Biomarkers can detect rapid aging of organs and disease risks
- EB Virus Could Be Infected by Kiss: A Hidden Threat Linked to Cancer
- The Silent Threat: How Gas Stoves Pollute Our Homes and Impact Health
- Paternal Microbiome Perturbations Impact Offspring Fitness
- New Report Casts Doubt on Maradona’s Cause of Death and Rocks Manslaughter Case
- Chinese academician unable to provide the exact source of liver transplants
- Early Biomarker for Multiple Sclerosis Development Identified Years in Advance
Biomarkers can detect rapid aging of organs and disease risks
- AstraZeneca Admits for the First Time that its COVID Vaccine Has Blood Clot Side Effects
- Was COVID virus leaked from the Chinese WIV lab?
- HIV Cure Research: New Study Links Viral DNA Levels to Spontaneous Control
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Biomarkers can detect rapid aging of organs and disease risks
Researchers have employed a machine learning model to analyze blood proteins, estimating the biological age of body organs rather than chronological age. This method, which predicts the risk of diseases such as heart failure, Alzheimer’s, and diabetes in seemingly healthy individuals, could aid in earlier intervention.
Chronological age does not always align with physiological age. The aging process can lead to the degradation of organ structure and function, increasing the risk of most chronic diseases. While there are various methods to assess biological aging, most provide a singular whole-body measurement without specific organ age information.
A study led by researchers from Stanford University School of Medicine identified protein biomarkers that can estimate the biological age of organs, subsequently predicting an individual’s disease risk.
The lead author of the study, Tony Wyss-Coray, stated, “We can estimate the biological age of a specific organ in an outwardly healthy individual. This, in turn, can predict the risk of diseases associated with that organ.”
Researchers studied 11 organs, organ systems, or tissues: heart, lungs, brain, kidneys, liver, pancreas, intestines, muscles, fat, immune system, and blood vessels (vascular system). They measured 4979 proteins in 5676 adults aged 20 to 90 years and identified proteins with gene expression levels at least four times higher in a specific organ than in others. Out of all measured proteins, 856 (17.9%) met this definition.
They trained a machine learning algorithm using high-expression proteins as input to estimate the age of 11 organs. They also trained an “organ” model using organ non-specific plasma proteins and a “general” model using all proteins (regardless of specificity) to compare the contribution of specific organs to common aging features. For each individual, the algorithm generated an “age gap,” indicating the biological age relative to peers. Previous studies found an association between age gap and the risk of death.
Researchers observed that individuals with the same general age gap exhibited different aging characteristics in specific organs, with some showing extreme aging in one or more organs compared to the general population.
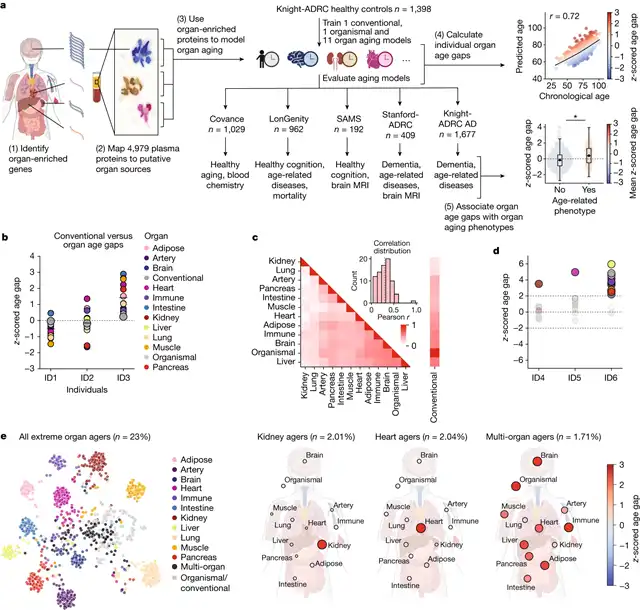
Wyss-Coray explained, “When we compared the biological ages of these organs in each person with the corresponding organ ages in a large group of individuals without apparent serious diseases, we found that, in people aged 50 or above, 18.4% had at least one organ aging faster than the average. We found that these individuals had an increased risk of developing diseases related to that organ in the next 15 years.”
Individuals with an organ aging faster faced a 20% to 50% higher risk of death. While only 1.7% showed extreme aging in multiple organs, their risk of death was significantly increased, 6.5 times higher than those without any visibly aging organs.
Concerning specific organs, individuals with accelerated heart aging had a 250% increased risk of heart failure, as well as an elevated risk of heart attacks and atrial fibrillation. An increase of one standard deviation in brain age gap correlated with a 34% increased clinical risk of cognitive decline within five years. Accelerated aging of the brain and blood vessels independently predicted the progression of Alzheimer’s disease, separate from the current blood-based Alzheimer’s biomarker—tau protein levels—and with similar strength. Additionally, kidneys with extreme aging (two standard deviations beyond normal) showed a close association with hypertension and diabetes.
Identifying rapidly aging organs in outwardly healthy individuals could mean earlier treatment. Determining organ-specific proteins indicating excessive aging and related disease risks could lead to new targeted drugs.
Researchers stated, “The organ aging model generated can predict mortality, organ-specific functional decline, disease risks and progression, and aging heterogeneity between tissues. This method is minimally invasive, requiring only a small blood sample, and can easily be applied to understand the effects of health interventions at the organ level, such as lifestyle adjustments and drug therapy.”
This research was published in the journal “Nature.”
Biomarkers can detect rapid aging of organs and disease risks
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



