It’s not Amyloid β plaquesdoes to cause Alzheimer’s disease
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
It’s not Amyloid β plaquesdoes to cause Alzheimer’s disease
It’s not Amyloid β plaquesdoes to cause Alzheimer’s disease. Amyloid beta plaque doesn’t cause Alzheimer’s disease. This may rewrite the clinical treatment of dementia!
Alzheimer’s disease was widely known as the “long farewell” in the late 20th century due to the slow deterioration of brain function and memory. However, more than 100 years ago, scientist Alois Alzheimer (Alois Alzheimer) first discovered plaques in the brains of patients with this disease. Since then, one of the hallmarks of Alzheimer’s disease (AD) has been the accumulation of amyloid beta plaques in the brain. Scientists have been focusing on treatments to eliminate plaque.
On June 7, 2021, the U.S. Food and Drug Administration (FDA) approved a new drug aducanumab for the treatment of amyloid plaques. The drug can target amyloid deposits in the brains of AD patients and then activate the immune system. The deposited proteins in the brain are removed. This new drug was approved through a special channel of accelerated approval. It was developed and produced by American biotech company Bojian. It is called the world’s first new drug for the potential pathogenesis of Alzheimer’s disease-beta amyloid deposition. .
Beta amyloid deposits are closely related to Alzheimer’s disease. In the past few decades, Alzheimer’s disease caused by this protein deposition has become a mainstream hypothesis in the academic field. But before the approval of Adukamazumab, a clinical trial of a new drug under this line of thinking failed almost every year. Because none of the previous research and development of new drugs has succeeded, the academic community and the industry have increasingly doubted this hypothesis.
But is this drug really effective? Many experts are skeptical!
Up to now, there are two most mainstream hypotheses about the pathogenesis of Alzheimer’s disease, namely the amyloid cascade hypothesis and the microtubule-associated protein (Tau protein) hypothesis. The amyloid cascade hypothesis believes that the deposition of beta amyloid, the main component of plaque in the patient’s brain, is the pathological source of Alzheimer’s disease. Neurofibrillary tangles, cell reduction, blood vessel reduction and even dementia are all direct results of this deposition.
After a series of failures, scientists and new drug companies have gradually turned their attention to other possible Alzheimer’s disease pathogenesis, with more than 30 related hypotheses. Among them, the clinical research phase involves more hypotheses about the relationship between the structure and function of the brain itself that cause Alzheimer’s disease, such as neuroimmune response, neuroinflammatory response, etc. But the amyloid cascade hypothesis “rules” the development of new drugs for Alzheimer’s disease for more than 20 years.
The current mainstream view of Alzheimer’s disease is that this disease is related to beta amyloid plaques. But so many medicines have failed. In fact, everyone has been questioning whether β-amyloid protein is a mechanism for curing diseases or a result of getting sick. Whether it is a cause or a result is actually still controversial.
A recent study published in EClinicalMedicine has once again subverted the traditional view that the accumulation of amyloid beta plaques in the brain is the cause of Alzheimer’s disease. Studies have found that it is not plaques that cause cognitive impairment. Amyloid plaques are the result of Alzheimer’s disease, not the cause.
The team believes that cognitive impairment may be due to the reduction of soluble amyloid beta peptides rather than the corresponding accumulation of amyloid plaques. To test their hypothesis, they analyzed the brain scans and spinal fluid of 600 people who participated in the Alzheimer’s Neuroimaging Program study, all of whom had amyloid plaques. From there, they compared the number of plaques and peptide levels between cognitively normal individuals and cognitively impaired individuals. They found that individuals with high levels of peptides are cognitively normal regardless of the number of plaques in the brain.
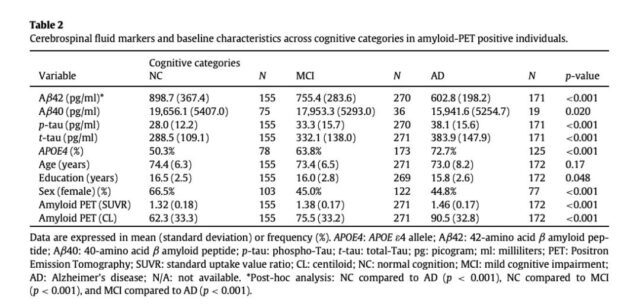
NC: Normal Cognition; MCI: Mild Cognitive Impairment; AD: Alzheimer’s Disease Aβ42: Amyloid β peptide of 42 amino acids; Aβ40: Amyloid β peptide of 40 amino acids
They also found that the level of soluble Aβ42 observed in NC (864.00 pg/ml) is higher than that of MCI (768.60 pg/ml) or AD (617.46 pg/ml). Higher soluble Aβ42 levels are also associated with better neuropsychological effects. Function is related to the larger size of the hippocampus, which is the most important area of the brain for memory. Despite increased cerebral amyloidosis, normal cognition and hippocampal volume are associated with maintaining high levels of soluble Aβ42.
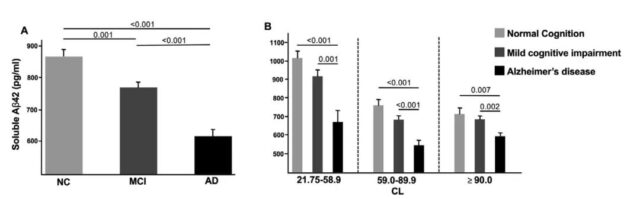
The picture compares with subjects with mild cognitive impairment and Alzheimer’s disease, the adjustment analysis of amyloid-positive normal cognition. (A) Soluble Aβ42 levels in each diagnostic category; (B) Soluble Aβ42 levels in each diagnostic category in the CL tertiles.
According to the authors, most people will develop amyloid disease as they get older, but very few people will develop dementia. They say that in fact, by the age of 85, 60% of people will develop these amyloid diseases, but only 10% will develop dementia.
The key finding of the study is that the symptoms of Alzheimer’s disease seem to depend on the consumption of the normal soluble Aβ42 protein, which is in a soluble state rather than aggregated into plaques, Espay said. A related future treatment will be to supplement these brain soluble Aβ42 proteins to normal levels.
The research team is now working to test their findings in animal models. If successful, the treatments in the future may be very different from the treatments tried in the past two decades. Espay said treatment may include increasing the normal soluble Aβ42 protein to keep the brain healthy while preventing the protein from hardening into plaque.
The Alzheimer’s Disease Drug Development Product Line: 2021 report released by the American Alzheimer’s Disease Society in May this year shows that as of January 2021, a total of 152 clinical experiences are in progress, covering 126 research projects. drug. Among them, 41 clinical trials were phase III, 87 were phase II, and 24 were phase I. More than 80% of the drugs under development belong to the category of altering disease progression, that is, acting against the underlying biological mechanism of Alzheimer’s disease. The remaining two categories are aimed at improving cognitive function and reducing neuropsychiatric symptoms.
However, among the drugs under development in the category of changing disease progression, the main targets of β-amyloid and tau are 16 and 11, respectively, accounting for 26% of the total. Among all drugs under investigation in Phase III clinical trials, the number of drugs targeting β-amyloid and tau protein accounted for 35%. This means that the two hypotheses, especially the β amyloid hypothesis, are still in an important position.
Along with a series of major failures, pharmaceutical companies’ investment in this disease has also shown a downward trend in the past few years. By 2021, only 49% of funders will come from the biopharmaceutical industry. The rest includes 29% from academic medical centers funded by the National Institutes of Health (NIH), 14% from public-private partnerships, and 7% from others. The report pointed out that only when new therapies are approved for marketing by the drug regulatory authority, the field of Alzheimer’s disease may attract more researchers and investors.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



