World’s Second Pig Heart Transplant Recipient Announced Deceased
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
World’s Second Pig Heart Transplant Recipient Announced Deceased
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
World’s Second Pig Heart Transplant Recipient Announced Deceased After 6 Weeks
Oct 31, the University of Maryland Medical Center in the United States published a memorial article on its official website.
The world’s second recipient of a pig heart transplant tragically passed away on October 30 [1].

Latest memorial article Source: University of Maryland Medical Center
“We are deeply saddened to announce the passing of Lawrence Faucette, a 58-year-old late-stage heart disease patient who underwent the world’s second genetically modified pig heart transplant. Mr. Faucette underwent the transplant surgery on September 20 and survived nearly six weeks post-surgery.”
The first pig heart transplant, which occurred after 2 months, was declared a failure.
The second pig heart transplant recipient was initially admitted on September 14 due to heart failure. The patient’s condition was critical, and without a heart transplant, it would likely have led to death. However, due to peripheral vascular disease and complications from internal bleeding, the evaluation indicated that he did not meet the criteria for heart transplant surgery. The only hope was an experimental treatment that had been used for the first time last year – “pig heart transplant.”
As a result, on the 9th day of admission, the University of Maryland Medical Center in the United States announced that following the global failure of the first “pig heart transplant” in March of last year, the team attempted once again to transplant a genetically engineered pig’s heart into a human on September 20.
The first recipient was a 57-year-old male who was hospitalized for severe heart failure and required a heart transplant. Because he did not meet the standard criteria for conventional heart transplants and mechanical circulatory support after being rejected by four regional and national heart transplant programs, the selection committee at the University of Maryland School of Medicine agreed to consider experimental cross-species heart transplantation.
The research team’s single-patient investigational new drug (IND) application was approved by the FDA, using a combination of 10 gene-edited pig donors (Revivicor), humanized anti-CD40 monoclonal antibody (Kiniksa Pharmaceuticals), and XVIVO heart perfusion system (XVIVO Perfusion).
Additionally, the FDA assessed the research team’s preclinical experience, detailed informed consent, and a plan for monitoring and preventing zoonotic diseases. After three institutional and one external independent psychiatric evaluations confirmed the patient’s informed consent, the pig heart transplant plan was smoothly approved.
The transplant surgery was successfully carried out, and after the surgery, the “pig heart” maintained a sinus rhythm at 70-90 beats per minute, with a normal or high-powered LVEF. Endomyocardial biopsies did not show rejection reactions.
However, starting on the 50th day after the surgery, the levels of free DNA derived from the xenotransplanted heart (xdcfDNA), specific IgG, and IgM serum levels reached their peaks. On the 56th day, endomyocardial biopsies revealed pathological antibody-mediated rejection, with 40% of myocardial cells necrotic, leading to irreversible damage to the xenograft.
On the 60th day post-surgery, with the consent of the family, life support systems were withdrawn, and the world’s second pig heart transplant recipient was declared deceased.
Learning from the First Case, the Second One Still Ends in Failure
After the first patient’s death, the investigation into the reasons for failure continued.
The research team conducted an initial autopsy of the patient’s heart, and the results did not align with typical xenotransplant rejection. The patient’s lead surgeon stated in a webinar hosted by the American Transplantation Society that pig cytomegalovirus may have had a destructive impact on the heart transplant and was a potential cause of the patient’s death. The detailed report of this case was published on June 22, 2022, in NEJM [3].
 Source: Reference [3]
Source: Reference [3]
In fact, during the first transplant, the research team had employed strict breeding practices for the donor pigs, including early weaning, the use of biosafety facilities, and regular pathogen monitoring. They also conducted regular post-operative tests on the patients.
The research team believed that the monitoring results indicated that breeding practices such as avoiding colostrum intake, early weaning, and the use of biosafety facilities could successfully eliminate pig viral pathogens. Testing 60 days after the transplant also confirmed that the virus had not spread to the organ recipient.
However, regardless of how unexpected it was, the positive result for pig cytomegalovirus remained a significant “suspect” in the transplant failure. The team believed that it was possible that the virus had been dormant in the body, evading detection, and was reactivated after surgery, leading to a destructive immune response.
Another case report published on June 29, 2023, in The Lancet confirmed this analysis: after an autopsy, a comprehensive evaluation of recipient organs and the donor heart for pig cytomegalovirus/pig roseola virus was performed using PCR, and viral DNA was detected in all [4].
Therefore, during the second transplant, the team improved infection prevention measures.
The biotechnology company that provided the genetically engineered pig hearts developed a new, more sensitive test for viral DNA and tested donor pig tissue samples, not just nasal swabs. The testing also included pig cytomegalovirus antibodies, which could not only detect whether an infection was ongoing but also whether it had occurred in the past.
Simultaneously, due to the analysis in the above-mentioned report, which found evidence of antibody-mediated rejection in the first recipient, the second case also involved a new attempt at antibody therapy, using an experimental antibody called tegoprubart to block the protein CD154 involved in immune system activation.
These measures briefly boosted the team’s confidence in the second transplant. A month after the surgery, the University of Maryland Medical Center released a video in which Faucette was undergoing physical rehabilitation.
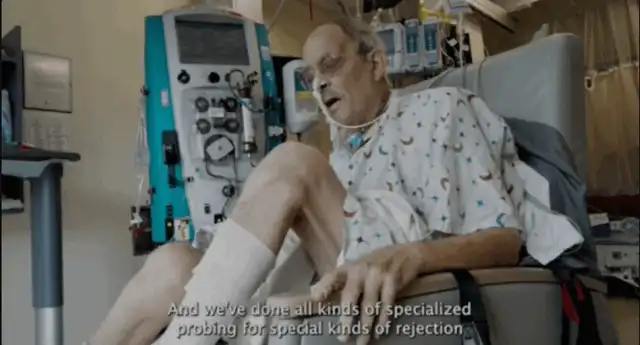 Video screenshot, source: Maryland University Team
Video screenshot, source: Maryland University Team
Muhammad Mohiuddin, the director of the University of Maryland Medical Center’s Xenotransplantation Heart Program, explained, “We’ve stopped all the heart support medications, and his heart is surviving on its own.” Bartley Griffith, the director of the Heart and Lung Transplant Program at the University of Maryland Medical Center who performed the surgery, also added, “Currently, we have seen no evidence of infection or rejection.”
However, similar to the first case, in the absence of clear evidence of rejection initially, the patient subsequently began to show preliminary signs of rejection and passed away within days.
Will Pig Heart Transplants Continue?
In June of last year, a commentary accompanying the case report in the New England Journal of Medicine (NEJM) stated, “Whether this xenotransplant opens a new future for transplantation or not, this study provides a new perspective on how researchers are rapidly and profoundly addressing medical problems through genetic bioengineering and developmental biology.”
After the failure of the first case, conducting the second trial was a more cautious process. A team of 30 FDA experts reviewed over 400 pages of data from the first transplant and ongoing research on non-human primates submitted before approving the second patient at the hospital.
“The ongoing animal studies have not resulted in any deaths; the longest survival post-surgery was 3 months, and it will take at least 1-2
years to obtain data that satisfies FDA requirements,” explained project director Mohiuddin.
However, this experimental treatment was once again rapidly approved through the FDA’s IND application, as patients like the one in this case did not have two years to wait. The experimental treatment of pig heart transplantation could at least give them a chance to survive.
The team stated that the long-term goal of the “pig heart transplant” project is to move from a one-time experiment to formal clinical trials.
After the first patient’s death, the patient’s son stated in a statement released at the hospital, “We are grateful for every innovative moment, every wild dream, and every sleepless night in this historic endeavor. We hope that this story is not the end but the beginning of hope.”
Ann Faucette, the wife of the second patient, expressed her gratitude to the treatment team and her support for the continuation of the research despite her grief.
She mentioned that her husband “had always been open-minded and had full confidence in the treatment team. He knew he might not have much time left, and this was his last chance to contribute to others. He never thought he would live this long or provide so much valuable information for the cross-species transplant project.”
In a memorial article released by the University of Maryland Medical Center, project director Mohiuddin stated, “Mr. Faucette was also a scientist, and in this experiment, he not only understood the results of his own biopsies but also the significant contributions he made to advancing this field.”
“Like the first patient, we plan to conduct extensive analysis to identify factors that can be prevented in future transplants. This will help us move forward and share our experiences with our colleagues.”
Image source: Maryland University Team
World’s Second Pig Heart Transplant Recipient Announced Deceased After 6 Weeks
References:
[1] https://www.umms.org/ummc/news/2023/announcing-the-passing-of-lawrence-faucette
[2] https://www.umms.org/ummc/news/2023/um-medicine-clinicians-perform-second-historic-transplant-of-pig-heart-into-patient
[3] Griffith BP, Goerlich CE, Singh AK, et al. Genetically Modified Porcine-to-Human Cardiac Xenotransplantation. N Engl J Med. 2022;387(1):35-44. doi:10.1056/NEJMoa2201422
[4] Mohiuddin MM, Singh AK, Scobie L, et al. Graft dysfunction in compassionate use of genetically engineered pig-to-human cardiac xenotransplantation: a case report. Lancet. 2023;402(10399):397-410. doi:10.1016/S0140-6736(23)00775-4″
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



