Radioactive iodine therapy increases risks of new cancers and younger age with more risks
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Radioactive iodine therapy increases risks of new cancers and younger age with more risks
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Radioactive iodine therapy increases risks of new cancers and younger age with more risks.
Weighing the pros and cons of treatment modalities is an eternal topic in clinical practice, especially in this age of precision medicine and personalized treatment.
In the past 10 years, with the deepening of the understanding of the disease, for some thyroid cancers with a lower degree of malignancy, such as differentiated thyroid cancer (DTC), the treatment concept has changed from the previous aggressive to mild, which not only includes surgical resection. It also includes radioactive iodine therapy (RAI) to adapt to changes in the population [1].
Currently, there is a consensus that radioactive iodine therapy is not required for patients with differentiated thyroid cancer with single focal microcarcinoma (diameter ≤10 mm) [2].
For other low-risk differentiated thyroid cancer patients, the latest clinical studies have shown that the long-term prognosis of patients who do not receive RAI is not worse than that of patients who receive RAI [3]. For these patients, RAI not only fails to improve the prognosis, but may bring additional risks of hematological tumors such as leukemia [4].
However, the effect of RAI on the risk of solid second primary malignancy (SPM) remains inconclusive [5].
Recently, the team of Professor Cari M. Kitahara from the National Cancer Institute of the United States assessed the risk of solid SPM in differentiated thyroid cancer patients treated with RAI before the age of 45 by using the SEER database .
The results of the study showed that RAI treatment was associated with a 23% increase in the risk of solid SPM , and the younger the age, the greater the risk of solid SPM in the future . The relevant research results were published in the Journal of Clinical Oncology (JCO) [6].

Since the 1940s, due to its high tissue specificity, RAI has become one of the main treatments for thyroid cancer, improving the prognosis of thyroid cancer patients [7].
Compared with traditional radiotherapy, patients have better tolerance to RAI, but in the long run, many patients still suffer from complications such as salivary gland dysfunction, conjunctivitis, and infertility [8], while The risk of secondary malignancy after radiation exposure is of particular concern, especially in younger patients.
Therefore, exploring the risk of RAI for secondary malignant tumors in young differentiated thyroid cancer population will help to balance the benefits and risks of RAI treatment .
The team of Prof. Cari M. Kitahara included in the SEER database of patients in the United States diagnosed with differentiated thyroid cancer (papillary or follicular carcinoma) before the age of 45 from 1975 to 2017, and excluded cases with distant metastases at diagnosis , to assess the risk of solid SPM in patients 5 years after diagnosis of differentiated thyroid cancer .
A total of 36,311 young patients with non-metastatic differentiated thyroid cancer were included in the study, of whom 81% were women and 45% received RAI therapy . RAI use was higher in male patients (50%) than in female patients (44%), and more than half (55%) of patients under the age of 15 received RAI therapy .
Overall, RAI usage increased from 9% to 55% between 1975-2009 and then gradually declined, dropping to 39% in 2017. RAI usage showed the same trend in patients with tumors <1 cm and those with tumors ≥1 cm.
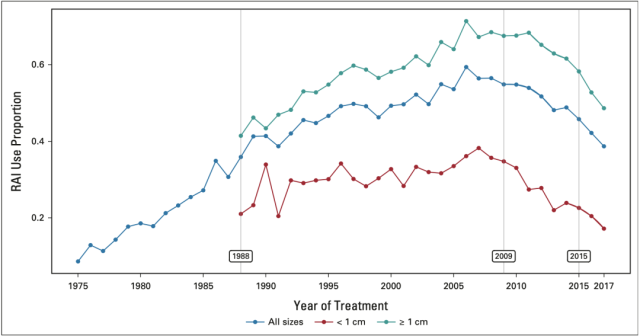
Changes in RAI usage in patients with differentiated thyroid cancer from 1975 to 2017
The researchers mainly analyzed the risk of solid SPM in patients (27,050 with analyzable data) 5 years after the diagnosis of differentiated thyroid cancer . RESULTS: During the follow-up period (median follow-up time 15.6 years), 1524 people were suffering from real SPM, and RAI treatment was associated with an increased risk of solid SPM (RR=1.23, 95%CI: 1.11-1.37) .
At 20 years after the diagnosis of differentiated thyroid cancer , the cumulative incidence of solid SPM was 5.6% (95% CI, 5.0-6.0) and 5.0% (95% CI, 95% CI: 4.6-5.4) .
This difference increased with follow-up time, with the cumulative incidence of solid SPM in RAI-treated and RAI-naïve patients 30 years after diagnosis of differentiated thyroid cancer was 12.5% (95% CI, 11.3-13.8) and 10.2% (95% CI, 9.5-11.0) .
Among organs with high radiation exposure (≥0.5 Gy), salivary gland cancer (RR=2.15, 95%CI: 0.91-5.08), gastric cancer (RR=1.61, 95%CI: 0.70-3.69) and renal cancer (RR=1.34) , 95%CI: 1.14-2.09) with a higher risk of SPM associated with RAI therapy.
For organs with relatively low radiation exposure (<0.5 Gy), uterine-related cancers (RR=1.55, 95%CI: 0.99-1.40), breast cancer (RR=1.18, 95%CI: 0.99-1.40) and lung cancer (RR=1.18, 95%CI: 0.99-1.40) 1.42, 95%CI: 1.03-2.32) had a higher risk.
With the exception of bladder cancer, cervical cancer, and liver cancer, most other solid SPMs had RRs higher than 1.
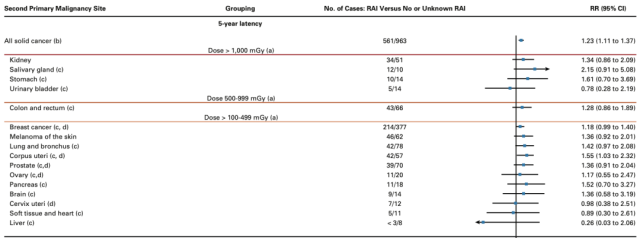
RAI treatment and entity SPM risk relationship
Younger patients had a higher RR with solid SPM compared with older patients (P=0.07). The RR was 1.60 (95% CI, 1.07-2.40) for patients before age 25, and 1.16 (95% CI, 1.02-1.33) for patients 35-45 years of age.
With the extension of follow-up time after the diagnosis of differentiated thyroid cancer , the RR of solid SPM increased (P=0.07).
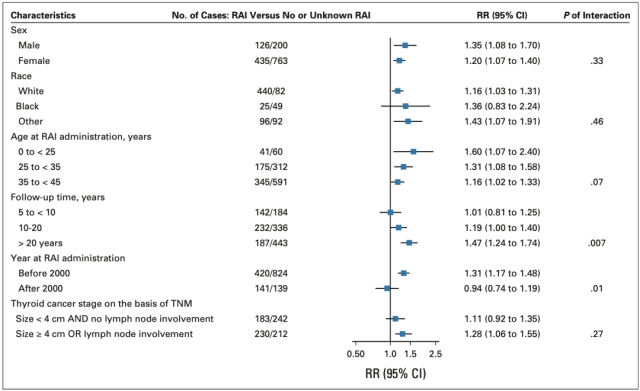
Risk stratification of solid SPM in patients with differentiated thyroid cancer
Professor Cari M. Kitahara’s team also analyzed the effect of RAI treatment on the risk of hematological tumors in patients with differentiated thyroid cancer .
Two years after the diagnosis of differentiated thyroid cancer , 146 people were diagnosed with hematologic SPM during the follow-up period (median follow-up time 13 years) .
RAI treatment increased the risk of leukemia (RR=1.92, 95%CI: 1.04-3.56) , especially non- lymphocytic leukemia (RR=2.17, 95%CI: 1.03-4.55) . The risk of Hodgkin or non-Hodgkin lymphoma was not increased with RAI treatment.

RAI treatment and risk of hematologic SPM
Although the study has some limitations, such as not considering the interaction between RAI treatment and cancer risk factors such as smoking and obesity, and not including patients with differentiated thyroid cancer over the age of 45 , it still shows that RAI treatment will lead to better outcomes. High risk associated with solid SPM , especially in patients with differentiated thyroid cancer <25 years of age .
The results of this study are an important reference for doctors to weigh whether to give RAI therapy to patients with differentiated thyroid cancer .
On the basis of correctly identifying patients who are most likely to benefit from RAI therapy, it is also necessary to consider whether the therapy will give patients The adverse effects brought by, comprehensive analysis and medical decision-making , this is the real precision medicine and personalized treatment.
Thinking of the previous “New England Journal of Medicine” results that RAI treatment did not improve the prognosis of patients with low-risk differentiated thyroid cancer , coupled with RAI will increase the risk of SPM patients .
The answer is almost there, RAI treatment will gradually It is no longer the main treatment for patients with low-risk differentiated thyroid cancer , and the indications for RAI therapy will be further strictly controlled.
References:
1. Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, Pacini F, Randolph GW, Sawka AM, Schlumberger M et al: 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer : The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26(1):1-133.
2. Verburg FA, Flux G, Giovanella L, van Nostrand D, Muylle K, Luster M: Differentiated thyroid cancer patients potentially benefitting from postoperative Mol I-131 therapy: a review of the literature of the past decade. Eur J Nucl Med Imaging 2020, 47(1):78-83.
3. Leboulleux S, Bournaud C, Chougnet CN, Zerdoud S, Al Ghuzlan A, Catargi B, Do Cao C, Kelly A, Barge ML, Lacroix L et al: Thyroidectomy without Radioiodine in Patients with Low-Risk Thyroid Cancer. N Engl J Med 2022, 386(10):923-932.
4. Molenaar RJ, Sidana S, Radivoyevitch T, Advani AS, Gerds AT, Carraway HE, Angelini D, Kalaycio M, Nazha A, Adelstein DJ et al: Risk of Hematologic Malignancies After Radioiodine Treatment of Well-Differentiated Thyroid Cancer. J Clin Oncol 2018, 36(18):1831-1839.
5. Yu CY, Saeed O, Goldberg AS, Farooq S, Fazelzad R, Goldstein DP, Tsang RW, Brierley JD, Ezzat S, Thabane L et al: A Systematic Review and Meta-Analysis of Subsequent Malignant Neoplasm Risk After Radioactive Iodine Treatment of Thyroid Cancer. Thyroid 2018, 28(12):1662-1673.
6. Pasqual E, Schonfeld S, Morton LM, Villoing D, Lee C, Berrington de Gonzalez A, Kitahara CM: Association Between Radioactive Iodine Treatment for Pediatric and Young Adulthood Differentiated Thyroid Cancer and Risk of Second Primary Malignancies. J Clin Oncol 2022: JCO2101841.
7. Lim H, Devesa SS, Sosa JA, Check D, Kitahara CM: Trends in Thyroid Cancer Incidence and Mortality in the United States, 1974-2013. JAMA 2017, 317(13):1338-1348.
8. Piek MW, Postma EL, van Leeuwaarde R, de Boer JP, Bos AME, Lok C, Stokkel M, Filipe MD, van der Ploeg IMC: The Effect of Radioactive Iodine Therapy on Ovarian Function and Fertility in Female Thyroid Cancer Patients: A Systematic Review and Meta-Analysis. Thyroid 2021, 31(4):658-668.
Radioactive iodine therapy increases risks of new cancers and younger age with more risks
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



