‘Hangover drug’ Myrkl launched in UK: First batch has sold out
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
‘Hangover drug’ Myrkl launched in UK: First batch has sold out
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
‘Hangover drug’ Myrkl launched in UK: First batch has sold out.
A new ‘pre-drinking pill’ from Sweden hits the UK market today.
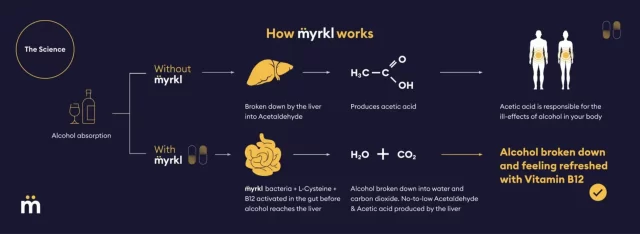
The “hangover drug”, named Myrkl, is said to prevent the formation of toxic acetaldehyde by breaking down alcohol before it reaches the liver.
Acetaldehyde is the main product the liver gets when it metabolizes ethanol in the blood, according to the Duke University Partnership for Alcohol Pharmacology Education.
It’s responsible for alcohol-related flushing, headaches, nausea, and increased heart rate and contributes to the overall horror of a hangover.
So this is definitely something drinkers want to avoid as much as possible.
Myrkl, developed by Swedish probiotic company DeFaire Medical, costs £2 for a two-tablet dose.
It contains strains including Bacillus subtilis and B. coagulans, plus L-cysteine and vitamin B12 and other odds and ends like fermented rice bran, dextrin, fatty acid magnesium salts, and calcium phosphate.
The drug is designed to place gut flora in the eater’s gut, specifically selecting for their ability to metabolize alcohol before it’s absorbed into the bloodstream.
So, it doesn’t change the damaging effects of alcohol on the stomach, but it does seem to drastically reduce the amount of alcohol ingested in the body.
Myrkl’s scientific credibility appears to be built on a single clinical study, funded by DeFaire Medical itself and published last month in the peer-reviewed Nutrition and Metabolic Insights.
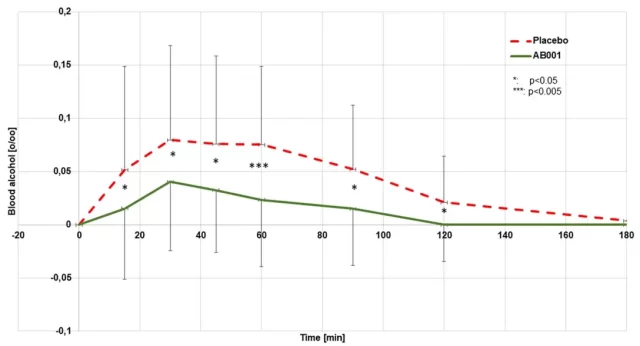
The small study of 24 healthy Caucasian subjects did not fully replicate how Myrkl might be used in the real world, in part because of ethical concerns.
The study did not test the effect of taking a single dose two hours before drinking, but had subjects take one dose per day for a week and then tested the effect of a single drink, which yielded a result of 0.3 grams of alcohol per kilogram of body weight.
So the lightest subjects drank a little less than two standard 30ml glasses of 40% alcohol, while the heaviest drank about three.
In this case, the team found that the study group’s blood alcohol levels peaked at about 50 percent of the control group’s, measured 30 minutes after drinking.
After 60 minutes, the gap between the two groups widened to about 70 percent, with the study group averaging zero blood alcohol after two hours, while the control group took three hours to reach the same level.
The drug appeared to be less effective at reducing the amount of alcohol detectable in the breath — the strongest effect was around 30 percent.
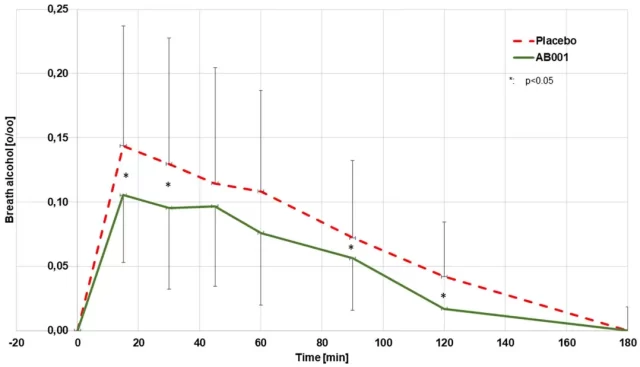
The study’s limitations, however, are fairly obvious — in 10 cases, or 42 percent of the study group, small amounts of alcohol given to participants did not result in measurable blood alcohol concentrations at all.
Researchers are limited to this number during the IRB’s study approval process.
In fact, the team mentions that it was followed through an unpublished but placebo-controlled trial that gave subjects twice the amount of alcohol after only one dose of the drug, with a significantly lower effect, both in the study and control groups.
The difference reaches around 10 percent in blood alcohol levels and 7 percent in breath alcohol levels.
The study doesn’t appear to track acetaldehyde or acetate levels, so it’s unclear how DeFaire supports its advertising claim that the liver doesn’t produce acetaldehyde and acetate.
If alcohol gets into the blood, it ends up in the liver, which produces these substances as it works to neutralize the alcohol and bring it into the body’s waste elimination system.
But drinkers have to understand this: Myrkl doesn’t reduce the damage to the system from alcohol while retaining the “fun” one wants to experience when going to a bar, it just reduces the amount of alcohol that gets into the bloodstream.
Since it’s neutralizing, it’s in a sense a waste of alcohol that people spend a lot of money drinking in their stomachs.
Drinkers can switch every second alcoholic drink to water and drastically reduce blood alcohol, breath alcohol, hangover symptoms, dehydration and the cost of going out at night.
But of course, logic and reason tend to take a back seat when the reason to go out in the first place is to break the logical, rational, employable workday mentality and get yourself a little crazy and have a good time.
As with many things in the modern world, it’s fair to assume that if alcohol had been invented today it would never have been legalized.
The simplicity of alcohol production, the total failure of Prohibition, and the weight of thousands of years of alcoholism all outweigh the well-known damage this stuff does to individuals, families, and society as a whole.
A UK study published by the Independent Scientific Committee on Drugs in 2010 ranked alcohol as the most harmful drug in existence, including heroin, crack, methamphetamine, cocaine, tobacco, cannabis, ketamine, GHB , MDMA and LSD.
But it’s part of the culture – which in the UK now includes a two-liquid “hangover pill” that may give people the impression that they can drink more without suffering any consequences.
At present, the first batch of Myrkl has sold out, which shows that there is a lot of interest in the tablet.
‘Hangover drug’ Myrkl launched in UK: First batch has sold out
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



