Cell Journal: This miRNA can reverse Alzheimer’s memory loss
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Cell Journal: This miRNA can reverse Alzheimer’s memory loss
Cell Journal: This miRNA can reverse Alzheimer’s memory loss. Alzheimer’s disease (AD), commonly known as “Alzheimer’s disease”, is a serious neurodegenerative disease.
Patients usually suffer from memory decline and weakened learning ability, accompanied by emotional regulation. Obstacles and loss of athletic ability greatly affect the development of individuals, families and even society.
Currently, about 50 million people worldwide suffer from Alzheimer’s disease. As the average life expectancy of human beings increases and the aging society intensifies, the prevalence of Alzheimer’s disease is also rising. It is estimated that by 2050, Alzheimer’s patients will increase to more than 150 million.
Neural stem cells located in the hippocampus maintain neurogenesis in the adult brain for life, and adult hippocampal neurogenesis (AHN) is functionally related to memory and cognition. In Alzheimer’s disease (AD), the process of producing new neurons in the hippocampus is hindered.
When considering adult hippocampal neurogenesis (AHN) as a therapeutic target for Alzheimer’s disease, a deeper understanding of the molecular mechanisms involved in adult hippocampal neurogenesis (AHN) becomes particularly important. miR-132 may play a role in adult neurogenesis, but little is known about the role of miR-132 in adult rodents and human hippocampus. The role of miR-132 in adult hippocampal neurogenesis (AHN) in the context of Alzheimer’s disease The role of this is still elusive.
Recently, a research team from the University of Leuven, Belgium, the Netherlands Institute of Neuroscience and other institutions published a research paper titled Restoring miR-132 expression rescues adult hippocampal neurogenesis and memory deficits in Alzheimer’s disease in the Cell Stem Cell journal, a subsidiary of Cell.
The study confirmed that adult hippocampal neurogenesis (AHN) is directly affected by the pathology of Alzheimer’s disease (AD) and revealed the potential therapeutic potential of increasing miR-132 levels in neurodegenerative diseases.

The research team first used two AD models to detect the level of miR-132 in the dentate gyrus (part of the hippocampal structure) in the control group and the disease group, and found that compared with the wild type, the miR-132 in the dentate gyrus of AD had an early downward trend.
BrdU injection experiments showed that the AD group had impaired the potential of inducing the proliferation of neuron progenitor cells when running. The effect of AD on the expression of miR-132 and AHN injury in two different mouse models is emphasized.
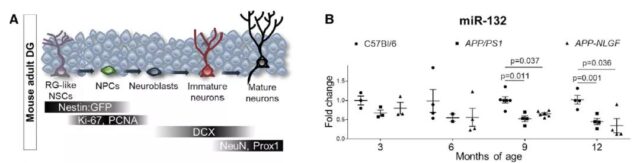
Next, the research team evaluated the localization and expression of miR-132 in the adult dentate gyrus. They used green fluorescent protein tracking technology based on the Nestin promoter to label neural stem cells and neuron precursors in the dentate gyrus.
The results show that miR-132 is recruited by adult neural stem cells and progenitor cells as part of the response to exercise or aging-related stimuli, but the response to the above stimuli is lost in AD.
Next, the research team assessed whether miR-132 regulation is also involved in the differentiation of human neuron precursors into neurons.
After neural induction of primary human embryonic stem cells into neuron precursors, the level of miR-132 increased significantly, and further increased during the process of neuron maturation, supporting the involvement of miR-132 in the process of human neurogenesis.
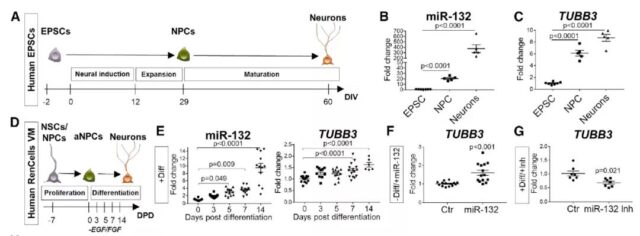
Further research suggests that miR-132 regulates human neurogenesis, and AD-related serum-derived factors inhibit the expression of miR-132 in human stem cells.
In order to clarify the miR-132 regulatory mechanism of AHN, the research team used single-cell RNA sequencing to evaluate the miR-132-specific nestin transcription response. The results showed that there were five significant changes in mRNA, indicating that these transcripts may be regulated by miR-132 Part of the direct effector of a molecular network.
Finally, the functional rescue experiment showed that increasing the level of miR-132 induced the proliferation and differentiation of nerve cells in the dentate gyrus, and significantly improved the memory of the AD mouse model.
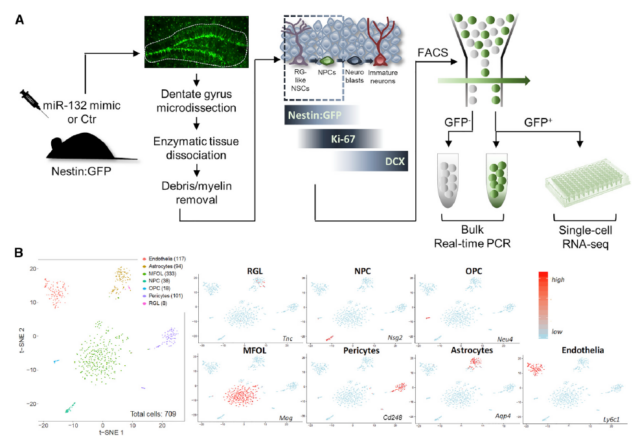
In conclusion, the study confirmed that miR-132 acts as a signal sensor in the nervous system and helps memory formation. Alzheimer’s disease leads to a lack of miR-132 and ultimately impairs adult hippocampal neurogenesis. Delineating the molecular and cellular mechanisms behind the neurogenic effects of miR-132 may provide impetus for identifying new strategies for therapeutic adult hippocampal neurogenesis in Alzheimer’s disease.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



