Gene Therapy for Genetic Heart Disease to Begin Clinical Trials Soon
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Gene Therapy for Genetic Heart Disease to Begin Clinical Trials Soon
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Gene Therapy for Genetic Heart Disease to Begin Clinical Trials Soon
Arrhythmogenic Cardiomyopathy (ACM), a hereditary heart disease affecting approximately 1 in every 2000-5000 individuals globally, is set to undergo clinical trials for a groundbreaking AAV gene therapy. The condition is characterized by arrhythmias and may lead to sudden cardiac arrest. Current treatments involve anti-arrhythmic medications and implantable cardioverter-defibrillators (ICD), but these approaches only manage symptoms without addressing the root cause.
ACM is a progressive condition where cardiac tissue is gradually replaced by fatty tissue over time, leading to a deterioration of heart function and, eventually, heart failure. In severe cases, heart transplantation becomes the only viable option, but the probability of finding a suitable donor is exceedingly low. Therefore, there is an urgent need to develop effective treatments targeting the underlying causes of ACM.
Recently, researchers from Utrecht University Medical Center published a study titled “Therapeutic efficacy of AAV-mediated restoration of PKP2 in arrhythmogenic cardiomyopathy” in the journal Nature Cardiovascular Research.
The study lays the foundation for gene therapy for ACM using adeno-associated virus (AAV) delivery of the PKP2 gene. Clinical trials for this therapy are scheduled to commence in the United States in 2024.

ACM is often associated with mutations in desmosome-related genes, responsible for connecting adjacent cardiac cells and ensuring synchronized contractions for coordinated pumping of blood by the heart.
The most common mutation in ACM is in the PKP2 gene, which encodes plakophilin-2, a vital component of desmosomes. Patients with this gene mutation typically have lower levels of plakophilin-2 protein in their cardiac cells, weakening the connections between these cells and leading to the development of arrhythmias.
To address this, the research team aimed to develop gene therapy targeting PKP2 mutations to fundamentally treat ACM. Introducing the correct PKP2 gene into affected cardiac cells could potentially restore plakophilin-2 protein to normal levels, strengthening desmosomes and reducing the occurrence of arrhythmias.
Using experimental models of ACM, the team demonstrated that delivering the correct PKP2 gene to human cardiac cells derived from stem cells restored plakophilin-2 levels and improved their sodium ion conduction, crucial for the contraction ability of cardiac cells.
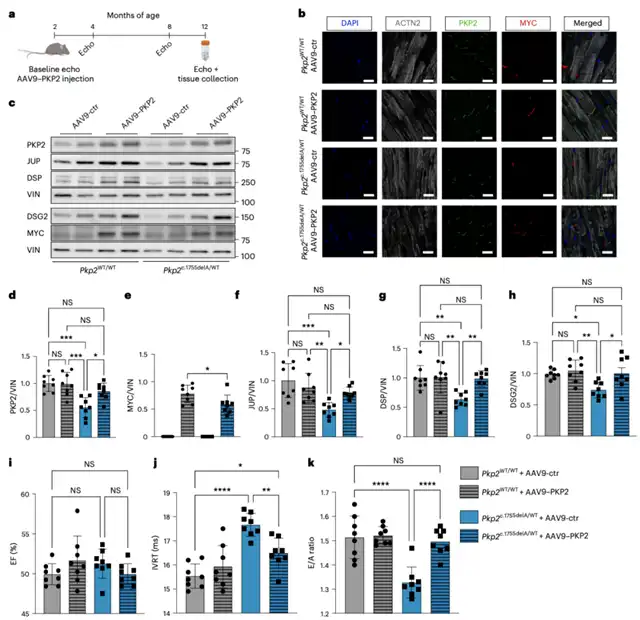
Subsequently, the team confirmed the therapeutic effect of this approach on the contractile force of engineered human cardiac cells in the laboratory. Finally, the researchers validated the efficacy of PKP2 gene therapy in a mouse model of ACM, successfully improving desmosomes and restoring cardiac function.
Following promising preclinical research progress, the next step is to explore the clinical treatment potential of this gene therapy in ACM patients carrying PKP2 gene mutations. Eirini Kyriakopoulou, the first author of the paper, announced that three US companies plan to initiate clinical trials next year to assess the therapeutic effects of this gene therapy in patients. While it remains uncertain whether this method can reverse existing cardiac damage in advanced stages of ACM, it may be effective in preventing the progression of early-stage disease to more severe phases.
Despite the significant hope brought by preclinical results and upcoming clinical trials for ACM, the true commercialization of this therapy may take several years. Besides confirming its effectiveness in human patients, any safety concerns must be addressed and resolved before clinical application.
Gene Therapy for Genetic Heart Disease to Begin Clinical Trials Soon
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



