How to treat cough after being infected with COVID-19?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
How to treat cough after being infected with COVID-19?
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
How to treat cough after being infected with COVID-19? ICS is here to help!
Nebulized inhalation of budesonide can improve the cough symptoms after COVID-19 infection and improve the curative effect of treatment.
Recently, many people infected with the new coronavirus still cough intermittently after becoming “negative”, which lasts for several days or even weeks, which affects work and life to a certain extent. Therefore, “cough after new coronavirus infection” was once listed Popular searches on major platforms.
The follow-up studies [1] also showed that about 50% of patients with new coronavirus infection will develop cough, and nearly one-fifth of patients have long-term cough (Figure 1). It can be seen that coughing after COVID-19 infection is an urgent problem for most patients.
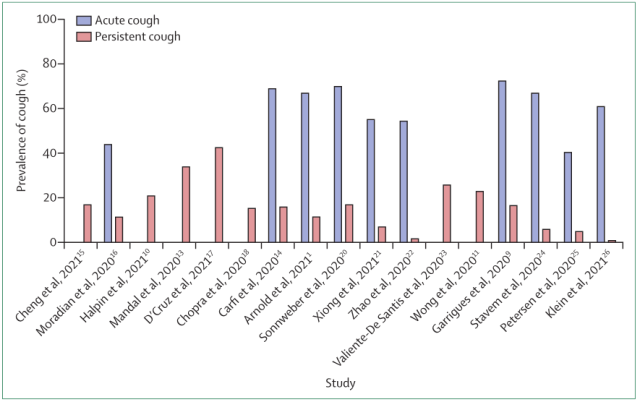
Figure 1 Follow-up study results of cough after COVID-19 infection
Cough after infection with the new coronavirus, ICS may have a good effect
Studies have found that [2] the new coronavirus can widely infect a variety of human cells through the angiotensin-converting enzyme 2 (ACE2) receptor, thereby causing an inflammatory response and causing multiple organ system symptoms including cough, chest pain, and diarrhea.
Sensory neurons also have ACE2 receptors, so the new coronavirus may invade sensory neurons and glial cells of the vagus nerve, and induce the release of neuropeptides and inflammatory mediators, thereby causing cough.
Neuropeptides and neuroinflammatory mediators can further recruit and activate immune cells, leading to lung and airway inflammation and enhanced cough sensitivity (Figure 2) [1] .
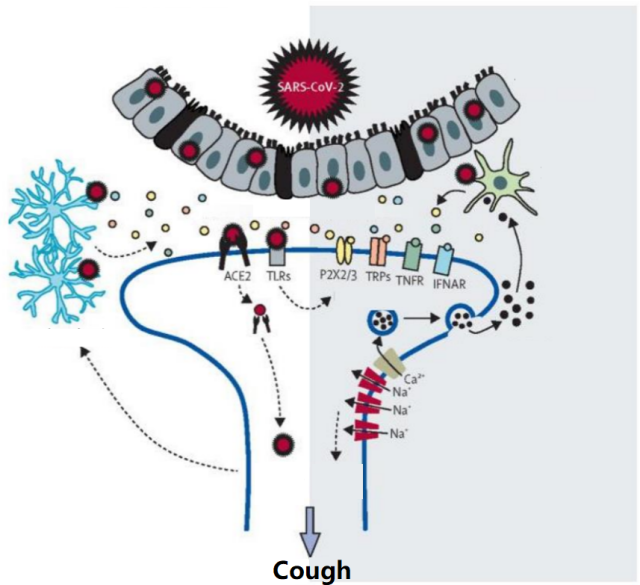
Figure 2 The possible pathogenesis of cough after COVID-19 infection
Previous studies [3] have shown that viral infection of the upper respiratory tract may also induce eosinophilic airway inflammation and airway hyperresponsiveness, which is the subacute stage of eosinophilic bronchial inflammation or cough variant asthma.
The above mechanisms suggest that inhaled glucocorticoids (ICS) may inhibit the release of inflammatory factors from airway epithelial cells through their anti-inflammatory effects, reduce eosinophilic airway inflammation and airway hyperresponsiveness, and thus alleviate the symptoms of COVID-19. Cough symptoms after viral infection.
A previous study [4] analyzed the gene expression of ACE2 in sputum cells of 330 asthmatic subjects and 79 healthy controls.
The results showed that the expression of ACE2 in patients treated with ICS was significantly lower than that in patients without ICS treatment, suggesting that ICS may Related symptoms such as cough after infection with the new coronavirus can be improved by inhibiting the expression of ACE2.
The recently published “Expert Consensus on the Diagnosis and Treatment of Cough Infected by Novel Coronavirus” [5] also pointed out that for patients with subacute cough infected by the new coronavirus, if eosinophilic inflammation or airway hyperresponsiveness is found, ICS is recommended for treatment. .
In addition, for patients with chronic cough caused by cough variant asthma (CVA), eosinophilic bronchitis (EB) and allergic cough (AC), the Guidelines for the Diagnosis and Treatment of Cough (2021) [6] also ICS treatment is recommended.
Several studies have confirmed that inhaled budesonide. Improve cough and other symptoms after infection with the new coronavirus
Among many ICS, budesonide has more evidence-based medical evidence for the treatment of new coronavirus infection.
In vitro studies [7] have shown that inhalation of budesonide can reduce the titer of the new coronavirus, suggesting that budesonide may improve the clinical symptoms of patients with new coronavirus infection.
In order to further evaluate the efficacy of inhaled budesonide on patients with new coronavirus infection, British scholars conducted two open-label randomized controlled trials.
One of the studies (STOIC study) [8] included 146 patients with early COVID-19 infection in the community, 73 received conventional treatment and 73 received budesonide.
The results showed that the average recovery time of the budesonide group was 8 days, while the average recovery time of the conventional treatment group was 12 days, suggesting that inhaled budesonide can shorten the recovery time of symptoms (Figure 3).
In addition, fewer people in the budesonide group had persistent symptoms on day 14 than those in the conventional treatment group (10% vs 30%, p=0.003), indicating that inhaled budesonide can reduce early symptoms in patients with COVID-19 infection.
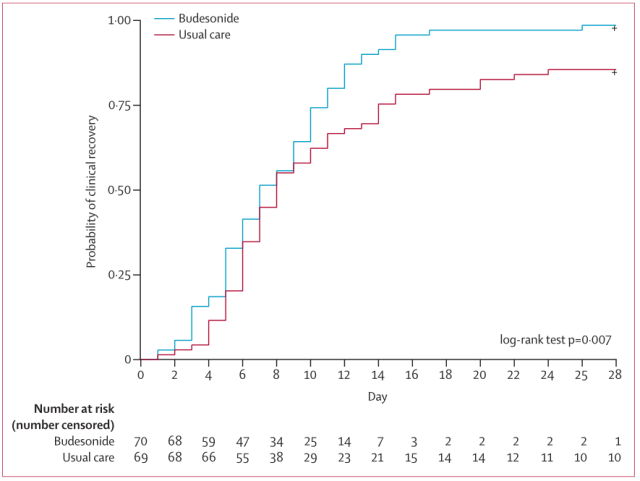
Figure 3 Compared with the conventional treatment group, the average recovery time of the budesonide group was shorter
Another study (PRINCIPLE study) [9] included 2530 elderly and complication patients with COVID-19 infection in the community, of which 787 were in the budesonide group, 1069 in the conventional treatment group, and 974 received other treatments.
The results of the study showed that the first self-reported recovery time of symptoms in the budesonide group was 11.8 days, while that in the conventional treatment group was 14.7 days, and inhaled budesonide could shorten the recovery time of patients by 2.9 days (Figure 4).

Figure 4 The first self-reported recovery time was shorter in the inhaled budesonide group compared with the usual care group
It can be seen that inhalation of budesonide has a significant effect on alleviating the early clinical symptoms of patients with COVID-19 infection.
In addition, the US Food and Drug Administration (FDA) pregnancy classification of budesonide is Class B, and it is relatively safe to use in pregnant women infected with the new coronavirus.
Summary
1. About 50% of patients with COVID-19 infection will have a cough, and nearly one-fifth of the patients have a long-term cough.
2. The new coronavirus can widely infect a variety of cells through the ACE2 receptor, and ICS treatment significantly reduces the expression of ACE2.
3. For subacute cough patients with new coronavirus infection, if eosinophilic inflammation or airway hyperresponsiveness is found, the expert consensus recommends the use of ICS for treatment.
4. In vitro studies have found that inhalation of budesonide can reduce the titer of the new coronavirus.
5. The STOIC study and the PRINCIPLE study showed that inhaled budesonide can shorten the recovery time of patients infected with the new coronavirus and alleviate early symptoms.
6. The FDA pregnancy classification of budesonide is Class B, and it is relatively safe to use in pregnant women infected with the new coronavirus.
references:
[1] Song WJ, Hui CKM, Hull JH, et al. Confronting COVID-19-associated cough and the post-COVID syndrome: role of viral neurotropism, neuroinflammation, and neuroimmune responses. Lancet Respir Med. 2021 May; 9(5 ):533-544.
[2] Crook H, Raza S, Nowell J, et al. Long covid-mechanisms, risk factors, and management. BMJ. 2021 Jul 26;374:n1648.
[3]Lai K, et al. Respirology, 2016, 21(4):683-688.
[4] Peters MC, Sajuthi S, Deford P, et al. COVID-19-related Genes in Sputum Cells in Asthma. Relationship to Demographic Features and Corticosteroids. Am J Respir Crit Care Med. 2020 Jul 1; 202(1): 83-90.
[5] Expert consensus on the diagnosis and treatment of cough caused by novel coronavirus [J/OL]. Chinese Journal of Tuberculosis and Respiratory Medicine, 2023, 46: Internet pre-published.
[6] Asthma Group of Respiratory Branch of Chinese Medical Association. Guidelines for Diagnosis and Treatment of Cough (2021) [J]. Chinese Journal of Tuberculosis and Respiratory Medicine, 2022, 45(1): 13-46.
[7] Heinen N, Meister TL, Klöhn M, et al. Antiviral Effect of Budesonide against SARS-CoV-2. Viruses. 2021 Jul 20;13(7):1411.
[8] Ramakrishnan S, Nicolau DV Jr, Langford B, et al. Inhaled budesonide in the treatment of early COVID-19 (STOIC): a phase 2, open-label, randomized controlled trial. Lancet Respir Med. 2021 Jul;9 (7):763-772.
[9] Yu LM, Bafadhel M, Dorward J, et al. Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial .Lancet. 2021 Sep 4;398(10303):843-855.
How to treat cough after being infected with COVID-19?
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



