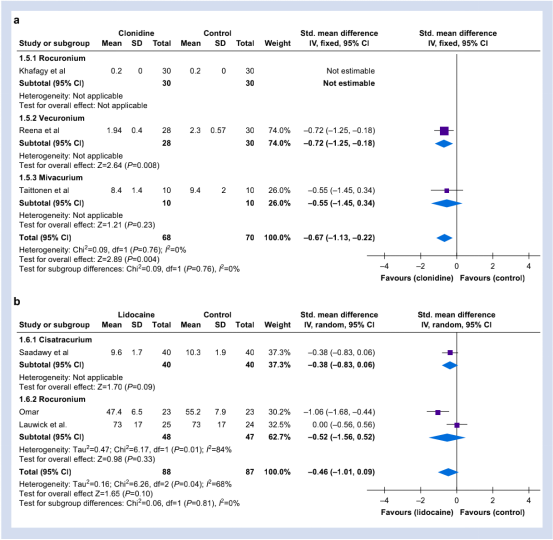
Figure 3: The NMBA dose required during the perioperative period in each subgroup vs. the control group. a: clonidine; b: lidocaine.
Secondary research results:
①Hypotension:
Compared with the normal saline group, the risk of hypotension in patients with or without magnesium is similar (n=9) (RR=1.00 [0.46, 2.21], P=0.99, I2=0%); Youmei Detomidine had the same outcome (n=3) (RR=0.71[0.30, 1.64], P=0.42, I2=0%).
② Bradycardia:
A total of 9 studies reported the occurrence of bradycardia, and meta-analysis showed that there was no difference in the risk of bradycardia with magnesium (RR=2.17 [0.63, 7.50], P=0.22, I2=0%).
③Induction time:
One study reported data on induction time. The average induction time of magnesium group (67.1 [9.3]s) and clonidine group (59.8[6.3]s) was significantly shorter than that of normal saline group (88.1[10.2]s; P<0.05).
④Extubation time:
Twelve studies (533 patients) reported data on time to extubation. The meta-analysis showed that there was no difference in the extubation time of the magnesium group (n=9) or the clonidine group (n=2). Dexmedetomidine (7.7 [3.1] min, vs saline group: 10.8[3.8] min; P=0.01) and lidocaine (7.2[3.6] min vs control group: 11[5.8] min; P= After 0.01), the extubation time was reduced.
⑤ Residual neuromuscular block:
Two studies reported data on TOF ratio when entering PACU. For magnesium, the TOF ratio of the intervention group and the saline group were both greater than 0.9, and lidocaine was associated with a higher TOF ratio.
Conclusion:
Magnesium, dexmedetomidine, and clonidine can reduce the dose of NMBA required to maintain satisfactory neuromuscular block without affecting patient safety and recovery.
Expert Comments:
A systematic review and Meta analysis showed that magnesium, dexmedetomidine, and clonidine can reduce the dose requirement of non-depolarized NMBA during the perioperative period. However, there are references to α2-adrenergic receptor agonists, and muscle relaxants are not the main content, and muscle relaxant monitoring is not used for objective monitoring, so the quality of the evidence is low. The evidence for the effect of lidocaine, nicardipine, diltiazem, or dexamethasone as adjuvants to reduce the dose of non-depolarized NMBA during the perioperative period is not clear.
Due to the difference in the results, we can only estimate the relative reduction of the combined administration of magnesium, dexamethasone or clonidine. Magnesium can reduce the use of NMBA by about 25%. After the publication of this article, this judgment was also supported by a randomized controlled trial. The trial reported that patients who received 50 mg/kg magnesium randomly, followed by continuous infusion at a rate of 15 mg/kg/h during the operation, the intraoperative Roku The demand for ammonium bromide has been reduced by 20%. There was no difference in recovery time and the incidence of nausea and vomiting.
Another article shows that for patients with myasthenia gravis, combined use of magnesium can reduce the amount of NMBA by 60%. But in critical care medicine, its 25% reduction may not be clinically meaningful. Magnesium is widely used clinically as a first-line medication for pregnancy-induced hypertension and an arrhythmia auxiliary medication, and its side effects are also well known to clinicians. However, there are few reports of adverse events in this systematic review, and the incidence of side effects within the therapeutic dose is not higher than that of the control group.
This article does not mention inhaled anesthetics, which may reduce the sensitivity of the membrane behind the neuromuscular junction to depolarization. By changing the Na-K channel to increase the endplate potential to generate an action potential threshold, it enhances the activity of inhibitory postsynaptic combination. And inhibit the activity of excitatory synaptic association, thereby enhancing muscle relaxation.
There are many experimental data. Compared with propofol, it can even reduce the amount of NMBA by 40%. It is also the most commonly used clinical general anesthetic for synergistic muscle relaxation.
(source:internet, reference only)
Disclaimer of medicaltrend.org



