Nature: New cancer vaccine improve immunotherapy by nearly 3 times!
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Nature: Improve the effect of immunotherapy by nearly 3 times! This anti-cancer vaccine has entered phase 1 clinical
Nature: New cancer vaccine improve immunotherapy by nearly 3 times! Immunotherapy is currently one of the most successful therapies in the field of cancer treatment, and clinical research and application of immune checkpoint inhibitors (Checkpoint inhibitors) are the most widely used.
In layman’s terms, immune checkpoints are the “brake points” of immune cell production, which can regulate the immune response to avoid “injury” to normal tissues. When this effect is used by tumor cells, immune checkpoint inhibitors can relieve this regulatory effect. , To reactivate immune cells to “fight against the enemy.”
Although immune checkpoint inhibitor therapy has already shined in the field of cancers such as melanoma, squamous cell carcinoma, lung cancer, kidney cancer and liver cancer, the problem is that some patients still cannot benefit from this therapy.
Excitingly, immunologists at the University of Konstanz have developed a new type of cancer vaccine that can increase the effectiveness of immune drugs from 20% to 75% in mouse experiments. The relevant report was published in the sub-Journal “Nature Communications” of “Nature” on May 18, local time.
Regarding the reason for the poor effect of immune checkpoint inhibitor therapy in some cancer patients, some people believe that it may be due to the role of immune checkpoint inhibitors being the “icing on the cake”, helping the body to prolong the immune response, and once the immune system Without the ability to distinguish tumor cells, immune checkpoint inhibitors also lose their effectiveness. From this perspective, it is very important to help the immune system see through the “camouflage” of tumor cells.
Dendritic cells (DC cells) are the most powerful antigen-presenting cells (APC), which can efficiently ingest, process, and present antigens, which is also the key to re-awakening the immune system.
Therefore, the researchers designed a new anti-cancer vaccine targeting DC cells with degradable particles. The vaccine has a diameter of 1 micron and uses polylactic acid-glycolic acid copolymer (PLGA) microparticles that have been approved for use in humans. Nanoparticles, encapsulating tumor proteins and GMP-level immune system promoter Riboxxim.
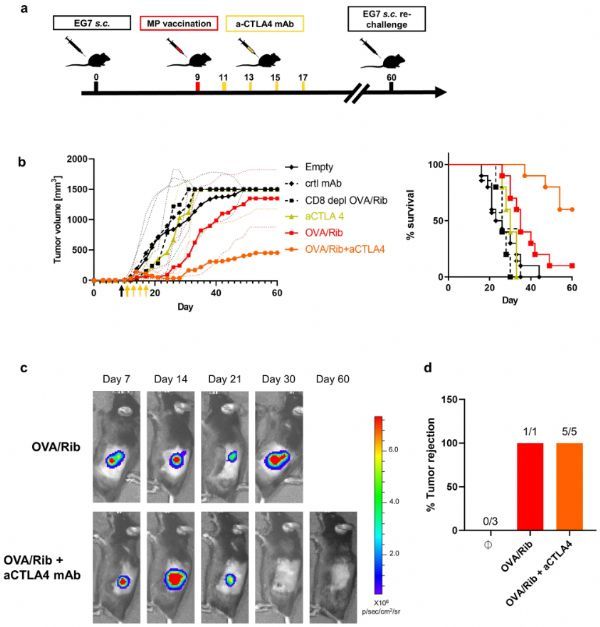
In mice, a single dose of the vaccine can induce DC cell maturation and enhance tumor antigen presentation, and promote the further proliferation of antigen-specific CD8 + T cells, prolonging the immune response to the 8th week. However, despite the strong immune response, it did not trigger a serum cytokine response and histological changes or damage to major organs. At the same time, the mouse endotoxin level was lower than 0.5EU/ml, which further proved the effectiveness and safety of the vaccine. .
Of course, “actual combat” is the most intuitive form of testing the effectiveness of vaccines. Researchers observed the immune response and tumor progression in mice with thymic cancer after being vaccinated. Bioluminescence imaging analysis showed that starting from the 8th day after the immune response reached its maximum, the tumors in the mice began to shrink gradually. In addition, this vaccine can produce a strong cellular immune response to multiple tumor types such as prostate cancer, breast cancer, and melanoma, indicating that the vaccine may be suitable for a variety of cancers.
More importantly, with the natural down-regulation of the human immune response, the tumor suppression effect of the vaccine will gradually weaken, but if the vaccine is used in combination with an immune checkpoint inhibitor such as CTLA-4 monoclonal antibody, it can be restarted. The combination of CD28 molecules and B7 molecules on APC cells activates the immune response and prolongs the duration of the therapeutic effect. After 10 mice received this treatment plan, the large tumor masses in the body were eliminated, and 6 mice had a more significant survival rate and complete remission rate.
Marcus Groettrup, senior author of the report and director of the Department of Immunology at the University of Konstanz, said: “Our research shows that if this clinically applicable cancer vaccine is combined with immune checkpoint inhibitors, it can cure mice with tumors. The proportion increased to 75%, long-term protection from the impact of tumor recurrence, which will have a profound impact on the immunotherapy of certain cancers.”
It is reported that a Dutch company currently cooperating with the research team is carrying out a phase 1 clinical trial for the combination therapy formed by this vaccine and immune checkpoint inhibitors to explore the effects of the therapy in humans.
(source:internet, reference only)
Disclaimer of medicaltrend.org



