FDA Investigates T-Cell Malignancy Risk in CAR-T Cell Therapy
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
FDA Investigates T-Cell Malignancy Risk in CAR-T Cell Therapy
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Breaking News: FDA Investigates T-Cell Malignancy Risk in CAR-T Cell Therapy
FDA Investigates Serious Risk of T-Cell Malignancy in Patients Following CAR-T Cell Therapy, Involving All 6 Approved Products.
Over the past decade, CAR-T cell therapy has revolutionized the field of oncology, offering a cure for previously incurable blood cancers.
In 2010, Professor Carl June pioneered CAR-T cell therapy in human clinical trials, successfully “curing” several leukemia patients.
In 2017, the FDA approved CAR-T therapy for commercial use, and since then, six CAR-T therapies have received FDA approval for treating blood cancers like leukemia and lymphoma.
The success of CAR-T therapy has rekindled hope for cancer patients awaiting bone marrow transplants and symbolized the arrival of the era of cell therapy.
CAR-T cell therapy, or chimeric antigen receptor T-cell immunotherapy, involves engineering a patient’s immune T-cells outside the body to recognize antigens on the surface of tumor cells.
These modified cells are then reintroduced into the patient’s body to identify and eliminate cancer cells.
Scientists have been exploring the application of CAR-T therapy beyond blood cancers, including solid tumors, autoimmune diseases, chronic infections, heart diseases, and age-related conditions.
Recently, the U.S. FDA announced an investigation into the serious risk of T-cell malignancy in patients who underwent autologous CAR-T cell immunotherapy targeting BCMA or CD19.

Tumor immunotherapy: What is the difference between CAR-T and TCR-T?
The FDA has received reports of T-cell malignancies in patients treated with autologous CAR-T cell immunotherapy targeting BCMA or CD19, both from clinical trials and adverse event data of marketed CAR-T cell therapies.
The FDA has determined that the risk of T-cell malignancy applies to all currently approved autologous CAR-T cell immunotherapies targeting BCMA or CD19. Incidences of T-cell malignancies have been observed in patients treated with these products. The FDA-approved CAR-T therapies include:
- Yescarta and Tecartus developed by Gilead Sciences’ Kite Pharma,
- Kymriah developed by Novartis,
- Breyanzi developed by Bristol Myers Squibb,
- Carvykti developed by Legend Biotech and Johnson & Johnson,
- Abecma developed by Bristol Myers Squibb.
Despite the overall benefits of these CAR-T cell therapies outweighing potential risks, the FDA is investigating the severe consequences of confirmed T-cell malignancy, including hospitalization and death, and evaluating the need for regulatory action.
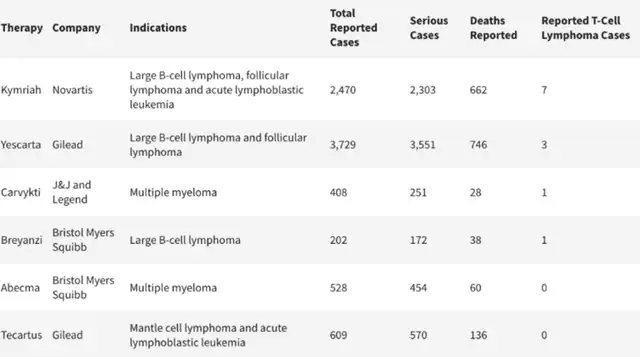
Similar to all gene therapy products using integrated vectors (such as retroviral or lentiviral vectors integrating into the host cell’s genome), potential risks of secondary malignancies are flagged as a class warning in prescriptions for FDA-approved autologous CAR-T cell immunotherapies targeting BCMA and CD19. Approval for these products includes a requirement for 15 years of long-term follow-up observational safety studies post-treatment to assess long-term safety and the risk of secondary malignancies.
The FDA advises patients receiving CAR-T cell therapy and participants in clinical trials to undergo lifelong monitoring for new malignancies. In case of new malignancies post-treatment, patients are urged to report adverse events to the developing company and provide patient samples for detecting chimeric antigen receptor (CAR) transgenes.
In response to the FDA investigation, Gilead Sciences expressed confidence in the overall safety of Tecartus and Yescarta. The company stated that, to date, these therapies have treated 17,700 cancer patients with no evidence suggesting a causal relationship with the development of new malignancies. Gilead emphasizes its rigorous monitoring procedures and commitment to full cooperation with the FDA’s data analysis related to this investigation.
Novartis also expressed confidence in the risk-benefit profile of its CD19-CAR-T product, Kymriah, stating no evidence of a causal relationship between treatment and secondary malignancies among over 10,000 patients treated with Kymriah.
Bristol Myers Squibb, the developer of Abecma and Breyanzi, reported no cases of CAR-positive T-cell malignancies among the 4,700 patients treated with these therapies. The company is responding to FDA requests and maintains confidence in the safety and clinical value of its cell therapies.
Legend Biotech noted that T-cell malignancies might occur in multiple myeloma patients even without CAR-T cell therapy. The company highlighted that other treatments related to multiple myeloma, such as alkylating agents, immunomodulatory drugs, and autologous stem cell transplants, are associated with an increased risk of secondary cancers. Johnson & Johnson shared monitoring data with the FDA for Carvykti, which has been used in 2,000 patients with a favorable benefit-risk ratio.
Yescarta, the first FDA-approved CAR-T cell therapy in 2017, generated $1.13 billion in revenue in the first nine months of 2023, making it the world’s top-selling CAR-T therapy.
Overall, the FDA believes that the benefits of these CAR-T cell therapies still outweigh the potential risks. While investigating the risk of T-cell malignancy associated with treatment, the FDA currently has no plans to withdraw approval for these six CAR-T cell therapies.
The cancer risk associated with CAR-T cell therapy is believed to stem from the use of gene delivery vectors, particularly retroviral or lentiviral vectors. These viral vectors integrate into the host cell’s genome, providing stability to the therapeutic effect. However, if the insertion site is near DNA sequences associated with cancer, there is a potential risk of causing malignancies.
It’s worth noting that on November 8, 2023, researchers from Stanford University published a study in Nature titled “Latent human herpesvirus 6 is reactivated in CAR T cells.”
Using comprehensive genomic analysis, the study linked specific human cell states to the reactivation of latent viruses. Through reanalysis of existing sequencing datasets, the research team discovered the reactivation of human herpesvirus 6 (HHV-6), a betaherpesvirus, in standard CD4+ T cell cultures. Using single-cell genomics, the team described a rare CAR-T cell state with high HHV-6 expression in both in vitro cell cultures and patients.
Based on these findings, the research team suggests that the possibility of latent virus reactivation should be widely considered in current and future cell therapy products.

FDA Investigates T-Cell Malignancy Risk in CAR-T Cell Therapy
Reference:
1.fda.gov/vaccines-blood-biologics/safety-availability-biologics/fda-investigating-serious-risk-t-cell-malignancy-following-bcma-directed-or-cd19-directed-autologous
2. fiercepharma.com/pharma/fda-investigates-serious-risk-secondary-cancer-following-car-t-therapy-treatment
3. nature.com/articles/s41586-023-06704-2
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



