CRISPR gene editing has great potential for Alzheimer’s disease
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
CRISPR gene editing has great potential for the prevention and treatment of Alzheimer’s disease
CRISPR gene editing has great potential for Alzheimer’s disease. Alzheimer’s disease (AD), commonly known as “Alzheimer’s disease”, is a serious neurodegenerative disease. Patients usually suffer from memory decline and weakened learning ability, accompanied by emotional regulation. Obstacles and loss of athletic ability greatly affect the development of individuals, families and even society.
Currently, about 50 million people worldwide suffer from Alzheimer’s disease. As the average life expectancy of human beings increases and the aging society intensifies, the prevalence of Alzheimer’s disease is also rising. It is estimated that by 2050, Alzheimer’s patients will increase to more than 150 million.
The cause of Alzheimer’s disease is complex, and the scientific community has not yet deciphered the specific mechanism of Alzheimer’s disease. It is estimated that about 1%-5% of Alzheimer’s disease is highly genetically related to family history. It belongs to familial Alzheimer’s disease, which is caused by the gene encoding amyloid beta precursor protein (APP) and presenilin 1 It is caused by a dominant mutation of the presenilin 2 gene, which has a clear genetic cause and can be diagnosed before the appearance of cognitive impairment, but there is still no effective treatment method. In China alone, more than 500,000 people suffer from hereditary familial Alzheimer’s disease.
Recently, researchers from Hong Kong University of Science and Technology, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, and California Institute of Technology published a titled Brain-wide Cas9-mediated cleavage of a gene causing familial Alzheimer’s disease alleviates amyloid-related pathologies in Nature Biomedical Engineering. in mice’s research paper.
The research team has developed a whole-brain level CRISPR-Cas9 genome editing technology that can reduce the pathology associated with pale powder-like proteins in a mouse model of Alzheimer’s disease. This research provides great hope for the development of new long-acting treatments for Alzheimer’s disease.

The use of genome editing technologies such as CRISPR-Cas9 as a treatment strategy for diseases caused by gene mutations (such as familial Alzheimer’s disease) has great potential, especially to correct pathogenic gene mutations before symptoms appear, which is considered to be The “one-and-for-all” treatment method, because once the genetic mutation is corrected, the effect can last a lifetime.
However, the well-known blood-brain barrier hinders the clinical development and application of gene editing for brain-related diseases. The blood-brain barrier lacks an effective non-invasive method to deliver gene editing components to the brain. In addition, the existing gene editing technology cannot produce beneficial results in the entire brain.
The team of Professor Ye Yuru, vice president of the Hong Kong University of Science and Technology, used the AAV-PHP.eB vector. This serotype AAV virus can cross the blood-brain barrier after intravenous injection and target the entire brain.
Through this AAV-PHP.eB vector, the CRISPR genome editing system that targets the amyloid beta precursor protein (APP) gene is injected intravenously.
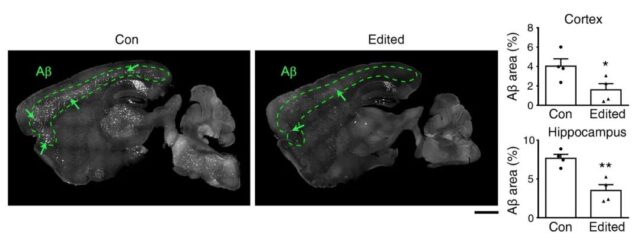
After 6 months of this gene editing treatment in Alzheimer’s disease transgenic mice, the amyloid plaques in the brain that are thought to cause Alzheimer’s neurodegenerative diseases still remain low. It shows that the single treatment of this method can maintain long-term curative effect, and there are no obvious side effects.
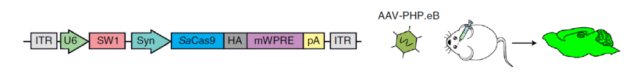
This method can not only cross the blood-brain barrier, but also deliver optimized genome editing tools to the entire brain through a single, non-invasive intravenous injection, achieving efficient whole-brain gene editing.
Through this method, the research team effectively destroyed the familial Alzheimer’s disease gene mutation in the Alzheimer’s transgenic mouse model, and improved the pathological symptoms of Alzheimer’s disease throughout the brain, which is a new treatment for the disease. Development paved the way.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



