COVID-19 breakthrough infection: 5800 are still infected and 74 died
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
COVID-19 breakthrough infection: 5800 people are still infected and 74 people died after COVID-19 vaccination
COVID-19 breakthrough infection: 5800 are still infected and 74 died . On April 21, NEJM published a paper reporting two cases of “breakthrough infection” of the new coronavirus after immunization with mRNA vaccines.
This paper comes from Rockefeller University. Since the fall of 2020, Rockefeller University will conduct new coronavirus nucleic acid tests for employees and students every week.
Between January 21st and March 17th, 2021, 417 of the tested persons completed two doses of vaccination, and 1491 were not vaccinated. It was tested that 2 people were infected after completing two doses of vaccination, while 32 of the unvaccinated people were infected.
According to the US Centers for Disease Control and Prevention, as of April 2021, 5814 cases of “breakthrough infection” and 74 deaths have been found among more than 75 million people who have completed vaccination in the United States.
Breakthrough infection or virus mutation
“Breakthrough infection”, as the name implies, refers to a pathogen breaking through the vaccine’s defenses, causing people who have completed the vaccination to be infected with diseases that the vaccine should prevent.
Because the level of immune response after vaccination varies greatly among individuals, no vaccine is currently 100% effective in preventing infection. Factors such as viral mutations will also lead to an increase in the frequency of breakthrough infections.
At present, most of the new coronavirus vaccines that have been approved around the world still use the virus gene sequence based on the sequence of the virus strain isolated from the Wuhan infected person in January 2020. However, with the global pandemic of the new coronavirus, mutant strains are constantly appearing around the world, and some of these mutant strains have been confirmed to weaken the protective efficacy of vaccines.
The Rockefeller University sequenced the viral genomes of 2 patients with breakthrough infections, and the results showed that the virus sequence of one of the infected patients was relatively close to the B.1.526 lineage.
The Rockefeller University paper records that an infected person developed symptoms of COVID-19 and tested positive for the virus 19 days after completing the second dose of vaccination. On the 4th day after the diagnosis, the researchers collected the serum of the infected person and tested the neutralizing antibody titers. As a result, it was found that the serum neutralizing antibody titer of this infected person was as high as 3209, which was much higher than the average value of 451 for vaccinators.
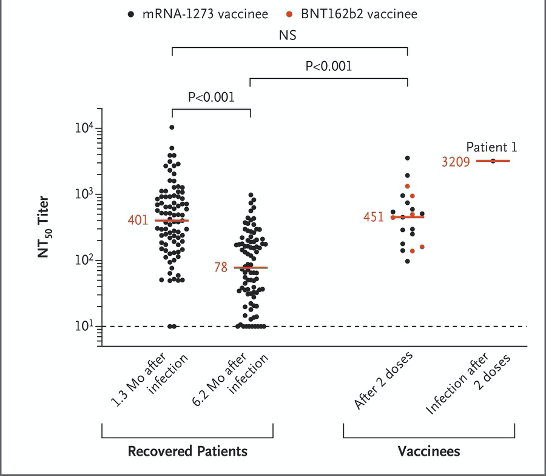
The serum neutralizing antibody titers of people who have broken through the vaccine: The two columns on the left are the titers of COVID-19 patients at 1.3 months and 6.2 months; the third column from the left is the titers of mRNA vaccination. The black dot represents the vaccination of Moderna company vaccine, the red dot represents the vaccination of Pfizer/BioNTech vaccine; the far right is the titer of the vaccinee after diagnosis (Source: NEJM paper)
At the same time, although the infected strain is a mutant strain similar to the B.1.526 lineage, the patient’s serum has a strong neutralizing ability against the original strain, the E484K single point mutant strain and the B.1.526 strain.
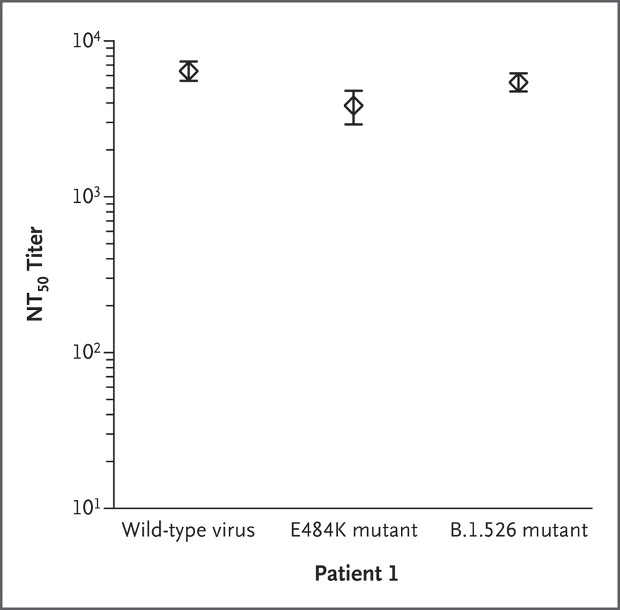
In other words, the vaccinated vaccine has induced an excellent antibody response in the patient. In this case, the virus has broken through the protection provided by the vaccine and infected the patient.
The previous breakthrough infection cases in Xi’an have also been confirmed, and the infection sequence is close to the lineage of the British epidemic strain B.1.1.7. According to the detailed analysis published by China CDC, the patient had previously sampled two returnees carrying B.1.1.7.
According to the data of published papers, B.1.1.7 lineage strains cannot escape the neutralizing antibodies after inoculation of Beijing biological inactivated vaccines. However, due to the lack of samples, it is temporarily impossible to determine the level of neutralizing antibodies of confirmed cases in Xi’an before infection.
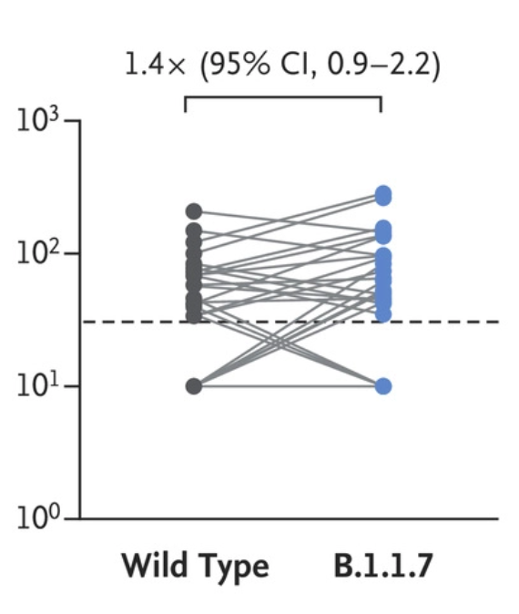
Although it is not completely certain whether the above cases of breakthrough infections are directly caused by viral mutations, the newly emerged mutant strains obviously need to be paid attention to.
For example, the B.1.351 strain that was previously circulating in South Africa has shown that it has significantly escaped the neutralizing antibodies induced by the existing COVID-19 vaccines that have been approved for marketing. The clinical trials of vaccines conducted by AstraZeneca and Johnson & Johnson in South Africa have shown that the protective efficacy is low. Based on test results in other regions.
Subsequently, many vaccine companies have launched strategies to respond to the mutant strains. Moderna initiated the development of the mRNA-1273.351 vaccine, replaced the gene sequence of the vaccine with the B.1.351 strain, and started clinical trials in South Africa.
Vaccines can still prevent severe illness
If the vaccine is still infected, does it mean that the vaccine has no effect? In fact, although vaccines cannot prevent infection 100%, they can still effectively prevent severe illness.
The breakthrough infection cases reported by Rockefeller University were relieved within one week after symptoms such as sore throat, sinus congestion, headache and olfactory failure, and did not develop into severe illness.
The breakthrough infection cases reported in Xi’an, China , showed symptoms of fever, cough and headache, were diagnosed as common type of new coronavirus pneumonia, and recovered and were discharged from the hospital on April 9.
Wang Huaqing, chief expert of the China Disease Control and Immunization Program, said in an interview that the better effect of vaccines is to prevent the occurrence of severe illness, that is to say, the protection efficiency of the COVID-19 vaccine for severe illness prevention is higher. “It is true that after the vaccination, a small number of people have failed to protect. This may be due to individual reasons and some other reasons.”
So, what are the other possible causes of “breakthrough infection”? To discuss this issue, we need to talk back to antibody immunity and cellular immunity.
At the level of antibody immune response, some viruses induce antibody responses that last for decades, and these viruses can often cause viremia. On the contrary, the virus that infects mucosal cells is not easy to cause viremia, and its antibody response can usually only last for months or years. The new coronavirus belongs to the latter.
The serum neutralizing antibody level of the new coronavirus usually peaks about one month after infection or vaccination, then gradually decreases, and drops by about half after 3 months, but it is not known how long it will last.
More importantly, how high levels of neutralizing antibodies can prevent infection? Can the infection be blocked by only relying on neutralizing antibodies? It is still unclear.
As for the “breakthrough infection” mentioned above, the vaccinated persons who have produced neutralizing antibodies in the serum are still infected. This may be due to the insufficient level of antibodies in their mucosal sites, or the cellular immune response, especially the tissue resident memory. The cell response is not strong enough.
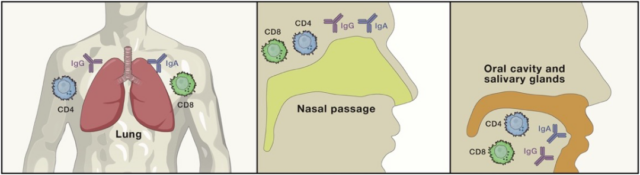
Components of local immunity
Source: References
In order to prevent the disease from becoming severe, T cells may play a more important role than B cells/antibodies. And this has been clinically proven: patients with gamma globulin (IgG) hypotension are usually less severe than patients with impaired T cell function.
The long-term immune response is jointly completed by long-lived plasma cells, memory B cells, CD4+ and CD8+ T cells. Six months after the onset of new coronavirus infections, memory B cells, CD4+ and CD8+ T cells against the new coronavirus can still be detected in their peripheral blood. Based on the experience of influenza viruses, this immune memory may last for many years.
Vaccination can induce memory B cells and memory T cells in the human body. Even if the infection is not blocked, the memory cells will be activated after infection. Compared with unvaccinated people without immune memory, vaccinated people can produce plasma cells and effector T cells faster, speed up the clearance of the virus, and prevent excessive inflammation and the development of severe illness.
This may also provide new ideas for future vaccine design-vaccines immunized through the mucosal route may have the advantage of inducing a higher level of mucosal immunity and enhance the ability to block infection.
Expert Comments:
The breakthrough infection after immunization with the COVID-19 vaccine reminds me of the report released in August last year that the survivors were secondly infected with the COVID-19 virus. The immune effect of the COVID-19 vaccine is actually the same as that of natural infections. It will induce antibody responses against viral antigens in the body. But whether it is a secondary infection of the survivor or a breakthrough infection after vaccination, at least two facts can be told:
1. Although the body has a certain amount of neutralizing antibodies for the new coronavirus, it may still not be 100% guaranteed that re-infection will not occur;
2. Fortunately, the incidence of such re-infection/breakthrough infection is very low for general strains. Unfortunately, new virus mutant strains that continue to appear may be gradually increasing.
Therefore, the current priority for global epidemic prevention is still to speed up the approval of new vaccines, speed up the vaccination of approved vaccines, expand the production capacity of vaccines under the premise of strict prevention and control, and try our best to reverse the disadvantage that the vaccination rate is not as fast as the mutation rate of the virus.
In addition, it is also necessary to make early arrangements for the development of vaccines against mutant strains and introduce targeted regulatory policies. For example, if vaccine manufacturers have been approved to market COVID-19 vaccines, can they have more changes when developing mutant vaccines with the same technical route Fast clinical and approval marketing guidance path.
At present, the new coronavirus has not spread in large numbers in non-human hosts, so there is a time window for the complete elimination of the virus. Only with more vaccines and faster vaccination can we try our best to seize the limited window period.
(source:internet, reference only)
Disclaimer of medicaltrend.org



