COVID-19 vaccines: Summary of Antigen Design and Delivery Materials
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
COVID-19 vaccines: Summary of Antigen Design and Delivery Materials
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
COVID-19 vaccines: Summary of Antigen Design and Delivery Materials.
Abstract:
Coronavirus is prevalent, and the COVID-19 vaccine has attracted much attention.
The vaccine development platform mainly develops low-dose, long-lasting vaccines for the SARS-CoV-2 spike protein antigen.
At present, most of the materials used in COVID-19 vaccines and adjuvants are at the nanometer or micrometer level.
Among them , the antigen design of mRNA vaccines, subunit vaccines, and virus particle vaccines using polymer display technology to form nanoparticles has received widespread attention.
This article mainly summarized the antigen design and delivery materials of the new COVID-19 vaccine.
Introduction
At present, the main mechanism of COVID-19 vaccine is to deliver the antigen to the immune system to trigger an adaptive immune response.
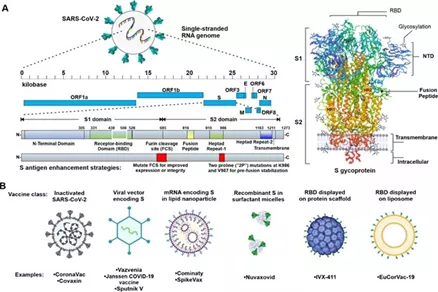
As shown in the figure, SARS-CoV-2 is a complex enveloped single-stranded RNA coronavirus .
Figure A is a schematic diagram of the SARS-CoV-2 genome and its S glycoprotein.
Among them, the S glycoprotein has been developed by most vaccines. central focus, but the membrane (M) , envelope (E) or nucleocapsid (N) ; protein in the future are likely to occur as a supplement to a vaccine antigen B diagram shows the vaccine and have clinical representative .
3 experiment of SARS- CoV-2 vaccine category. The advanced vaccine materials used can allow the S antigen itself or the protein encoded by the sequence to produce the corresponding antigen protein in situ in the host.
At the same time, the vaccine adjuvant can enhance the antigen presentation through the antigen library effect, induce the production of chemokines and cytokines, and recruit the injection site.
Immune cells and other mechanisms of action promote the transport of antigens to the origin of the response immune response.
Although some new vaccines such as mRNA vaccines can produce antigen proteins in situ in the host in a safe and highly modular manner, the high molecular weight of nucleic acids and their strong negative charge hinder their delivery on the cell membrane and are easy Degraded in the body.
Therefore, new vaccines often require delivery systems , and research on related materials is still hot.
The first part of this issue will first review the design of new COVID-19 vaccine antigens.
Principles of Antigen Design and Delivery
The receptor binding domain ( RBD ) in the S subunit of SARS-CoV-2 has a lower degree of glycosylation than S , which can improve its immunogenicity and induce high-frequency neutralizing antibodies, which is expected to become a neutralizing antibody induction Target.
Meanwhile, RBD of low molecular weight, reduces its immunogenicity, thus increasing antigen density of the RBD Strategies multimerization increasingly popular.
Among them, nanoparticles can be used to display the antigen density. This delivery platform should be flexible enough to quickly adjust and adapt to cope with virus mutations with mutations that evade neutralization.
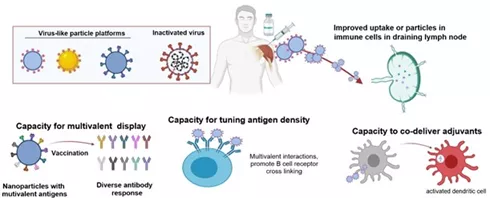
The figure shows the potential advantages of some viral particle platforms and nanoscale antigen delivery systems.
Many vaccine platforms, such as inactivated viruses, have nano-scale sizes that can enhance the uptake of antigens by immune cells in draining lymph nodes.
In addition, antigen delivery systems can usually promote multivalent antigen responses, regulate antigen density, and in some cases can deliver adjuvants together with antigens.
Key vaccine antigen design
mRNA vaccine
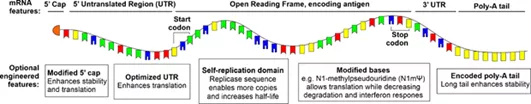
The optimization of mRNA vaccines through mRNA modification and stable delivery methods has become the focus of the development of COVID-19 vaccines.
As shown in the figure, the 5’cap, 5’untranslated region (UTR), coding sequence, 3’UTR, and polyadenylation tail in the mRNA molecule are all optimized targets, especially n1-methylpseudouridine (N1mΨ) Instead of uridine, it has been used in Pfizer’s Pfizer/BioNTech BNT162b2 vaccine and Modern mRNA-127 vaccine.
Modification and optimization have improved the stability of the vaccine to a certain extent.
An emerging method of mRNA vaccines is developed from amplified RNA (saRNA), which encodes a replicase sequence, and it is possible to reduce the injection dose by extending the half-life of mRNA.
Protein subunit vaccine
Protein subunit vaccines do not contain viral genetic material and are highly safe. For example , the S glycoprotein of SARS-CoV-2 and its RBD have always been the key research objects of protein subunit vaccines.
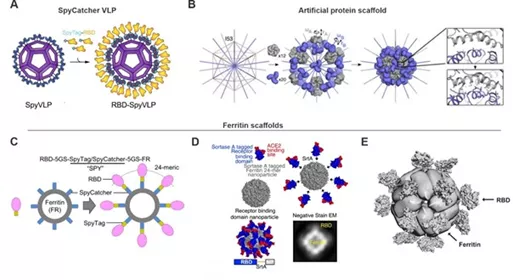
As shown in the table, the Binding Technology show common antigen component nanoparticles protein subunits and subunit vaccines connection technologies, such as connection table based inverse streptococcal protein SpyTag / SpyCatcher system or artificial protein scaffold Artificial Protein the scaffold , the individual The protein folding subunits are separated from the final macromolecular structure assembly.
The above operations can effectively enhance the immunogenicity of the vaccine and realize the low-dose and high-efficiency characteristics of the vaccine.
Virus-like particle vaccine
As a component of subunit vaccines, protein scaffolds use self-assembling proteins to form virus-like particles (VLP) , which have a clear monomer size and number, and can be displayed on their surface through coupling, gene fusion or tag coupling The antigen of interest. Because of its small molecular weight, RBD is often used as the main subject of virus-like particles (VLP) research.
As shown in the figure, SpyVLP has successfully realized the transformation to RBD-SpyVLP virus-like particle vaccine through the SpyCatcher003-VLP technology platform ;
the SpyTag/Spycatcher system can be used to couple RBD to 24 poly subunit ferritin particles; the expression contains c- terminal Classification enzyme a donor sequence and Helicobacter pylori ferritin nanoparticle sequence RBD , constructed SARS-CoV-2RBD nanoparticles ;
surface functionalized ferritin can self-assemble, using SpyTag/Spycatcher technology to make SARS-CoV-1 /2RBDS is attached to ferritin nanoparticles and forms virus-like particles (VLP) through self-assembly .
At the same time, SpyTag/Spycatcher mentioned aboveTechnology can not only promote the host to produce a specific cellular response, but also help the host produce neutralizing antibodies to neutralize other coronaviruses, and provide cross-protection to a certain extent.
Summary
The development of advanced technology promotes the optimization of vaccine antigen design.
The currently optimized mRNA vaccine will be combined with cationic lipid materials for delivery stage research, and the self-assembly technology of protein subunit vaccines has also been developed to the stage of clinical trials. In the future, more information about the effectiveness and safety of these candidate vaccines will appear, and the next part will continue to summarize the delivery materials of the new COVID-19 vaccine.
References:
Advanced Materials for SARS-CoV-2 Vaccines
COVID-19 vaccines: Summary of Antigen Design and Delivery Materials
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



